Advanced Organocatalytic Route for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
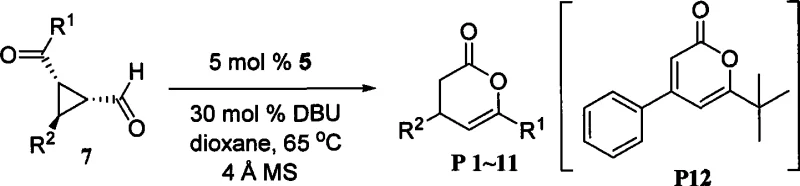
The pharmaceutical and fine chemical industries are constantly seeking robust, metal-free synthetic pathways to produce high-value heterocyclic scaffolds. Patent CN101481369A introduces a groundbreaking methodology for the efficient synthesis of 4,6-substituted 3,4-dihydro-pyran-2-one derivatives, a critical class of intermediates widely utilized in drug discovery and agrochemical development. This technology leverages the power of nitrogen-heterocyclic carbene (NHC) organocatalysis to facilitate the ring expansion of aldehyde-substituted cyclopropane compounds. Unlike traditional transition-metal catalyzed processes that often suffer from heavy metal contamination issues requiring costly removal steps, this novel approach operates under mild conditions using purely organic catalysts. The significance of this patent lies in its ability to generate complex lactone structures with high atom economy and exceptional functional group tolerance, addressing a long-standing challenge in the synthesis of 5-unsubstituted pyran-2-one derivatives.
Furthermore, the versatility of this catalytic system allows for the modification of substituents at the 4 and 6 positions, enabling the rapid generation of diverse chemical libraries for structure-activity relationship (SAR) studies. For procurement managers and supply chain directors, the elimination of precious metal catalysts represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. The process utilizes commercially available bases such as DBU and common organic solvents, simplifying the supply chain logistics and reducing the dependency on volatile metal markets. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a sustainable and economically viable alternative to legacy synthetic routes.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,6-substituted 3,4-dihydro-pyran-2-one derivatives has been plagued by significant technical hurdles that impact both yield and purity profiles. Conventional literature methods predominantly rely on strong acidic conditions or elevated temperatures to drive the cyclization of corresponding acids or esters. These harsh reaction environments often lead to the degradation of sensitive functional groups, resulting in complex impurity profiles that are difficult to separate during downstream processing. Moreover, many existing protocols require the presence of electron-withdrawing groups at the 5-position of the pyranone ring to facilitate the reaction, severely limiting the structural diversity of the accessible chemical space. This restriction forces medicinal chemists to design around synthetic limitations rather than optimizing for biological efficacy.
Additionally, traditional methods frequently employ transition metal catalysts which, while effective, introduce the risk of heavy metal residues in the final product. For pharmaceutical applications, removing these metal traces to meet stringent regulatory standards (often in the ppm range) necessitates additional purification steps such as specialized scavenging resins or repeated recrystallizations. These extra unit operations not only extend the production lead time but also drastically increase the overall cost of goods sold (COGS). The operational complexity associated with handling strong acids and high temperatures also poses safety risks in large-scale manufacturing environments, making these conventional routes less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN101481369A utilizes an N-heterocyclic carbene (NHC) catalytic system to achieve the ring expansion of aldehyde-substituted cyclopropanes under remarkably mild conditions. This organocatalytic strategy completely bypasses the need for strong acids or transition metals, thereby eliminating the associated safety hazards and contamination risks. The reaction proceeds efficiently at temperatures ranging from 25°C to 70°C, which is significantly lower than the thermal energy required by traditional acid-catalyzed condensations. This mildness preserves the integrity of sensitive substituents, allowing for the successful synthesis of derivatives containing halogens, ethers, and other labile functional groups that would otherwise decompose.
The core innovation lies in the use of NHC precursor salts, such as thiazolium or triazolium salts, which generate the active carbene species in situ upon treatment with a base like DBU. This catalytic cycle facilitates the nucleophilic attack on the cyclopropane ring, triggering a smooth ring expansion to form the desired six-membered lactone. The scope of this reaction is exceptionally broad, accommodating various aryl and alkyl substituents at the 4 and 6 positions without the mandatory requirement for electron-withdrawing groups at the 5-position. This flexibility empowers R&D teams to access previously difficult-to-synthesize chemical motifs, accelerating the drug discovery timeline. Furthermore, the workup procedure is straightforward, typically involving simple filtration and solvent evaporation, which streamlines the manufacturing process and enhances overall throughput.
Mechanistic Insights into NHC-Catalyzed Ring Expansion
The mechanistic pathway of this transformation is a testament to the elegance of organocatalysis, relying on the unique electronic properties of the nitrogen-heterocyclic carbene. The reaction initiates with the deprotonation of the NHC precursor salt by a base, such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), to generate the free carbene species. This nucleophilic carbene then attacks the aldehyde moiety of the cyclopropane substrate, forming a Breslow intermediate. This key intermediate undergoes a polarity inversion (umpolung), rendering the carbonyl carbon nucleophilic. Subsequently, the electron-rich center attacks the strained cyclopropane ring, inducing ring opening and subsequent intramolecular cyclization to form the six-membered dihydro-pyran-2-one core. The catalyst is then eliminated to regenerate the active species and release the final product.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Since the catalytic cycle does not involve redox-active metal centers, there is no risk of metal-mediated side reactions such as over-oxidation or uncontrolled radical polymerization. The specificity of the NHC for the aldehyde functionality ensures high chemoselectivity, minimizing the formation of byproducts derived from other reactive sites on the molecule. Additionally, the use of 4 Å molecular sieves in the reaction mixture, as recommended in the patent examples, effectively scavenges trace water that could otherwise hydrolyze the intermediate or deactivate the catalyst. This attention to moisture control ensures consistent reaction performance and high isolated yields, often exceeding 90% for optimal substrates like 4,6-diphenyl-3,4-dihydropyran-2-one.
How to Synthesize 4,6-Substituted 3,4-Dihydro-pyran-2-one Efficiently
The practical implementation of this synthesis route is designed for reproducibility and ease of operation, making it highly suitable for both laboratory scale optimization and pilot plant production. The standard protocol involves dissolving the aldehyde-substituted cyclopropane substrate in a dry polar aprotic solvent such as 1,4-dioxane or tetrahydrofuran. To this solution, a catalytic amount of the NHC precursor salt (typically 5 mol%) and a stoichiometric amount of the base (typically 30 mol% DBU) are added. The inclusion of activated 4 Å molecular sieves is critical to maintain anhydrous conditions throughout the reaction course. The mixture is then heated to approximately 65°C and stirred under an inert atmosphere until conversion is complete, as monitored by TLC or HPLC.
- Prepare the reaction mixture by combining 2-acyl-1-formylcyclopropane substrate, thiazolium salt catalyst precursor (5 mol%), and DBU base (30 mol%) in dry 1,4-dioxane.
- Add 4 Å molecular sieves to the reaction vessel to maintain anhydrous conditions and stir the mixture at 65°C under argon protection.
- Upon completion, filter through celite, wash with dichloromethane, evaporate solvent, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage is the drastic simplification of the raw material supply chain. By eliminating the need for expensive and supply-constrained transition metal catalysts, manufacturers can mitigate the risks associated with metal price volatility and geopolitical supply disruptions. The reagents used in this process, including the NHC precursor salts and organic bases, are commodity chemicals that are readily available from multiple global suppliers, ensuring a stable and continuous supply of critical inputs for production schedules.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free process is profound. Traditional methods often incur hidden costs related to the procurement of precious metals and the subsequent validation of metal clearance procedures. By removing these requirements, the new method significantly reduces the cost of raw materials and quality control testing. Furthermore, the mild reaction conditions (65°C) consume less energy compared to high-temperature reflux processes, contributing to lower utility costs per kilogram of product. The simplified workup procedure, which avoids complex extraction or scavenging steps, also reduces labor hours and solvent consumption, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, where delays can halt clinical trials or commercial launches. This synthesis route enhances reliability by utilizing robust, non-hazardous reagents that are easy to transport and store. The absence of heavy metals simplifies the regulatory filing process, as there is no need to submit extensive data on metal residue limits and clearance validation to health authorities. This streamlined regulatory pathway accelerates the time-to-market for new drug candidates. Additionally, the high yields and purity achieved reduce the need for re-processing batches due to quality failures, ensuring a more predictable and consistent output of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often reveals hidden bottlenecks, but this organocatalytic method is inherently scalable. The reaction exotherm is manageable due to the mild conditions, reducing the engineering controls required for heat dissipation in large reactors. From an environmental perspective, the process aligns with green chemistry principles by avoiding toxic heavy metals and reducing waste generation. The solvents used, such as dioxane, can be efficiently recovered and recycled, minimizing the environmental footprint. This compliance with increasingly strict environmental regulations protects the company from potential fines and enhances its corporate social responsibility profile, making it a preferred partner for sustainability-conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for R&D and operations teams evaluating this process for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the primary advantages of this NHC-catalyzed method over traditional acid-catalyzed routes?
A: Unlike conventional methods requiring strong acidic conditions or high temperatures which often lead to decomposition, this NHC-catalyzed process operates under mild conditions (25°C to 70°C) and eliminates heavy metal residues, ensuring higher purity suitable for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process utilizes readily available organic solvents like dioxane and common bases like DBU, avoiding expensive transition metal catalysts. The simple workup procedure involving filtration and standard chromatography supports robust commercial scale-up.
Q: What is the substrate scope for this ring expansion reaction?
A: The method demonstrates broad compatibility with various substituted aryl groups and alkyl chains at the 4 and 6 positions of the pyran-2-one ring, allowing for the synthesis of diverse derivatives without requiring electron-withdrawing groups at the 5-position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Substituted 3,4-Dihydro-pyran-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic technologies in modern drug synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN101481369A can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4,6-substituted 3,4-dihydro-pyran-2-one derivatives meets the highest quality standards required for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this metal-free synthesis route can optimize your supply chain and reduce costs. Contact our Customized Cost-Saving Analysis department today to discuss your specific project requirements. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules, helping you accelerate your development timeline with confidence and efficiency.
