Optimizing Intumescent Flame Retardant Production With Continuous Triazine Synthesis Technology
Optimizing Intumescent Flame Retardant Production With Continuous Triazine Synthesis Technology
Introduction to Advanced Triazine-Based Char Forming Technology
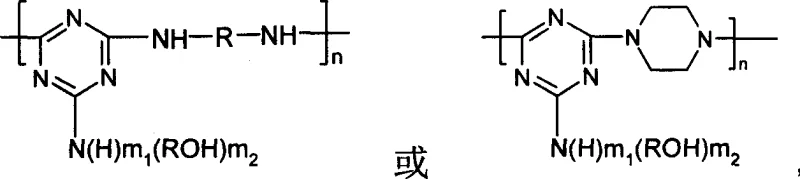
The development of high-performance intumescent flame retardants (IFR) represents a critical frontier in polymer safety engineering, specifically addressing the limitations of traditional halogenated systems. Patent CN1288140C introduces a groundbreaking macromolecular triazine series char-forming-foaming agent that fundamentally alters the synthesis landscape for polymer additive suppliers. This technology leverages the unique reactivity of cyanuric chloride to create oligomeric structures with superior thermal stability and char-forming capacity. Unlike conventional methods that suffer from discontinuous processing and excessive solvent consumption, this innovation utilizes a continuous nucleophilic substitution pathway. The resulting material exhibits a softening point between 225-250°C and maintains structural integrity up to 270°C, making it an ideal candidate for high-temperature polymer processing applications. By integrating hydroxyl and amine functionalities into a triazine backbone, the process ensures excellent compatibility with polyphosphate ammonium systems, thereby enhancing the overall fire resistance of the final polymer composite.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for triazine-based char formers have historically been plagued by significant operational inefficiencies and environmental drawbacks that hinder large-scale adoption. Existing technologies often rely on discontinuous batch processes where intermediates must be isolated, washed, and dried before proceeding to the next reaction step, leading to prolonged production cycles exceeding 29 hours. Furthermore, these legacy methods frequently utilize environmentally unfriendly organic solvents such as xylene, which pose substantial challenges for distillation recovery and waste management. The thermal stability of products generated through these older pathways is often compromised, with char residues at 800°C dropping as low as 5%, severely limiting their effectiveness in intumescent flame retardant formulations. Additionally, the use of acid-binding agents in non-aqueous systems complicates post-reaction purification, resulting in higher impurity levels that can negatively impact the mechanical properties of the host polymer matrix.
The Novel Approach
The innovative methodology described in the patent data revolutionizes this landscape by implementing a continuous, one-pot synthesis strategy that drastically reduces reaction time to approximately 16-18 hours. By strategically controlling reaction temperatures to activate specific chlorine atoms on the cyanuric chloride ring, the process eliminates the need for intermediate isolation, thereby streamlining the workflow and minimizing material loss. A key advancement is the substitution of hazardous organic solvents with water or water-acetone mixtures, which facilitates easy solvent recovery through simple distillation during the temperature ramping phases. This shift not only aligns with stringent environmental compliance standards but also significantly lowers the operational costs associated with solvent procurement and disposal. The resulting macromolecular oligomers demonstrate a char formation rate exceeding 35% at 800°C under nitrogen, providing a robust carbonaceous barrier that effectively insulates the underlying polymer substrate from heat and oxygen during combustion events.
Mechanistic Insights into Stepwise Nucleophilic Substitution
The core chemical innovation lies in the precise exploitation of the differential reactivity of the three chlorine atoms attached to the s-triazine ring, which allows for controlled stepwise functionalization without cross-reactivity. At low temperatures ranging from -5 to 10°C, the first chlorine atom undergoes nucleophilic substitution with alkanolamines, forming a stable monosubstituted intermediate while preserving the remaining chlorines for subsequent reactions. As the temperature is elevated to the 30-45°C range, the second chlorine atom becomes activated, permitting the introduction of diamine linkers that begin to establish the oligomeric backbone structure. Finally, raising the temperature to reflux conditions activates the third chlorine position, driving the polymerization to completion and forming the high molecular weight macromolecular network essential for effective char formation. This temperature-programmed approach ensures high regioselectivity and minimizes the formation of unwanted by-products, resulting in a product with a narrow molecular weight distribution and consistent performance characteristics.
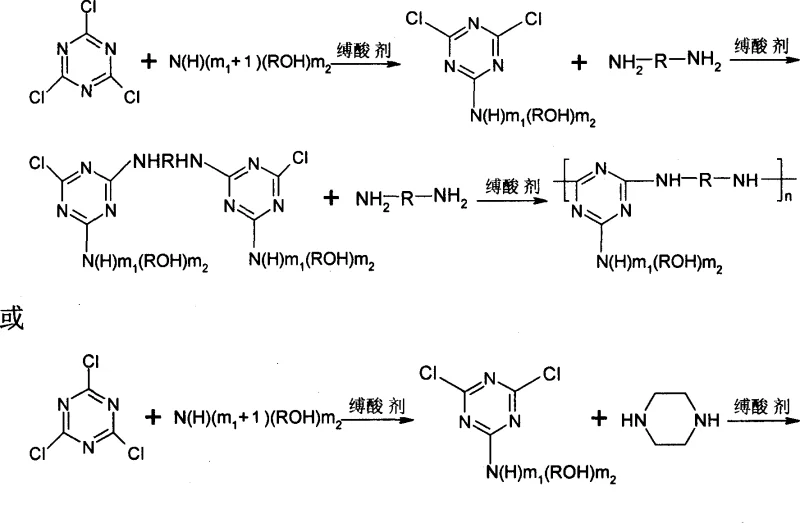
Impurity control is intrinsically built into this mechanism through the rigorous maintenance of pH levels between 5 and 8 during each substitution stage. By using inorganic bases such as sodium hydroxide or sodium bicarbonate as acid-binding agents, the process neutralizes the hydrochloric acid by-product generated during nucleophilic attack without introducing organic contaminants. The use of water as a primary solvent further aids in the removal of inorganic salts through simple filtration and washing steps, ensuring the final white powder product meets high purity specifications required for sensitive electronic or automotive polymer applications. The presence of hydroxyl groups from the alkanolamine reactants provides additional sites for cross-linking during the charring phase, enhancing the cohesive strength of the intumescent layer. This mechanistic precision guarantees that the final triazine oligomer possesses the necessary thermal resilience to withstand processing temperatures while remaining reactive enough to expand and char upon exposure to fire.
How to Synthesize Macromolecular Triazine Char-Forming Agent Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and reagent addition rates to maximize yield and molecular weight consistency. The process begins with the dispersion of cyanuric chloride in a chilled aqueous or acetone-water medium, followed by the controlled addition of alkanolamines to initiate the first substitution phase. Operators must monitor pH levels continuously to prevent localized acidity that could lead to hydrolysis of the triazine ring, ensuring the reaction proceeds exclusively via nucleophilic substitution. Detailed standardized synthesis steps see the guide below.
- Disperse cyanuric chloride in water or acetone-water mixture at -5 to 10°C, then dropwise add alkanolamine and inorganic base to maintain pH 5-8 for monosubstitution.
- Raise temperature to 30-45°C and add diamine with base control, then increase to 50-60°C to complete the disubstitution reaction forming the intermediate.
- Add remaining diamine and base, raise temperature to solvent reflux for 6-8 hours to polymerize, then filter, wash, and dry to obtain the final oligomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis technology offers compelling advantages that directly address cost volatility and supply chain resilience in the polymer additive sector. The elimination of intermediate isolation steps significantly reduces labor costs and equipment occupancy time, allowing for higher throughput within existing manufacturing infrastructure. By shifting from expensive, hard-to-recycle solvents like xylene to water-based systems, manufacturers can achieve substantial cost savings in raw material procurement and waste treatment compliance. The continuous nature of the reaction also minimizes the risk of batch-to-batch variability, ensuring a consistent supply of high-quality char-forming agents that meet rigorous customer specifications without the need for extensive rework or blending. This reliability is crucial for maintaining just-in-time inventory levels and preventing production stoppages in downstream compounding operations.
- Cost Reduction in Manufacturing: The transition to a continuous process inherently lowers energy consumption per unit of output by eliminating repeated heating and cooling cycles associated with batch isolation. Furthermore, the ability to recover and reuse acetone during the reflux stage creates a closed-loop solvent system that drastically reduces raw material expenditure over the lifecycle of the plant. The use of commodity chemicals such as cyanuric chloride, ethanolamine, and caustic soda ensures that input costs remain stable and predictable, shielding the supply chain from the price fluctuations often seen with specialized organic reagents. These efficiencies collectively contribute to a more competitive cost structure for high-purity polymer additive manufacturing without compromising on product performance or safety standards.
- Enhanced Supply Chain Reliability: The simplified workflow reduces the number of critical control points where delays or errors could occur, thereby shortening the overall lead time from raw material intake to finished goods. Water-based chemistry mitigates the regulatory and logistical burdens associated with transporting and storing large volumes of flammable organic solvents, simplifying site permitting and insurance requirements. The robustness of the reaction conditions allows for scalability from pilot plants to multi-ton commercial reactors with minimal process re-engineering, ensuring that supply can be rapidly ramped up to meet surging market demand for flame retardant solutions. This flexibility provides a strategic buffer against supply disruptions and enables faster response times to custom formulation requests from key accounts.
- Scalability and Environmental Compliance: Adopting this green chemistry approach aligns production capabilities with increasingly stringent global environmental regulations regarding volatile organic compound (VOC) emissions and wastewater discharge. The high char yield and thermal stability of the product reduce the loading levels required in final formulations, indirectly lowering the carbon footprint of the end-user's polymer products. Waste streams are primarily composed of benign inorganic salts and water, which are easier and cheaper to treat compared to the complex organic waste generated by traditional xylene-based processes. This sustainability profile enhances the marketability of the supply chain to eco-conscious brands and helps secure long-term contracts with multinational corporations committed to reducing their environmental impact.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and performance of this triazine-based synthesis technology in industrial settings. These answers are derived directly from the experimental data and process parameters outlined in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this char-forming agent into existing intumescent flame retardant supply chains.
Q: How does this continuous synthesis method improve thermal stability compared to traditional batch processes?
A: The continuous method allows precise temperature staging for chlorine activation on the triazine ring, reducing side reactions and ensuring higher molecular weight oligomers with decomposition temperatures exceeding 270°C.
Q: What are the solvent recovery advantages of using water-acetone mixtures in this process?
A: Using water or water-acetone mixtures enables efficient solvent recovery via distillation during the heating phase, significantly reducing organic waste and eliminating the need for environmentally harmful solvents like xylene.
Q: Can this char-forming agent be scaled for industrial intumescent flame retardant production?
A: Yes, the process eliminates intermediate isolation steps and uses common inorganic bases, making it highly scalable for commercial production of high-performance intumescent flame retardant systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Macromolecular Triazine Char-Forming Agent Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of thermal stability and process efficiency in the production of advanced polymer additives. Our CDMO expertise allows us to translate complex laboratory pathways like the continuous triazine synthesis into robust, commercial-scale operations with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure every batch of char-forming agent meets the exacting standards required for high-performance flame retardant applications. Our team is dedicated to optimizing reaction conditions to maximize char yield and minimize impurity profiles, delivering a product that consistently outperforms traditional alternatives in both safety and processing metrics.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can enhance your product portfolio and reduce overall manufacturing costs. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your operation. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and sourcing initiatives, ensuring a seamless transition to this superior technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
