Advanced Aralkyl-Tetrahydro-Pyridine Synthesis for Commercial Pharmaceutical Manufacturing
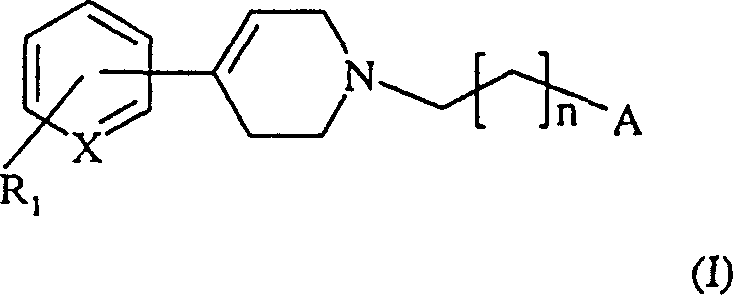
The pharmaceutical landscape is continuously evolving with the discovery of novel heterocyclic compounds that offer potent therapeutic benefits, particularly in the modulation of immune responses. Patent CN100335464C introduces a significant advancement in the field of aralkyl-tetrahydro-pyridines, detailing robust synthetic methodologies for compounds exhibiting strong TNF-alpha inhibitory activity. These molecules are critical for treating a wide array of inflammatory and autoimmune conditions, ranging from rheumatoid arthritis to multiple sclerosis. The patent outlines a versatile general formula where structural variations allow for fine-tuning of pharmacological properties, ensuring high efficacy and low toxicity profiles. By leveraging specific acylation and reduction strategies, this technology provides a reliable pathway for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards. The ability to synthesize these complex structures efficiently is paramount for drug developers seeking to advance candidates through clinical pipelines without supply chain bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating substituted tetrahydropyridines often suffer from significant drawbacks that hinder large-scale commercial production and cost-effectiveness. Many legacy processes rely on harsh reaction conditions that require extreme temperatures or pressures, leading to safety concerns and increased energy consumption in manufacturing facilities. Furthermore, conventional methods frequently utilize expensive transition metal catalysts that necessitate complex downstream purification steps to remove trace metal residues, which is a critical quality attribute for pharmaceutical ingredients. The formation of unwanted by-products and isomers is another common issue, resulting in lower overall yields and requiring extensive chromatographic separation that drives up production costs. Additionally, the use of unstable intermediates in older pathways can lead to inconsistent batch-to-batch quality, posing risks to supply chain reliability and regulatory compliance. These inefficiencies collectively create barriers to entry for generic manufacturers and increase the time-to-market for new therapeutic applications.
The Novel Approach
The methodology described in the patent data presents a streamlined and chemically elegant solution that overcomes the inherent inefficiencies of prior art techniques. By employing a sequential acylation, reduction, and dehydration strategy, the process achieves high selectivity and yield without the need for exotic reagents or specialized equipment. The use of borane-dimethylsulfide for reduction offers a safer and more controllable alternative to pyrophoric hydrides, significantly improving operational safety profiles in industrial settings. The dehydration step utilizes a simple acid mixture that facilitates rapid conversion to the desired tetrahydropyridine core, minimizing the formation of side products and simplifying isolation procedures. This approach allows for the introduction of diverse aralkyl groups, enabling the synthesis of a broad library of analogs for structure-activity relationship studies. Ultimately, this novel route enhances the economic viability of producing these high-value intermediates while maintaining the rigorous purity standards required by global health authorities.
Mechanistic Insights into Borane-Mediated Reduction and Dehydration
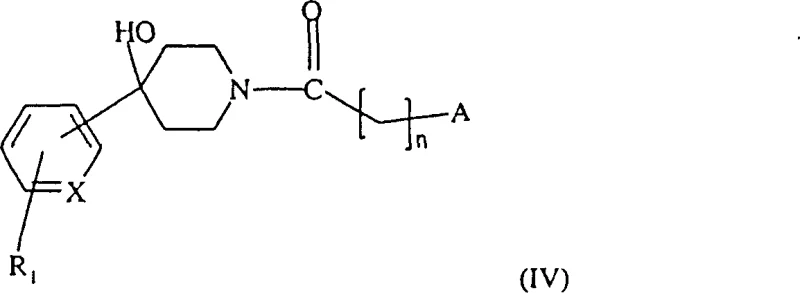
The core of this synthetic innovation lies in the precise control of the reduction and elimination steps that construct the tetrahydropyridine ring system with high fidelity. The reduction of the carbonyl intermediate is achieved using borane-dimethylsulfide, which acts as a mild yet effective hydride donor capable of selectively reducing ketones in the presence of other functional groups. This chemoselectivity is crucial for preserving the integrity of sensitive substituents on the aralkyl chain, ensuring that the final product retains its intended biological activity. The reaction proceeds through a coordinated transition state that minimizes steric hindrance, allowing for efficient conversion even with bulky substrates. Following reduction, the resulting piperidinol intermediate undergoes acid-catalyzed dehydration, where the hydroxyl group is protonated and eliminated as water to form the double bond within the ring. This elimination is driven by the stability of the conjugated system and is carefully controlled by temperature and acid concentration to prevent polymerization or rearrangement.
Impurity control is inherently built into this mechanism through the selection of reagents that minimize side reactions and the optimization of reaction parameters to favor the desired pathway. The use of halogenated solvents during the initial acylation step ensures solubility of reactants and facilitates heat dissipation, preventing localized hot spots that could degrade sensitive intermediates. During the dehydration phase, the specific ratio of acetic acid to sulfuric acid is critical for promoting elimination over substitution, thereby reducing the formation of ether by-products. The process also includes robust workup procedures, such as aqueous washes and pH adjustments, which effectively remove acidic residues and inorganic salts from the organic phase. These meticulous controls result in a crude product that requires minimal purification, often achievable through simple crystallization or short-column chromatography. Such mechanistic precision translates directly into higher process mass intensity and reduced waste generation, aligning with modern green chemistry principles.
How to Synthesize Aralkyl-Tetrahydro-Pyridine Efficiently
Implementing this synthesis requires a clear understanding of the sequential unit operations and the critical process parameters that govern each transformation. The process begins with the coupling of the amine and acid components, followed by the critical reduction and dehydration steps that define the molecular architecture. Operators must maintain strict control over temperature and addition rates to manage exotherms and ensure consistent reaction progress. Detailed standardized synthetic steps are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- React the aralkyl amine precursor with an activated acid derivative in a halogenated solvent with a tertiary amine base to form the keto-intermediate.
- Reduce the carbonyl group of the intermediate using borane-dimethylsulfide complex in an inert organic solvent under reflux conditions.
- Perform acid-catalyzed dehydration of the resulting piperidinol using an acetic acid and sulfuric acid mixture to yield the final tetrahydropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that directly address the pain points of procurement managers and supply chain directors in the pharmaceutical industry. The elimination of expensive noble metal catalysts significantly reduces the raw material costs associated with production, allowing for more competitive pricing structures in long-term supply agreements. The use of common organic solvents and reagents ensures that sourcing is not dependent on single suppliers or geopolitically restricted materials, thereby enhancing supply chain resilience and continuity. Furthermore, the simplified purification requirements reduce the consumption of chromatography media and solvents, leading to lower operational expenditures and a smaller environmental footprint. These factors combine to create a manufacturing process that is not only cost-effective but also scalable and sustainable.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts which are often subject to volatile market pricing and require costly recovery systems. By utilizing borane complexes and standard acid reagents, the overall cost of goods sold is significantly optimized without compromising product quality. The high yield and selectivity of the reaction minimize the loss of valuable starting materials, further contributing to economic efficiency. Additionally, the reduced need for extensive purification steps lowers the consumption of energy and consumables, resulting in substantial cost savings across the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals ensures that production schedules are not disrupted by raw material shortages or logistics delays. This stability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of downstream pharmaceutical manufacturers. The robustness of the chemistry also means that technology transfer between sites is straightforward, allowing for diversified manufacturing locations to mitigate regional risks. Consequently, partners can rely on a steady supply of high-quality intermediates to support their clinical and commercial programs without interruption.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up from laboratory to multi-ton production without significant re-engineering of the process equipment. The use of standard unit operations such as reflux and extraction facilitates integration into existing manufacturing facilities, reducing capital expenditure requirements. Moreover, the process generates less hazardous waste compared to traditional methods, simplifying compliance with environmental regulations and reducing disposal costs. This alignment with sustainability goals makes the technology attractive for companies seeking to improve their corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these aralkyl-tetrahydro-pyridine derivatives. The answers are derived directly from the patent specifications to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your supply chain.
Q: What are the key reaction conditions for the dehydration step?
A: The dehydration reaction is preferably carried out using a 3:1 volume mixture of acetic acid and sulfuric acid, heating at approximately 80-100°C for 1 to 3 hours to ensure complete conversion.
Q: Which reducing agents are suitable for the carbonyl reduction?
A: Borane complexes such as borane-dimethylsulfide are preferred, used in excess relative to the starting compound at reflux temperature in inert solvents like tetrahydrofuran.
Q: How is the final product isolated and purified?
A: The compound is isolated as a free base or salt via conventional techniques, often involving extraction with organic solvents like ethyl acetate and purification by silica gel column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aralkyl-Tetrahydro-Pyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to bring complex pathways like this from bench scale to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for your pharmaceutical intermediate requirements.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these critical compounds. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this advanced synthetic route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your specific project needs. Contact us today to initiate a conversation about securing a reliable and cost-effective supply of high-purity aralkyl-tetrahydro-pyridines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
