Advanced L-Prolineamide Catalysts: Revolutionizing Chiral Pharmaceutical Intermediate Manufacturing
Introduction to Next-Generation Organocatalysis
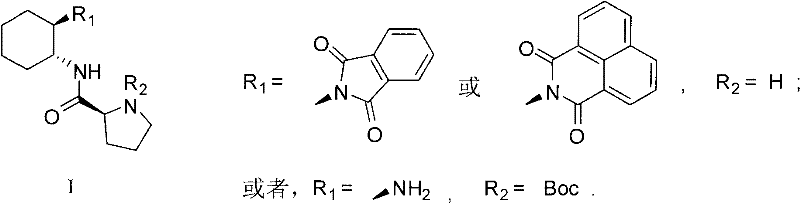
The landscape of asymmetric synthesis is undergoing a paradigm shift, driven by the urgent need for greener, more efficient manufacturing processes in the pharmaceutical and fine chemical sectors. Patent CN101891668B introduces a groundbreaking class of L-prolineamide derivatives, specifically designed to overcome the limitations of traditional catalytic systems. These novel compounds, characterized by the general Formula I, represent a significant leap forward in organocatalysis, offering a robust alternative to expensive and toxic transition metal catalysts. By leveraging the inherent chirality of L-proline combined with a rigid cyclohexane diamine backbone, these derivatives achieve exceptional stereocontrol in direct Aldol reactions. This technological advancement is not merely an academic curiosity; it provides a tangible pathway for a reliable pharmaceutical intermediate supplier to deliver high-purity chiral building blocks with reduced environmental footprints. The ability to operate under mild conditions without stringent anhydrous requirements marks a distinct improvement in process safety and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-carbon bonds via asymmetric Aldol reactions has relied heavily on two main approaches: stoichiometric chiral auxiliaries or transition metal catalysis. While effective, these methods suffer from inherent drawbacks that hinder large-scale adoption. Metal-catalyzed processes often require rigorous exclusion of moisture and oxygen, necessitating specialized equipment and increasing capital expenditure. Furthermore, the presence of residual heavy metals in the final active pharmaceutical ingredient (API) is a critical regulatory concern, demanding extensive and costly purification steps to meet strict ppm limits. Traditional enolate-based methods also struggle with atom economy, generating significant waste through the use of protecting groups and stoichiometric bases. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, creating bottlenecks for supply chain managers seeking agility in drug development.
The Novel Approach
The technology disclosed in CN101891668B offers a transformative solution by utilizing metal-free organic small molecule catalysts. These L-prolineamide derivatives function efficiently in common organic solvents and even tolerate trace amounts of water, drastically simplifying reaction setup and execution. The core innovation lies in the molecular design, which integrates a proline moiety with a chiral diamine scaffold to create a highly organized transition state. This architecture enables the direct reaction between unmodified ketones and aldehydes, bypassing the need for pre-formed enolates. For procurement teams, this translates to cost reduction in API manufacturing through simplified workflows and the elimination of expensive metal scavengers. The catalysts exhibit high turnover frequencies and can be recovered and reused multiple times, addressing the historical criticism of organocatalysis regarding high catalyst loading. This approach aligns perfectly with modern green chemistry principles while delivering the high stereoselectivity required for complex drug synthesis.
Mechanistic Insights into L-Prolineamide Catalyzed Aldol Reactions
The exceptional performance of these derivatives stems from a sophisticated interplay of steric hindrance and hydrogen bonding networks. The rigid cyclohexane ring in the catalyst backbone restricts conformational freedom, locking the molecule into a specific geometry that favors one enantiomeric pathway over the other. During the catalytic cycle, the secondary amine of the proline ring forms an enamine intermediate with the ketone substrate, activating it for nucleophilic attack. Simultaneously, the amide NH group acts as a hydrogen bond donor, coordinating with the carbonyl oxygen of the aldehyde electrophile. This dual activation mechanism lowers the energy barrier for the reaction while precisely orienting the reactants. The result is a highly ordered transition state that minimizes the formation of unwanted syn-diastereomers and racemic byproducts. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters for new substrates.

Furthermore, the impurity profile of reactions using these catalysts is remarkably clean. The high diastereoselectivity (dr values up to 99:1) and enantioselectivity (ee values up to 98%) mean that downstream purification is significantly less burdensome compared to traditional methods. The patent data indicates that electron-withdrawing groups on the aromatic aldehyde enhance reactivity, likely by increasing the electrophilicity of the carbonyl carbon, while bulky substituents improve stereocontrol through increased steric repulsion in the transition state. This predictability allows chemists to tailor reaction conditions, such as solvent choice and temperature, to maximize yield and purity. For instance, the use of acetonitrile at -20°C is identified as optimal for balancing reaction rate and selectivity. Such mechanistic clarity ensures that the transfer of this technology from the lab bench to commercial scale-up of complex pharmaceutical intermediates is seamless and reproducible.
How to Synthesize L-Prolineamide Derivative b Efficiently
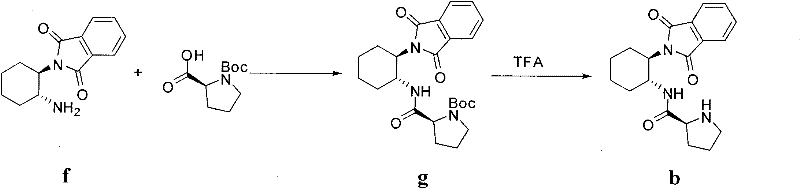
The preparation of the most effective catalyst, derivative 'b', follows a concise two-step synthetic route that utilizes readily available starting materials. The process begins with the coupling of N-Boc-L-Proline with a mono-protected (R,R)-1,2-cyclohexanediamine. This step requires careful control of temperature and activation agents to ensure high conversion without racemization. Following the isolation of the protected intermediate, a deprotection step unveils the active catalytic species. The robustness of this synthesis is evidenced by the high yields reported in the patent examples, making it an attractive candidate for in-house production or contract manufacturing. The detailed standardized synthesis steps for producing this high-performance catalyst are outlined in the guide below.
- Couple N-Boc-L-Pro with mono-protected (R,R)-1,2-cyclohexanediamine using ethyl chloroformate activation in anhydrous dichloromethane at 0°C.
- Purify the resulting intermediate g via flash column chromatography to obtain a white solid with high purity.
- Deprotect the Boc group using trifluoroacetic acid (TFA) in dichloromethane, followed by neutralization and recrystallization to yield the final catalyst b.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this catalytic technology offers compelling strategic benefits. The shift from metal-based to organocatalytic processes fundamentally alters the cost structure of chiral synthesis. By eliminating the need for precious metals like palladium or rhodium, manufacturers can insulate themselves from volatile commodity prices and supply disruptions associated with mining regions. Moreover, the simplified purification protocols reduce the consumption of silica gel and solvents during chromatography, leading to substantial waste reduction and lower disposal costs. The ability to recycle the catalyst further amplifies these savings, as a single batch of catalyst can service multiple production runs. This efficiency directly contributes to a more resilient and cost-effective supply chain.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound. Traditional metal-catalyzed routes often incur hidden costs related to metal removal validation and specialized waste treatment. In contrast, this organocatalytic method utilizes inexpensive, non-toxic organic acids as cocatalysts and operates in standard solvents. The high atom economy of the direct Aldol reaction means less raw material is wasted as byproducts. Additionally, the catalyst recovery process described in the patent allows for multiple reuse cycles without significant loss of activity, effectively amortizing the cost of the catalyst over a larger volume of product. This results in a significantly lower cost per kilogram of the final chiral intermediate.
- Enhanced Supply Chain Reliability: Supply chain heads prioritize vendors who can guarantee consistent delivery. The synthetic route for these L-prolineamide derivatives relies on bulk chemicals like L-proline and cyclohexanediamine, which are globally available and not subject to the geopolitical risks often associated with rare earth metals. The robustness of the reaction conditions—tolerating ambient moisture and air—reduces the risk of batch failures due to minor environmental fluctuations. This reliability ensures that production schedules are met consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing drug developers to accelerate their clinical timelines with confidence.
- Scalability and Environmental Compliance: As regulatory pressure mounts for greener manufacturing, this technology positions companies favorably. The absence of heavy metals simplifies environmental compliance and reduces the burden on wastewater treatment facilities. The patent explicitly demonstrates scalability through large-scale examples where enantioselectivity was maintained, proving that the chemistry translates well from gram to kilogram scales. The ease of handling and the potential for continuous flow processing make this an ideal candidate for modern, sustainable manufacturing facilities aiming to minimize their carbon footprint while maximizing output.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of L-prolineamide derivatives in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process chemists and project managers evaluating this technology for adoption.
Q: What are the primary advantages of L-prolineamide derivatives over traditional metal catalysts?
A: Unlike traditional metal catalysts which pose toxicity risks and require complex removal steps, L-prolineamide derivatives are metal-free organic small molecules. They operate under mild conditions, often in wet solvents or air, and can be easily separated and recycled from the product mixture, significantly reducing environmental impact and purification costs.
Q: Can this catalytic system be scaled for industrial production?
A: Yes, the patent data explicitly demonstrates scalability. Examples 59-62 show successful large-scale reactions maintaining high enantioselectivity and diastereoselectivity comparable to small-scale runs. Furthermore, the catalyst can be recovered and reused for at least 5 cycles with minimal loss in activity, supporting cost-effective industrial application.
Q: What is the substrate scope for this asymmetric Aldol reaction?
A: The system exhibits broad substrate universality. It effectively catalyzes reactions between various aromatic and heteroaromatic aldehydes (including those with electron-withdrawing or donating groups) and cyclic ketones like cyclohexanone or cyclopentanone, as well as aliphatic ketones like acetone, delivering high dr and ee values.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Prolineamide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale optimization to full-scale manufacturing is smooth and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our chiral intermediates meets the highest global standards. Whether you require custom synthesis of specific L-prolineamide derivatives or complete process development services, our infrastructure is designed to support your most challenging projects.
We invite you to collaborate with us to leverage this cutting-edge organocatalytic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthesis needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions that drive value and efficiency in your supply chain.
