Advanced Synthesis of Pyrazole-Modified Fused-Ring Arylimides for High-Performance Optoelectronic Materials
The landscape of advanced optoelectronic materials is undergoing a significant transformation, driven by the urgent need for compounds that offer superior stability, high electron mobility, and versatile coordination capabilities. Patent CN110818718A introduces a groundbreaking class of fused-ring arylimide derivative compounds that address these critical demands through a novel molecular architecture. These derivatives, characterized by a rigid planar macroconjugated structure modified with pyrazole ligands, represent a substantial leap forward in the design of functional materials for solar cells, organic light-emitting diodes (OLEDs), and coordination chemistry. The core innovation lies in the strategic integration of pyrazole rings onto the fused-ring arylimide backbone, which not only enhances the photoresponse properties and chemical stability but also introduces multifunctional coordination sites that were previously unattainable with standard monodentate ligands. This technical breakthrough provides a robust foundation for developing next-generation electronic chemicals that require precise control over molecular assembly and electronic characteristics.
For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, the implications of this patent are profound. The ability to synthesize these complex structures via a streamlined one-step process significantly lowers the barrier to entry for high-performance material production. The compounds described in Formula I, II, and III demonstrate exceptional potential for application in fields ranging from fluorescence sensing to anti-tumor drug development, showcasing the versatility of this chemical platform. By leveraging the unique acidic nature of the nitrogen-hydrogen bonds within the pyrazole moiety, these derivatives can participate in complex coordination behaviors, releasing protons under specific conditions to form stable complexes with various metal ions. This dual functionality makes them an invaluable asset for researchers aiming to push the boundaries of material science and commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functional arylimide derivatives has been constrained by the reliance on monodentate ligands, which inherently limit the structural diversity and functional output of the resulting complexes. These traditional ligands typically possess a single coordination site, leading to complexes with simple, often linear or planar structures that lack the three-dimensional complexity required for advanced optoelectronic applications. The simplicity of their coordination form restricts the ability to fine-tune the electronic properties of the material, resulting in suboptimal performance in devices such as organic photovoltaics and light-emitting diodes. Furthermore, conventional methods often involve multi-step synthetic routes that require harsh reaction conditions, expensive catalysts, and tedious purification processes, which collectively drive up production costs and extend lead times. The inability to introduce rich coordination points means that the resulting materials often suffer from poor solubility and limited processability, creating significant bottlenecks for supply chain heads looking to scale production for commercial markets.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a pyrazole-modified fused-ring system that fundamentally alters the coordination landscape. By incorporating pyrazole ligands, which are complex organic bridging ligands, the new derivatives offer multiple coordination sites and the ability to form hydrogen bonds that stabilize specific conformations. This structural innovation allows for the design of compounds with varying coordination angles and shapes, enabling the creation of materials with tailored fluorescence, catalytic, and electronic properties. The synthesis strategy is remarkably efficient, relying on a direct condensation reaction between tetracarboxylic dianhydrides and aminopyrazoles, which eliminates the need for intermediate protection and deprotection steps. This simplification not only accelerates the development cycle but also enhances the overall yield, with reported efficiencies consistently exceeding 80%. The result is a high-purity fused-ring arylimide that is ready for immediate integration into sophisticated electronic devices.
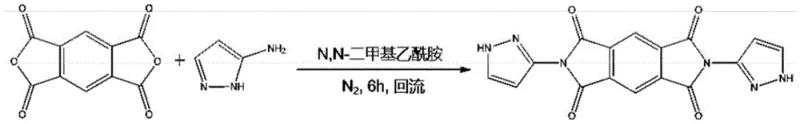
Mechanistic Insights into Pyrazole-Modified Imide Condensation
The chemical mechanism underpinning this synthesis is a classic yet highly optimized nucleophilic acyl substitution followed by cyclodehydration. In the case of the pyromellitic anhydride derivative (Formula I), the reaction initiates with the nucleophilic attack of the amino group from the aminopyrazole on the carbonyl carbon of the anhydride. This step is facilitated by the use of polar aprotic solvents like N,N-dimethylacetamide (DMAc), which stabilize the transition state and ensure complete dissolution of the reactants. The subsequent ring closure to form the imide structure is driven by thermal energy under reflux conditions, typically maintained for around 6 hours. The presence of the pyrazole ring does not interfere with this imidization process; rather, its weak acidity and hydrogen bonding capability can influence the solubility and crystallization behavior of the final product. This mechanistic pathway ensures that the rigid planar structure of the fused ring is preserved, which is critical for maintaining the high molar absorption coefficient and electron mobility required for optoelectronic performance.
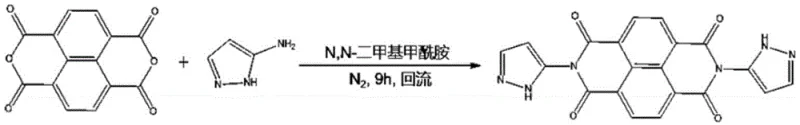
When scaling this mechanism to larger fused-ring systems such as the naphthalene and perylene derivatives (Formulas II and III), slight modifications to the reaction conditions are necessary to accommodate the increased steric hindrance and reduced solubility of the larger anhydride cores. For the naphthalene-based synthesis, the reaction time is extended to approximately 9 hours in N,N-dimethylformamide (DMF) to ensure complete conversion. For the perylene-based system, which is particularly relevant for high-end OLED material manufacturing, the process employs imidazole as both a solvent and a reaction medium, often in conjunction with a zinc acetate catalyst. This catalytic assistance is crucial for overcoming the activation energy barrier associated with the bulky perylene core, allowing the reaction to proceed efficiently at temperatures between 100°C and 160°C. The careful control of these parameters ensures that the final product retains its structural integrity and optoelectronic functionality, providing a reliable pathway for cost reduction in electronic chemical manufacturing.

How to Synthesize Pyrazole-Modified Fused-Ring Arylimides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the specified molar ratios and atmospheric conditions to guarantee reproducibility and high purity. The process begins with the preparation of the anhydride solution under a nitrogen blanket to prevent oxidation, followed by the controlled addition of the aminopyrazole reagent. The reaction mixture is then subjected to prolonged heating to drive the imidization to completion, after which the product is isolated through filtration and washing with appropriate solvents like dichloromethane or methanol. This straightforward protocol minimizes the generation of hazardous waste and reduces the consumption of energy compared to multi-step alternatives. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized synthesis steps provided below.
- Prepare the reaction vessel under nitrogen protection and dissolve the specific tetracarboxylic dianhydride (pyromellitic, naphthalene, or perylene) in a high-boiling polar aprotic solvent such as N,N-dimethylacetamide or imidazole.
- Introduce the aminopyrazole ligand (3-aminopyrazole, 4-aminopyrazole, or 5-methyl-3-aminopyrazole) to the reaction mixture, ensuring a molar ratio of anhydride to amine between 1: 2 and 1:3 for complete imidization.
- Heat the mixture to reflux temperatures ranging from 100°C to 160°C for 6 to 20 hours depending on the anhydride core, followed by cooling, filtration, and washing to isolate the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis technology offers substantial benefits for procurement managers and supply chain heads focused on efficiency and cost control. The primary advantage lies in the drastic simplification of the production process, which translates directly into lower operational expenditures and reduced dependency on complex supply chains for specialized reagents. By utilizing cheap and easily obtainable raw materials such as pyromellitic anhydride and aminopyrazoles, manufacturers can significantly mitigate the risk of raw material price volatility. Furthermore, the high yield nature of the reaction ensures that material throughput is maximized, reducing the cost per kilogram of the final active pharmaceutical ingredient or electronic chemical. This efficiency is critical for maintaining competitive pricing in the global market while ensuring that supply continuity is not compromised by low-yield batch failures.
- Cost Reduction in Manufacturing: The elimination of multi-step synthetic sequences and the avoidance of expensive transition metal catalysts (in most cases) lead to a streamlined production workflow that inherently lowers manufacturing costs. The use of common organic solvents and standard reflux equipment means that existing infrastructure can often be utilized without the need for capital-intensive upgrades. Additionally, the high conversion rates minimize the loss of valuable starting materials, ensuring that the overall material balance is optimized for economic production. This logical deduction of cost savings through process intensification makes the technology highly attractive for large-scale commercial operations.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis of these advanced derivatives ensures a robust and resilient supply chain. Since the key precursors are widely available from multiple global suppliers, the risk of single-source bottlenecks is significantly reduced. The simplicity of the post-processing steps, which involve basic filtration and washing rather than complex chromatography, further accelerates the turnaround time from reaction completion to final product shipment. This agility allows supply chain managers to respond more quickly to market demands and maintain consistent inventory levels for high-purity solar cell materials and other critical applications.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates easy scale-up from gram-scale laboratory synthesis to ton-scale industrial production without significant re-optimization. The process generates minimal byproducts, and the solvents used can often be recovered and recycled, aligning with modern environmental compliance standards and sustainability goals. The ability to produce these complex fused-ring arylimides with high purity and consistency supports the commercial scale-up of complex electronic chemicals, ensuring that the environmental footprint of the manufacturing process remains manageable and compliant with international regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fused-ring arylimide derivatives. The answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the technology's capabilities. These insights are intended to assist decision-makers in evaluating the feasibility of integrating these materials into their existing product lines or research pipelines. By clarifying the coordination properties, yield expectations, and scalability potential, we aim to eliminate uncertainties and foster confidence in the technology's commercial viability.
Q: What distinguishes these pyrazole-modified derivatives from conventional monodentate ligands?
A: Unlike conventional monodentate ligands which offer limited coordination sites and simple structures, these pyrazole-modified fused-ring arylimides possess rich coordination sites due to the nitrogen-hydrogen bonds on the pyrazole ring. This allows for the formation of complexes with diverse angles, shapes, and coordination numbers, significantly enhancing their utility in coordination chemistry and optoelectronic applications.
Q: What are the typical yields and purity levels achievable with this synthesis method?
A: The patent data indicates that this one-step condensation method consistently achieves high yields exceeding 80%, with specific examples demonstrating yields up to 91%. The post-processing purification is described as simple, involving filtration and washing, which facilitates the production of high-purity materials suitable for sensitive electronic applications like OLEDs and solar cells.
Q: Can this synthesis process be scaled for industrial production of electronic chemicals?
A: Yes, the method is explicitly designed for industrial scalability. It utilizes cheap and easily obtainable raw materials, involves ordinary organic synthesis steps without complex catalytic systems (except for specific perylene derivatives), and operates under standard nitrogen protection and reflux conditions, making it highly suitable for commercial scale-up of complex electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused-Ring Arylimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the pyrazole-modified fused-ring arylimide technology and are fully equipped to support its transition from patent to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are designed to handle the specific solvent systems and thermal conditions required for this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the optoelectronic industry. We understand that consistency is key in electronic material manufacturing, and our quality management systems are tailored to deliver high-purity fused-ring arylimides that perform reliably in your devices.
We invite you to collaborate with us to optimize your supply chain and unlock the full value of this innovative chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that evaluates how this synthesis route can reduce your overall production expenses. We encourage you to reach out to request specific COA data and route feasibility assessments that are tailored to your specific project requirements. By partnering with us, you gain access to a wealth of technical expertise and manufacturing capacity that will accelerate your time-to-market and strengthen your competitive position in the global electronics sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
