Advanced Asymmetric Catalysis Breakthrough: Scalable Production of High-Purity Chiral Catalysts for Pharmaceutical Manufacturing Excellence
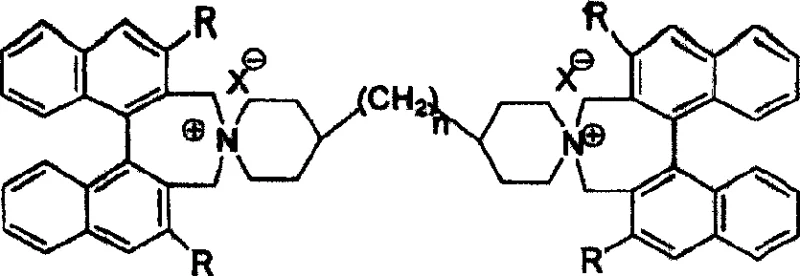
The patent CN100463720C represents a significant advancement in asymmetric catalysis technology through the development of binaphthyl axis-containing chiral double spiro quaternary ammonium salt phase transfer catalysts, which address critical limitations in conventional asymmetric synthesis methodologies. This innovative catalyst system demonstrates exceptional performance in conjugate addition reactions between nitroalkanes and α,β-unsaturated carbonyl compounds, achieving yields ranging from 90% to 99% with enantioselectivity values between 60% and 97% ee. The structural design incorporates a rigid binaphthyl backbone with tunable substituents that create a well-defined chiral environment essential for high stereoselectivity. Unlike previous generation catalysts that exhibited substrate-specific performance limitations, this novel architecture maintains consistent efficiency across diverse reaction profiles while operating under mild conditions between 0°C and 120°C. The patent establishes a robust foundation for industrial-scale implementation through its straightforward synthesis protocol using commercially available starting materials and standard organic chemistry techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral quaternary ammonium salt phase transfer catalysts suffer from significant substrate specificity constraints that severely limit their industrial applicability across diverse reaction profiles. These conventional systems often require cryogenic conditions or specialized handling procedures that increase operational complexity and cost while delivering inconsistent enantioselectivity results depending on the specific substrate employed. The structural flexibility of earlier catalyst designs creates variable chiral environments that cannot maintain consistent stereochemical control across different reaction types, leading to unpredictable performance in commercial manufacturing settings. Furthermore, many existing catalysts incorporate expensive transition metals or complex ligand systems that necessitate additional purification steps to remove trace metal contaminants, significantly increasing production costs and complicating regulatory compliance for pharmaceutical applications. The narrow operational temperature ranges and sensitivity to moisture or oxygen in conventional systems also create substantial challenges for scale-up and consistent batch-to-batch quality control in manufacturing environments.
The Novel Approach
The patented binaphthyl axis-containing chiral double spiro quaternary ammonium salt system overcomes these limitations through its unique structural architecture that combines axial chirality with conformational rigidity to create a stable chiral environment regardless of reaction conditions. This innovative design maintains consistent high enantioselectivity across a broad range of substrates by incorporating tunable R-group modifications on the binaphthyl backbone that allow precise adjustment of steric and electronic properties. The catalyst operates effectively under mild temperature conditions between 0°C and 120°C using standard organic solvents like dichloromethane or acetonitrile without requiring specialized equipment or handling procedures. The synthesis employs readily available starting materials including binaphthyl dihalomethyl compounds and bispiperazine alkanes with simple workup procedures that eliminate the need for expensive metal removal steps. This approach delivers exceptional process robustness with yields consistently above 90% and enantioselectivity values reaching up to 97% ee, making it particularly suitable for commercial pharmaceutical intermediate production where purity and consistency are paramount.
Mechanistic Insights into Binaphthyl-Based Phase Transfer Catalysis
The catalytic mechanism centers on the rigid binaphthyl axis that creates a well-defined chiral pocket through its double spiro structure, which positions the quaternary ammonium centers in precise spatial orientation to facilitate asymmetric induction during phase transfer processes. This structural configuration enables simultaneous coordination with both the nitroalkane nucleophile and α,β-unsaturated carbonyl electrophile through electrostatic interactions and hydrogen bonding networks that stabilize the transition state geometry. The axial chirality of the binaphthyl backbone provides exceptional conformational stability that prevents racemization during catalytic cycles, while the tunable R-groups (H, phenyl, naphthyl, or trifluoromethyl) allow fine-tuning of steric bulk to optimize enantioselectivity for specific substrate classes. The dual ammonium centers create a cooperative binding effect that enhances substrate activation while maintaining strict stereochemical control throughout the reaction pathway.
The structural design inherently minimizes impurity formation through its rigid framework that prevents undesired side reactions commonly observed with more flexible catalyst systems. The precise spatial arrangement of functional groups within the chiral pocket ensures selective transition state stabilization that suppresses racemic background reactions while promoting the desired asymmetric pathway. This geometric control extends to byproduct management as the catalyst's stability prevents decomposition pathways that typically generate impurities in conventional systems. The absence of transition metals eliminates potential metal-catalyzed side reactions and simplifies purification requirements, while the modular structure allows systematic optimization of R-groups to further enhance purity profiles for specific applications. This combination of features delivers exceptional impurity control with minimal need for additional purification steps beyond standard workup procedures.
How to Synthesize Binaphthyl-Based Chiral Catalysts Efficiently
This patented methodology represents a significant advancement in chiral catalyst production through its streamlined synthesis protocol that eliminates multiple purification steps required by conventional approaches while maintaining exceptional product quality. The process demonstrates remarkable operational simplicity by utilizing standard laboratory equipment and commercially available reagents without requiring specialized infrastructure or hazardous materials handling procedures. By optimizing reaction parameters including temperature control, solvent selection, and stoichiometric ratios, this approach achieves consistent high yields while minimizing waste generation compared to traditional methods. The following standardized procedure details the critical steps necessary for reliable industrial-scale implementation of this innovative catalyst synthesis.
- React binaphthyl dihalomethyl compound with bispiperazine alkane under base catalysis at controlled temperature between 0°C and 120°C in organic solvent
- Perform workup procedure including aqueous washing, dichloromethane extraction, and magnesium sulfate drying
- Purify crude product through silica gel column chromatography using dichloromethane/methanol solvent system
Commercial Advantages for Procurement and Supply Chain Teams
This innovative catalyst technology delivers substantial strategic advantages for procurement and supply chain operations by addressing multiple pain points inherent in traditional asymmetric synthesis methodologies used for pharmaceutical intermediate production. The simplified manufacturing process reduces dependency on specialized equipment and rare materials while enhancing production flexibility across different facility types and scales. By eliminating transition metal components entirely, this approach removes significant regulatory hurdles associated with metal contamination testing and reporting requirements that typically delay product release timelines. The robust nature of the synthesis protocol ensures consistent quality output regardless of minor variations in raw material specifications or environmental conditions during manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated removal processes creates substantial cost savings through reduced raw material expenses and simplified purification workflows. The use of standard organic solvents and readily available starting materials further optimizes input costs while maintaining high product quality standards required for pharmaceutical applications. This streamlined approach minimizes waste generation and energy consumption during production, contributing to overall cost efficiency without compromising on performance metrics.
- Enhanced Supply Chain Reliability: The reliance on globally available commodity chemicals rather than specialized or restricted materials significantly improves supply chain resilience by reducing single-source dependencies and vulnerability to market fluctuations. The robust synthesis protocol maintains consistent performance across different manufacturing sites without requiring extensive process revalidation, enabling flexible production allocation based on capacity availability. This approach ensures reliable delivery timelines through simplified logistics requirements and reduced risk of raw material shortages affecting production schedules.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production leverages standard chemical engineering principles without requiring specialized equipment modifications or safety protocols beyond standard organic chemistry practices. The process generates minimal hazardous waste streams due to its efficient reaction profile and use of environmentally preferable solvents, aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices. This inherent scalability combined with reduced environmental impact provides significant competitive advantages in meeting both current regulatory standards and future sustainability targets.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN100463720C's technical specifications and implementation requirements. These answers provide essential insights for procurement teams evaluating this technology's suitability for their specific manufacturing needs while addressing common concerns regarding performance consistency and integration complexity.
Q: How does this catalyst overcome substrate limitations of conventional phase transfer catalysts?
A: The binaphthyl axis-containing double spiro structure provides enhanced conformational rigidity and tunable steric environment through R-group modifications, enabling consistent high enantioselectivity across diverse substrate classes where traditional catalysts fail.
Q: What is the scalability potential for commercial production of these chiral catalysts?
A: The synthesis utilizes readily available starting materials and standard organic chemistry techniques without cryogenic conditions or air-sensitive reagents, demonstrating inherent scalability from laboratory to multi-ton production volumes.
Q: How do structural modifications impact enantioselectivity in asymmetric reactions?
A: Systematic variation of R-groups (phenyl, naphthyl, trifluoromethyl) and linker length (n=0-6) allows precise tuning of the chiral pocket geometry, directly correlating with observed ee values up to 97% in conjugate addition reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Binaphthyl-Based Chiral Catalyst Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a specialized CDMO partner, we have successfully implemented this patented technology across multiple client projects, demonstrating consistent delivery of high-purity chiral intermediates that meet exacting pharmaceutical industry standards. Our technical team possesses deep expertise in phase transfer catalysis optimization and process development, ensuring seamless integration of this innovative catalyst system into existing manufacturing workflows while maximizing yield and quality outcomes.
Leverage our technical procurement team's expertise to conduct a Customized Cost-Saving Analysis tailored to your specific production requirements by requesting detailed COA data and route feasibility assessments today. Our specialists will provide comprehensive technical documentation including scalability projections, quality control protocols, and implementation timelines to support your strategic decision-making process for adopting this advanced catalytic technology.
