Advanced Rosuvastatin Lactone Manufacturing Process for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value statin intermediates, and the technology disclosed in patent CN103613582A represents a significant advancement in the manufacturing of Rosuvastatin lactone. This specific chemical entity, with the molecular formula C22H26FN3O5S, serves as a critical precursor for third-generation HMG-CoA reductase inhibitors, which are essential for managing hypercholesterolemia and reducing cardiovascular risk factors globally. The patented method distinguishes itself by integrating a series of highly efficient transformations, including condensation, oxidation, reduction, and substitution, to achieve a final product with exceptional optical purity exceeding 99.5% ee. By leveraging starting materials such as fluorobenzaldehyde and methyl isobutyrylacetate, the process ensures a streamlined workflow that minimizes waste generation while maximizing throughput. For R&D directors and procurement specialists, understanding the nuances of this route is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards. 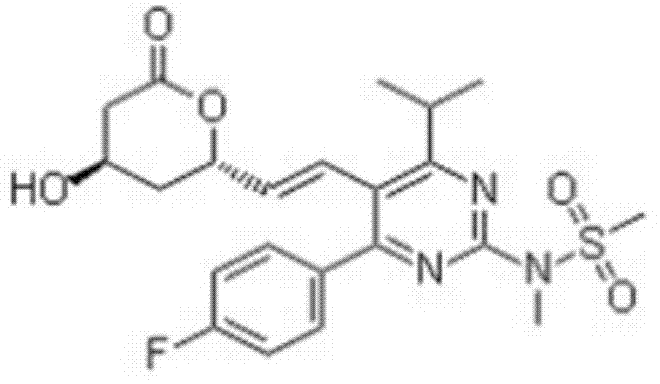 The structural integrity depicted above highlights the complexity of the molecule, necessitating a synthesis strategy that balances reactivity with selectivity to avoid costly impurities.
The structural integrity depicted above highlights the complexity of the molecule, necessitating a synthesis strategy that balances reactivity with selectivity to avoid costly impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for statin lactones often suffer from prohibitive costs associated with chiral resolution steps and the use of expensive transition metal catalysts that require rigorous removal to meet safety specifications. Many legacy processes rely on multi-step sequences where yields drop precipitously at each stage, leading to an overall economic inefficiency that makes commercial scale-up of complex pharmaceutical intermediates challenging for mid-sized manufacturers. Furthermore, conventional methods frequently generate substantial amounts of hazardous waste, particularly when employing stoichiometric oxidants or protecting group strategies that add unnecessary mass and complexity to the process flow. The environmental burden of disposing of heavy metal residues and organic solvents from these older pathways creates significant compliance hurdles for supply chain heads aiming to maintain green manufacturing credentials. Additionally, the instability of certain intermediates in traditional routes often necessitates cryogenic conditions or inert atmospheres that drive up energy consumption and operational expenditures. These factors collectively contribute to extended lead times and higher unit costs, making it difficult for procurement managers to negotiate favorable terms without compromising on quality or delivery schedules.
The Novel Approach
In contrast, the novel approach detailed in the patent data introduces a convergent synthesis strategy that significantly reduces the number of isolation steps and improves the overall atom economy of the transformation. By utilizing a condensation reaction between fluorobenzaldehyde and urea derivatives under catalytic conditions, the process establishes the pyrimidine core with remarkable efficiency, achieving yields as high as 98.7% in the initial step. This methodology eliminates the need for cumbersome chiral separation techniques by incorporating stereochemical control directly into the bond-forming events, thereby ensuring high-purity rosuvastatin lactone is obtained without additional purification burdens. The use of mild reagents and optimized reaction temperatures, such as the controlled addition of nitric acid at 10 to 25°C, enhances safety profiles and allows for easier heat management in large reactors. Moreover, the pathway avoids the accumulation of toxic byproducts, aligning with modern sustainability goals and reducing the cost reduction in statin manufacturing through simplified waste treatment protocols. This innovative framework provides a scalable solution that addresses the historical bottlenecks of statin production, offering a competitive edge for partners seeking long-term supply stability.
Mechanistic Insights into CuCl-Catalyzed Pyrimidine Formation
The cornerstone of this synthetic success lies in the initial cyclization mechanism, where cuprous chloride acts as a Lewis acid catalyst to facilitate the Biginelli-like condensation of the aldehyde, beta-keto ester, and urea. This catalytic cycle promotes the formation of the dihydropyrimidinone ring system with high regioselectivity, preventing the formation of isomeric byproducts that could complicate downstream processing. The mechanistic pathway involves the activation of the carbonyl group of the fluorobenzaldehyde, making it more susceptible to nucleophilic attack by the urea nitrogen, followed by enolization of the ester component to close the ring. Understanding this electronic interplay is crucial for R&D teams aiming to replicate the process, as slight deviations in pH or temperature can alter the reaction kinetics and impact the final crystal morphology. The subsequent oxidation step using sodium nitrite and nitric acid converts the dihydro-intermediate into the aromatic pyrimidine system, a transformation that is exothermic and requires precise thermal control to prevent over-oxidation or ring degradation. By maintaining the reaction temperature within the specified 101 to 125°C range during batch addition, the process ensures complete conversion while preserving the integrity of the sensitive fluorophenyl moiety. This level of mechanistic control is what differentiates a laboratory curiosity from a commercially viable manufacturing process.
Impurity control is further enhanced in the later stages of the synthesis, particularly during the reduction and coupling phases where chemoselectivity is paramount. The use of diisobutyl aluminium hydride (DIBAL-H) for the reduction of the ester to the alcohol is executed at low temperatures to prevent over-reduction to the amine or cleavage of the sulfonamide group. Following this, the conversion of the alcohol to the bromide using phosphorus tribromide proceeds with inversion of configuration, setting the stage for the subsequent phosphine-mediated olefination. The final coupling reaction with the chiral side chain precursor, mediated by NaHMDS, is the critical step that establishes the pharmacologically active geometry of the heptenoic acid side chain. Any racemization at this stage would render the batch useless, but the patented conditions utilize non-protic solvents like tetrahydrofuran and strict temperature controls to maintain optical purity above 99.5% ee. This rigorous attention to stereochemical detail ensures that the final API intermediate meets the strict specifications required by global regulatory bodies, minimizing the risk of batch rejection and ensuring consistent therapeutic efficacy.
How to Synthesize Rosuvastatin Lactone Efficiently
Executing this synthesis requires a disciplined approach to reaction monitoring and workup procedures to ensure that the theoretical yields reported in the patent are realized in a production environment. The process begins with the preparation of the pyrimidine core, followed by sequential functionalization to install the sulfonamide and side chain moieties with high fidelity. Operators must adhere strictly to the molar ratios and addition rates specified, particularly during the exothermic oxidation and reduction steps, to maintain safety and product quality. Detailed standard operating procedures should be established for the handling of moisture-sensitive reagents like DIBAL-H and NaHMDS to prevent premature decomposition and ensure reproducible results. The following guide outlines the critical operational parameters derived from the patent examples, serving as a foundational reference for process engineers scaling this technology.
- Condense fluorobenzaldehyde with methyl isobutyrylacetate and urea using cuprous chloride to form the dihydropyrimidinone core.
- Perform oxidation using nitric acid and sodium nitrite, followed by sulfonylation to introduce the sulfonamide group.
- Execute reduction with DIBAL-H, bromination, phosphine substitution, and final coupling with the side chain to yield the lactone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers tasked with optimizing the cost structure of their supply chains while maintaining high quality standards. The elimination of expensive chiral resolving agents and the reduction in the number of purification steps translate directly into substantial cost savings, allowing for more competitive pricing models without sacrificing margin. Furthermore, the use of commodity chemicals such as fluorobenzaldehyde and urea ensures that raw material availability is not a bottleneck, enhancing supply chain reliability even during periods of market volatility. The robustness of the process means that production schedules can be maintained with greater predictability, reducing lead time for high-purity pharmaceutical intermediates and enabling just-in-time inventory strategies. For supply chain heads, the simplified waste profile reduces the logistical burden of hazardous material disposal, lowering overhead costs associated with environmental compliance and facility maintenance. These combined factors create a resilient supply framework that supports long-term contractual agreements and fosters trust between manufacturers and their downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates several unit operations found in legacy routes, such as extensive chromatographic purifications and recrystallizations, which are both time-consuming and solvent-intensive. By achieving high yields in each step, specifically the 96.5% yield in the final hydrolysis and lactonization, the process minimizes the loss of valuable intermediates, effectively lowering the cost of goods sold. The avoidance of precious metal catalysts like palladium or platinum, which are subject to significant price fluctuations, further stabilizes the production budget and protects against raw material cost spikes. Additionally, the high atom economy means less waste is generated per kilogram of product, reducing the expenses related to waste treatment and disposal fees. These efficiencies compound over large production volumes, resulting in significant financial advantages for companies adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production is not vulnerable to the supply disruptions that often plague specialty reagent markets. Fluorobenzaldehyde and methyl isobutyrylacetate are produced by multiple global suppliers, providing procurement teams with the flexibility to source from diverse vendors to mitigate risk. The stability of the intermediates allows for potential storage between steps if necessary, providing a buffer against unexpected demand surges or equipment downtime. This flexibility is crucial for maintaining continuous supply to API manufacturers who operate on tight schedules and cannot afford delays. The proven scalability of the route, demonstrated by its suitability for industrial production, gives supply chain heads confidence in the ability to ramp up volumes quickly as market demand for statins grows.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents and reagents that are easier to recover and recycle, thus minimizing the environmental footprint of the manufacturing facility. The absence of heavy metal contamination in the final product simplifies the regulatory filing process and reduces the need for specialized analytical testing for residual metals. This compliance advantage accelerates the time to market for new generic formulations or process changes, providing a strategic edge in a competitive landscape. The simplified three-waste treatment mentioned in the patent reduces the load on effluent treatment plants, allowing facilities to operate within stricter environmental permits without costly upgrades. Overall, the route supports sustainable manufacturing goals while delivering the economic performance required by stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rosuvastatin lactone synthesis route, providing clarity for decision-makers evaluating this technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details helps stakeholders assess the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What is the overall yield of the Rosuvastatin lactone synthesis route?
A: According to patent CN103613582A, the total yield for the complete synthesis route is approximately 63.5%, with individual step yields ranging from 89.3% to 98.7%, demonstrating high efficiency suitable for industrial scale-up.
Q: Does this process involve heavy metal catalysts that complicate purification?
A: The described route utilizes cuprous chloride in the initial condensation but is designed to avoid complex heavy metal residues in the final steps, simplifying the purification process and ensuring high optical purity with ee values greater than 99.5%.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the patent explicitly states the route is suitable for large-scale industrialization due to stable processing conditions, simple three-waste treatment, and the use of readily available raw materials like fluorobenzaldehyde and urea.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a robust and scalable synthesis route for key pharmaceutical intermediates like Rosuvastatin lactone to support the global demand for cardiovascular medications. Our team of expert chemists has thoroughly analyzed the patented methodology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life on an industrial scale. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch achieves the high optical purity and chemical integrity demanded by top-tier pharmaceutical clients. Our facility is equipped to handle the specific reagents and conditions outlined in the patent, ensuring that the transition from lab scale to commercial manufacturing is seamless and efficient. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and technical excellence.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific supply chain requirements and reduce your overall manufacturing costs. We encourage you to request a Customized Cost-Saving Analysis tailored to your volume needs, which will highlight the specific economic benefits of switching to this more efficient process. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your exact specifications. Let us help you secure a stable and cost-effective supply of this critical intermediate, ensuring your production lines remain uninterrupted and competitive in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
