Advanced Refining Technology for High-Purity Suvorexant Commercial Production
The pharmaceutical industry continuously demands more efficient and environmentally sustainable processes for the production of critical active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in patent CN110818701A, which discloses a novel refining method for Suvorexant, a potent Orexin receptor antagonist used in the treatment of insomnia. This technology addresses the critical challenges of impurity control and solvent safety that have plagued previous manufacturing routes. By shifting from hazardous organic solvents to a greener ethanol-water system, this method not only enhances the safety profile of the manufacturing process but also drastically improves the final product's purity and yield. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediates supplier, understanding the technical nuances of this patent is essential for securing a stable supply chain. The following analysis dissects the chemical engineering principles behind this innovation, demonstrating how it facilitates cost reduction in pharmaceutical manufacturing while ensuring the highest standards of quality control.
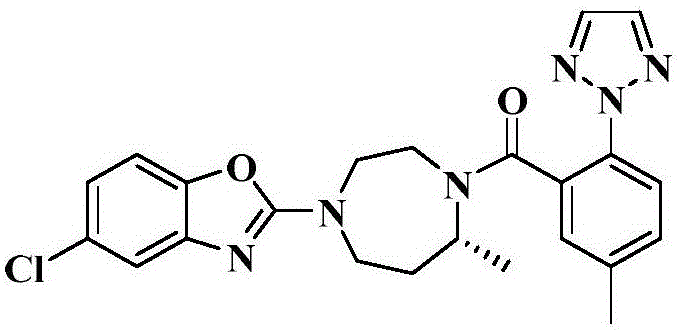
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the purification of Suvorexant relied heavily on solvent systems that presented significant regulatory and operational hurdles. Conventional methods, such as those disclosed in earlier patents like WO2013169610, utilized mixtures of acetonitrile and water. Acetonitrile is classified as a Class 2 solvent under ICH guidelines, meaning it has inherent toxicity limitations and requires stringent residual solvent testing, which complicates the regulatory filing process for the final drug product. Furthermore, other existing techniques employed mixtures of isopropyl acetate and n-heptane. While these organic solvents can induce crystallization, they often result in poor impurity removal profiles, failing to reduce isomer content to the rigorous levels required for medicinal grade API. The reliance on these solvents also introduces higher environmental disposal costs and safety risks associated with flammability and toxicity, creating bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The method described in CN110818701A represents a paradigm shift by employing a binary solvent system composed of C1-C4 alkanols and purified water, with ethanol being the preferred embodiment. This approach leverages the differential solubility of Suvorexant and its related impurities in hot versus cold aqueous alcohol solutions. By heating the mixture to a specific range of 50-90°C, preferably 70-80°C, the crude product is fully dissolved, allowing for the removal of insoluble mechanical impurities through hot filtration. Subsequent controlled cooling to 20±5°C induces selective crystallization of the target molecule while leaving key isomeric impurities in the mother liquor. This gradient crystallization technique is not only operationally simpler but also aligns with green chemistry principles by utilizing Class 3 solvents, which are considered less toxic and safer for large-scale industrial production. This transition significantly simplifies the solvent recovery process and reduces the overall environmental footprint of the manufacturing facility.
Mechanistic Insights into Ethanol-Water Recrystallization
The efficacy of this refining method lies in the precise thermodynamic control of the crystallization process. Suvorexant possesses specific polar and non-polar regions within its molecular structure, as seen in its chemical diagram, which interact differently with the ethanol-water matrix compared to its impurities. The addition of water to the alcohol solvent reduces the overall solubility of the organic compound, acting as an anti-solvent that drives supersaturation upon cooling. However, the key to high purity is the temperature gradient. By maintaining the solution at 70-80°C initially, the system ensures that all target material is in the solution phase, preventing premature nucleation which can trap impurities within the crystal lattice. The slow cooling phase allows for the formation of a stable crystal polymorph that inherently excludes structurally similar isomers. This mechanism is critical for R&D teams focusing on purity and impurity profiles, as it directly impacts the bioavailability and safety of the final dosage form.
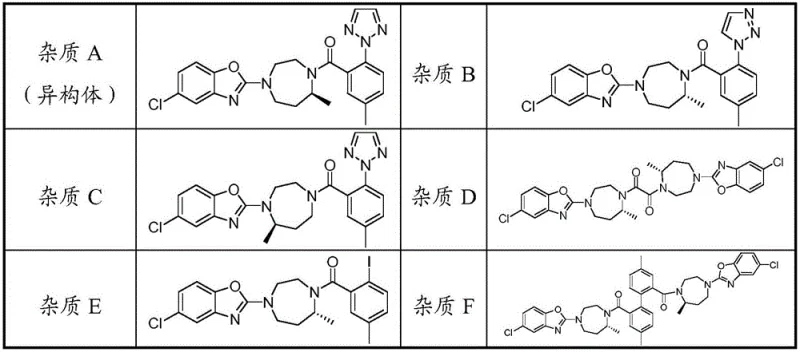
Impurity control is the cornerstone of this technology. The crude Suvorexant typically contains a complex array of by-products, including stereoisomers and synthetic intermediates. As illustrated in the impurity profiles, specific isomers (such as Impurity A) and related substances (Impurities B through F) share high structural similarity with the target molecule, making them difficult to separate via standard chromatography on a large scale. The ethanol-water recrystallization method exploits subtle differences in lattice energy and solvation shells. The polar hydroxyl groups of the alcohol and water form hydrogen bonds that stabilize the impurities in the solution phase more effectively than they stabilize the target crystal. Consequently, when the Suvorexant crystallizes out, these impurities remain dissolved in the mother liquor. This selective exclusion mechanism allows the process to reduce single impurity levels to below 0.1% and total impurities to under 0.2%, achieving a final purity of over 99.8% in a single pass. This level of control eliminates the need for multiple recrystallization cycles, which traditionally degrade yield.
How to Synthesize Suvorexant Efficiently
Implementing this refining protocol requires strict adherence to the temperature and solvent ratio parameters defined in the patent data. The process begins with the preparation of the solvent system, followed by the dissolution of the crude material under controlled heating. It is crucial to maintain the solution clarity before initiating the cooling phase to ensure that no mechanical particulates contaminate the final crystal form. The washing step using a cold mixture of ethanol and water is equally vital, as it removes surface-adhered mother liquor containing the concentrated impurities without redissolving the purified crystals. For detailed operational parameters, temperature ramping rates, and specific solvent ratios required for GMP compliance, please refer to the standardized synthesis steps provided in the technical guide below.
- Dissolve crude Suvorexant in a C1-C4 alcohol and water mixture at 50-90°C.
- Filter hot to remove mechanical impurities, then cool to 20°C for crystallization.
- Wash the filter cake with alcohol-water solution and dry at 60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this refining method offers substantial strategic benefits beyond mere technical specifications. The elimination of Class 2 solvents like acetonitrile directly translates to a reduction in regulatory compliance costs and hazardous waste disposal fees. Furthermore, the high yield achieved in a single refining step means that less crude material is required to produce the same amount of API, effectively lowering the raw material cost per kilogram. This efficiency enhances supply chain reliability by reducing the dependency on complex multi-step purification sequences that are prone to bottlenecks and yield losses. The use of ethanol and water, which are commodity chemicals with stable global pricing and availability, further insulates the manufacturing process from volatile solvent market fluctuations, ensuring consistent production costs and reliable [precise industry noun] supplier performance.
- Cost Reduction in Manufacturing: The shift to an ethanol-water solvent system eliminates the need for expensive and highly regulated solvent recovery systems associated with acetonitrile. Since ethanol is a Class 3 solvent with higher permissible daily exposure limits, the cost of testing for residual solvents is significantly reduced. Additionally, the high yield of over 90% in a single step minimizes material loss, which is a major cost driver in API production. By avoiding multiple recrystallization cycles, the process saves on energy consumption, labor hours, and equipment occupancy time, leading to substantial cost savings in pharmaceutical manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Ethanol and water are universally available commodities, unlike specialized solvent mixtures that may face supply constraints. This availability ensures that production schedules are not disrupted by raw material shortages. The robustness of the process, demonstrated by its success across various batch sizes from grams to kilograms, indicates high scalability. This scalability reduces lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more quickly to market demand spikes. The simplified process flow also reduces the risk of operational errors, ensuring a consistent and uninterrupted supply of critical medication ingredients to downstream partners.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (50-90°C) and atmospheric pressure, which reduces the energy load on manufacturing facilities. The use of non-toxic solvents simplifies the environmental permitting process and reduces the burden on wastewater treatment systems. This environmental compliance is increasingly important for multinational corporations aiming to meet sustainability goals. The method's ability to handle large batch sizes, as evidenced by the successful scaling in the patent examples, confirms its suitability for commercial scale-up of complex pharmaceutical intermediates, ensuring that the technology can meet global market volume requirements efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to industry standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own production lines or for procurement teams assessing the quality of potential suppliers.
Q: What solvent system is used in the novel Suvorexant refining method?
A: The method utilizes a mixture of C1-C4 alkanol (preferably ethanol) and purified water, replacing toxic Class 2 solvents like acetonitrile.
Q: What purity levels can be achieved with this recrystallization technique?
A: The process consistently achieves a chromatographic purity of over 99.8% with isomer content reduced to less than 0.1%.
Q: How does this method improve yield compared to conventional processes?
A: By optimizing the solvent ratio and temperature gradient, the method achieves yields exceeding 90% in a single refining step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Suvorexant Supplier
The technical potential of the ethanol-water recrystallization method for Suvorexant is immense, offering a pathway to high-purity production that meets the rigorous demands of the global pharmaceutical market. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the <0.1% isomer content and >99.8% purity benchmarks set by this patent. We understand the critical nature of API intermediates and are committed to delivering products that adhere to the highest international regulatory standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for Suvorexant or related compounds. By leveraging our expertise in process optimization and scale-up, we can help you secure a stable, high-quality supply of this critical insomnia medication ingredient.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
