Advancing Ester Reduction Technology with Novel Monovalent Magnesium Catalysts for Commercial Scale-up
Advancing Ester Reduction Technology with Novel Monovalent Magnesium Catalysts for Commercial Scale-up
The landscape of organic synthesis is undergoing a significant transformation with the emergence of main group metal catalysis, specifically highlighted by the groundbreaking technology disclosed in patent CN108276433B. This intellectual property introduces a revolutionary hydroboration method for esters utilizing a beta-diimine monovalent magnesium compound, marking a departure from conventional transition metal-dependent processes. The core innovation lies in the utilization of a magnesium-magnesium bonded species that acts as a potent two-center, two-electron reductant, enabling the efficient conversion of esters to alcohols or boronic esters under remarkably mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this represents a paradigm shift towards safer, more sustainable, and cost-effective manufacturing protocols. The ability to achieve high conversion rates without the need for expensive noble metals or hazardous high-pressure hydrogenation setups positions this technology as a critical asset for modern supply chains seeking resilience and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of esters to alcohols has relied heavily on stoichiometric hydride reagents such as lithium aluminum hydride (LiAlH4) or lithium borohydride (LiBH4), which, while effective, present severe safety hazards including pyrophoricity and poor functional group tolerance. Alternatively, transition metal-catalyzed hydrogenation requires elevated temperatures and high pressures, leading to significant energy consumption and potential equipment corrosion issues that complicate reactor maintenance. Furthermore, the use of late transition metals like ruthenium, rhodium, or palladium introduces the risk of heavy metal residue in the final product, necessitating costly and time-consuming purification steps to meet stringent pharmaceutical purity standards. These traditional methodologies often suffer from limited substrate scope, particularly when dealing with sterically hindered or sensitive functional groups, thereby restricting their utility in the synthesis of complex pharmaceutical intermediates where selectivity is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a beta-diimine monovalent magnesium catalyst that operates with exceptional activity at room temperature, eliminating the need for thermal input or pressurized reactors. This method utilizes pinacol borane as the reducing agent, which reacts rapidly with the ester substrate in the presence of the magnesium catalyst to achieve yields reaching up to 99% within a mere 10-minute timeframe. The catalytic system exhibits broad substrate universality, successfully processing diverse esters ranging from simple ethyl acetate to complex structures like lactide and benzyl benzoate without compromising efficiency. By shifting from stoichiometric reagents to a catalytic cycle driven by earth-abundant magnesium, this technology drastically simplifies the operational workflow and reduces the environmental footprint associated with waste disposal and energy usage.
Mechanistic Insights into Mg-Mg Bond Catalyzed Hydroboration
The mechanistic foundation of this transformative process rests on the unique electronic structure of the beta-diimine monovalent magnesium compound, which features a direct magnesium-magnesium bond capable of activating small molecules through oxidative addition or sigma-bond metathesis pathways. Unlike traditional Grignard reagents that act as nucleophiles, this monovalent species functions as a powerful reductant, facilitating the cleavage of the boron-hydrogen bond in pinacol borane and subsequent transfer of hydride equivalents to the carbonyl carbon of the ester. The catalytic cycle is sustained by the regeneration of the active magnesium species, allowing for high turnover numbers that are uncharacteristic of main group elements, which typically require stoichiometric quantities for similar transformations. This mechanistic efficiency ensures that the reaction proceeds with minimal catalyst loading, typically at a molar ratio of ester to catalyst of 20:1, yet maintains rapid kinetics that drive the reaction to completion almost instantaneously.

Impurity control is inherently superior in this system due to the absence of transition metals, which are notorious for forming difficult-to-remove organometallic byproducts that can poison downstream biological assays or catalytic steps. The use of magnesium, a biocompatible and easily removable metal, simplifies the workup procedure to basic aqueous extraction or filtration, ensuring that the final high-purity pharmaceutical intermediates meet rigorous quality specifications without the need for specialized scavenger resins. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups such as halides or alkenes, which might otherwise undergo unwanted side reactions under the harsh conditions of hydride reductions or high-temperature hydrogenation. This selectivity profile is crucial for the synthesis of complex drug candidates where preserving stereochemistry and functional group integrity is non-negotiable for biological activity.
How to Synthesize Beta-Diimine Monovalent Magnesium Compound Efficiently
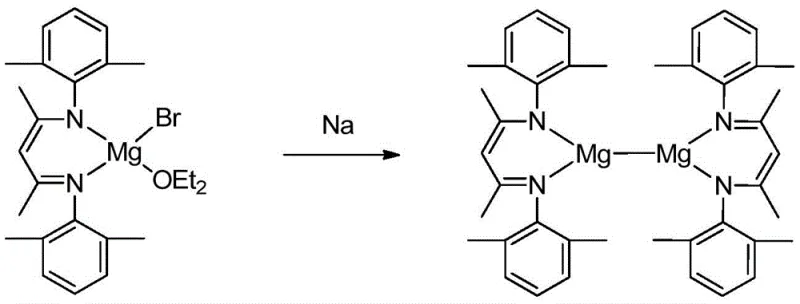
The preparation of the active catalyst involves a straightforward two-step synthetic sequence that begins with the formation of a magnesium bromide intermediate followed by a reduction step to generate the Mg-Mg bonded species. The process is designed to be robust and scalable, utilizing readily available starting materials such as beta-diimine ligands and methylmagnesium bromide, which are standard commodities in the fine chemical industry. The initial step requires careful temperature control during the addition of the Grignard reagent to ensure the formation of the correct coordination geometry around the magnesium center, setting the stage for the subsequent reduction. This synthetic route avoids the use of exotic reagents or extreme conditions, making it accessible for commercial scale-up in standard glass-lined or stainless steel reactors equipped with inert atmosphere capabilities.
- Dissolve beta-diimine ligand in ether, add methylmagnesium bromide at low temperature (-80 to -40°C), and react at room temperature to form the magnesium bromide intermediate.
- Dissolve the magnesium bromide intermediate in toluene and react with a sodium mirror under anhydrous conditions for 3 to 5 days to yield the yellow beta-diimine monovalent magnesium crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this magnesium-catalyzed hydroboration technology offers substantial cost reduction in fine chemical manufacturing by eliminating the dependency on volatile and expensive transition metal catalysts. The removal of precious metals like palladium or rhodium from the supply chain not only lowers raw material costs but also mitigates the geopolitical risks associated with the sourcing of these critical minerals, ensuring a more stable and predictable supply line for production planning. Additionally, the dramatic reduction in reaction time from hours or days to mere minutes translates into increased reactor throughput, allowing manufacturers to produce larger volumes of intermediates using existing infrastructure without the need for capital-intensive expansion. This efficiency gain directly impacts the bottom line by reducing utility costs and labor hours per batch, making the process economically attractive for both pilot-scale development and full-scale commercial production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in energy consumption due to room temperature operation lead to significant operational expenditure savings. By avoiding the need for high-pressure hydrogenation equipment and the associated safety protocols, facilities can lower their insurance premiums and maintenance costs while extending the lifespan of their reactor assets. The high atom economy of the hydroboration reaction further minimizes waste generation, reducing the costs associated with solvent recovery and hazardous waste disposal, which are increasingly regulated and taxed in global markets.
- Enhanced Supply Chain Reliability: Utilizing magnesium, an abundant and widely available element, decouples the production process from the supply constraints often faced with rare earth or platinum group metals. The simplicity of the reagent profile, relying on common solvents like toluene and ethers, ensures that raw material availability remains high even during market fluctuations, preventing production delays caused by supply shortages. This reliability is critical for maintaining continuous manufacturing schedules and meeting tight delivery deadlines for downstream pharmaceutical customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The benign nature of the magnesium catalyst and the absence of toxic heavy metals simplify the regulatory approval process for new drug applications, as residual metal limits are easier to satisfy. The process generates minimal hazardous waste, aligning with green chemistry principles and corporate sustainability goals, which are becoming increasingly important criteria for supplier selection by major multinational corporations. The scalability of the reaction from gram to kilogram scales has been demonstrated in the patent examples, providing confidence that the technology can be seamlessly transferred to multi-ton production facilities without loss of efficiency or selectivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel hydroboration technology in industrial settings. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for process evaluation and feasibility studies. Understanding these details is essential for technical teams assessing the integration of this method into existing synthetic routes for API intermediates or fine chemical products.
Q: What are the advantages of using monovalent magnesium catalysts over traditional hydride reagents?
A: Unlike traditional hydrides like LiAlH4 which pose fire hazards and selectivity issues, the monovalent magnesium catalyst operates safely at room temperature with high chemoselectivity and eliminates heavy metal contamination risks associated with transition metal catalysts.
Q: What is the reaction efficiency of this hydroboration method?
A: The patent data demonstrates exceptional efficiency, achieving up to 99% yield for various ester substrates including ethyl acetate and lactide within just 10 minutes at room temperature.
Q: Is this catalyst suitable for large-scale pharmaceutical manufacturing?
A: Yes, the mild reaction conditions (room temperature, ambient pressure) and the use of earth-abundant magnesium make this process highly scalable and economically viable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Diimine Magnesium Compound Supplier
As a leader in the custom synthesis and manufacturing of advanced chemical intermediates, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of this cutting-edge magnesium-catalyzed technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the quality of every batch. Our commitment to excellence means that we can deliver the high-purity beta-diimine ligands and magnesium precursors required for this process with the consistency and reliability that global supply chains demand.
We invite you to engage with our technical procurement team to discuss how this innovative hydroboration method can optimize your current manufacturing processes and reduce overall production costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecules and volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to tailor our solutions to your unique chemical challenges and help you achieve your strategic sourcing objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
