Advanced Synthesis of Z-Configuration Allyl Amino Acid Derivatives for Pharmaceutical Applications
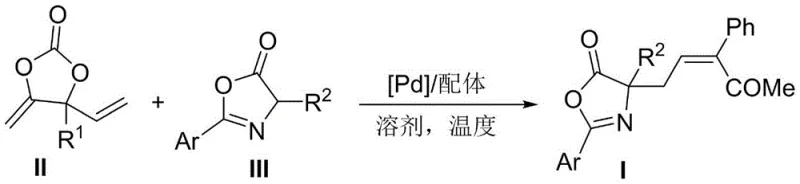
The pharmaceutical industry continuously seeks robust methodologies for constructing complex amino acid scaffolds, particularly those with defined stereochemistry that are often inaccessible through traditional routes. Patent CN115197161A introduces a groundbreaking approach for the synthesis of Z-configuration allyl amino acid derivatives, addressing a long-standing challenge in organic synthesis where thermodynamic stability typically favors the E-isomer. This innovation utilizes a palladium-catalyzed decarboxylative allylation strategy, coupling alkenyl cyclic ethylene carbonates with azlactones to generate quaternary carbon chiral centers with exceptional precision. The significance of this technology extends beyond academic interest, offering a practical pathway for producing high-purity pharmaceutical intermediates that serve as critical building blocks for next-generation therapeutics. By leveraging this novel catalytic system, manufacturers can access unique chemical space previously difficult to explore, thereby accelerating the discovery of new bioactive molecules.
Historically, the synthesis of allyl amino acids has been dominated by methods that yield the E-configuration due to lower steric hindrance and greater thermodynamic stability. Conventional approaches often struggle to control the geometry of the double bond, leading to mixtures that require tedious and costly separation processes, ultimately reducing overall process efficiency. Furthermore, existing methods frequently rely on harsh reaction conditions or expensive chiral auxiliaries that limit their scalability and economic viability for industrial applications. The inability to selectively access the Z-isomer has restricted the structural diversity available to medicinal chemists, potentially overlooking potent drug candidates that rely on this specific spatial arrangement for biological activity. This technological gap has necessitated the development of a more selective and operationally simple protocol that can reliably produce the Z-configuration without compromising yield or purity.
The novel approach disclosed in the patent overcomes these limitations by employing a specialized palladium catalytic system that directs the reaction trajectory towards the Z-isomer. By utilizing alkenyl cyclic ethylene carbonates as electrophiles and azlactones as nucleophiles, the reaction proceeds through a unique allyl-pi-allylpalladium intermediate that kinetically favors the formation of the Z-product. This method operates under mild conditions, typically between -10°C and 50°C, which preserves sensitive functional groups and reduces energy consumption compared to high-temperature alternatives. The use of readily available starting materials and a straightforward workup procedure involving column chromatography simplifies the manufacturing process, making it highly attractive for scale-up. This shift from thermodynamic control to kinetic control via catalyst design represents a significant advancement in synthetic methodology, enabling the efficient production of valuable amino acid derivatives.
Mechanistic Insights into Palladium-Catalyzed Decarboxylative Allylation
The core of this synthesis lies in the generation and reactivity of the pi-allylpalladium intermediate, which dictates the stereochemical outcome of the transformation. Upon activation by the palladium source, the alkenyl cyclic ethylene carbonate undergoes decarboxylation to form a highly reactive species that is subsequently intercepted by the azlactone nucleophile. The choice of ligand is critical in this process, as it modulates the electronic and steric environment around the metal center, thereby influencing the facial selectivity of the nucleophilic attack. Specifically, the use of bidentate phosphine ligands like L4 creates a rigid coordination sphere that stabilizes the transition state leading to the Z-configuration, effectively suppressing the formation of the E-isomer. This level of control is essential for maintaining high optical purity and ensuring that the resulting quaternary carbon center is formed with the correct absolute configuration required for downstream biological activity.
Impurity control is inherently built into the mechanism through the high stereoselectivity of the catalytic cycle, which minimizes the generation of geometric isomers that are difficult to separate. The reaction conditions are optimized to prevent side reactions such as polymerization or hydrolysis of the sensitive carbonate and azlactone moieties, ensuring a clean reaction profile. By maintaining a Z:E ratio greater than 20:1, the process significantly reduces the burden on downstream purification steps, leading to higher overall recovery of the desired product. Furthermore, the compatibility of the reaction with various substituted aryl and alkyl groups on both the carbonate and the azlactone allows for broad substrate scope without sacrificing selectivity. This robustness ensures that the impurity profile remains consistent and manageable across different batches, which is a critical requirement for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Z-Configuration Allyl Amino Acid Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls to maximize the efficiency of the palladium catalyst. The process begins with the dissolution of the alkenyl cyclic ethylene carbonate and the azlactone in a suitable organic solvent, with chlorobenzene identified as the optimal medium for achieving superior yields. Following this, the palladium source and the specific phosphine ligand are introduced to the mixture, initiating the catalytic cycle under controlled temperature conditions. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve alkenyl cyclic ethylene carbonate and azlactone in an organic solvent such as chlorobenzene.
- Add a palladium source like Pd(PPh3)2Cl2 and a phosphine ligand such as L4 to the reaction mixture.
- Stir the reaction at 0°C to 50°C, then separate and purify the product via column chromatography to obtain the Z-configuration derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis route offers substantial benefits by utilizing commercially available starting materials that are cost-effective and easy to source globally. The elimination of complex chiral resolving agents and the reduction in purification steps translate directly into lower manufacturing costs, making the final intermediates more competitive in the market. Additionally, the mild reaction conditions reduce the energy load on production facilities, contributing to a more sustainable and economically viable supply chain. These factors combined allow for a significant reduction in the cost of goods sold, providing a strategic advantage for companies looking to optimize their raw material expenses without compromising on quality.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot reaction eliminates the need for multiple protection and deprotection steps often required in traditional amino acid synthesis, thereby reducing reagent consumption and labor costs. By avoiding expensive chiral catalysts or resolution processes, the overall material cost is drastically lowered, allowing for more aggressive pricing strategies in the final API market. The high yield and selectivity further minimize waste generation, reducing the costs associated with waste disposal and environmental compliance. This economic efficiency makes the technology highly scalable and suitable for large-volume production runs.
- Enhanced Supply Chain Reliability: The reliance on stable and widely available reagents such as palladium salts and phosphine ligands ensures that supply chain disruptions are minimized, guaranteeing consistent production schedules. The robustness of the reaction against minor variations in temperature and concentration means that batch-to-batch variability is low, enhancing the reliability of supply for downstream customers. Furthermore, the simplicity of the workup procedure allows for faster turnaround times from synthesis to shipment, effectively reducing lead times for high-purity pharmaceutical intermediates. This reliability is crucial for maintaining continuous manufacturing operations in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are compatible with standard industrial reactor setups, facilitating a smooth transition from laboratory to commercial scale. The reduced generation of hazardous byproducts and the use of recyclable solvents align with green chemistry principles, simplifying regulatory approvals and environmental permitting. This compliance reduces the risk of production halts due to environmental violations and enhances the corporate sustainability profile. Consequently, manufacturers can scale up complex pharmaceutical intermediates with confidence, knowing that the process meets stringent global safety and environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for R&D and procurement teams evaluating this route. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What is the primary advantage of this synthesis method over conventional approaches?
A: Unlike conventional methods that predominantly yield thermodynamically stable E-configuration products, this novel palladium-catalyzed protocol achieves high stereoselectivity for the challenging Z-configuration, with Z:E ratios exceeding 20:1.
Q: What are the optimal reaction conditions for maximizing yield and selectivity?
A: The preferred conditions involve using Pd(PPh3)2Cl2 as the palladium source, Ligand L4 as the phosphine ligand, and chlorobenzene as the solvent at a temperature of 0°C, which balances high yield with exceptional stereocontrol.
Q: Can these derivatives be utilized for anticancer drug development?
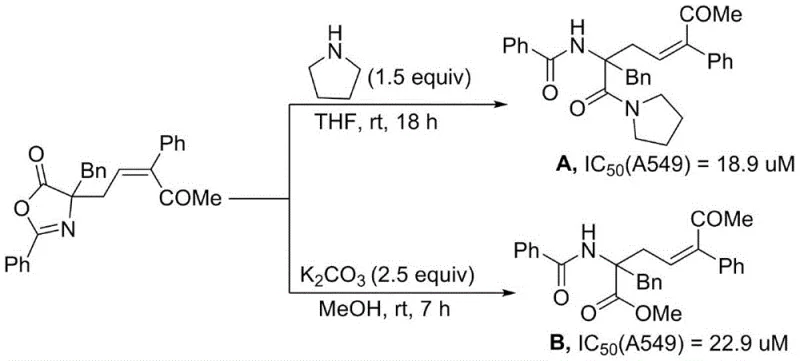
A: Yes, the patent demonstrates that specific derivatives synthesized via this route exhibit significant antitumor activity against A549 lung cancer cells, comparable to cisplatin, highlighting their potential as valuable scaffolds for oncology drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Z-Configuration Allyl Amino Acid Derivatives Supplier
As a leader in custom synthesis and process development, NINGBO INNO PHARMCHEM possesses the technical expertise to translate this innovative patent into commercial reality for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high stereoselectivity and purity demonstrated in the lab are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that the Z-configuration integrity is preserved throughout the manufacturing process, delivering intermediates that are ready for immediate use in drug development.
We invite potential partners to engage with our technical procurement team to discuss how this technology can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain insights into the specific economic benefits of adopting this route for your target molecules. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project needs. Let us help you overcome synthetic bottlenecks and secure a reliable supply of high-value amino acid derivatives for your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
