Advanced One-Step Synthesis of Optically Active Alpha-Hydroxy-Beta-Amino Acids for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive natural products. Patent CN101402582A introduces a groundbreaking methodology for the synthesis of optically active α-hydroxyl-β-amino acids, a critical structural motif present in potent peptidase inhibitors such as amastatin and bestatin. This innovation represents a significant leap forward from traditional multi-step syntheses, offering a robust, one-step four-component reaction that utilizes diazo compounds, alcohols, amines, and aldehydes. By leveraging a sophisticated dual-catalyst system comprising chiral phosphoric acids and either rhodium carboxylates or monovalent copper complexes, this process achieves exceptional levels of stereoselectivity and yield. For R&D directors and procurement managers alike, this technology promises not only a reduction in synthetic complexity but also a substantial optimization of the supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of optically active α-hydroxyl-β-amino acids has been plagued by significant inefficiencies that hinder commercial viability. Traditional routes often rely on multi-step synthesis strategies starting from expensive chiral pool materials like (1R)-8-phenylmenthol glyoxylate or involve cumbersome transformations of epoxy esters. These legacy methods suffer from low overall yields due to the accumulation of losses at each isolation step, requiring extensive purification protocols that drive up manufacturing costs. Furthermore, natural product separation techniques, while sometimes effective, are inherently limited by source availability and batch-to-batch variability, making them unsuitable for the consistent, large-scale supply required by the global pharmaceutical market. The operational complexity of these older methods, often involving harsh conditions and toxic reagents, also poses environmental and safety challenges that modern green chemistry initiatives strive to eliminate.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a convergent multicomponent reaction strategy that assembles the target molecule in a single operational step. This approach capitalizes on the high atom economy of combining four distinct building blocks—diazo compounds, alcohols, amines, and aldehydes—directly into the final chiral framework. 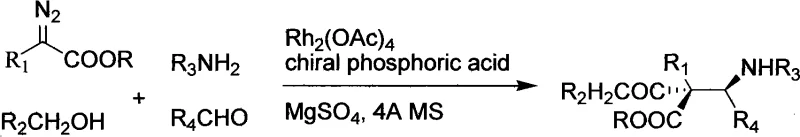 . The reaction is facilitated by a cooperative catalytic system where a chiral phosphoric acid works in tandem with a metal catalyst to control both reactivity and stereochemistry. This eliminates the need for intermediate isolation and protection group manipulation, drastically simplifying the workflow. The use of mild conditions and commercially available starting materials further enhances the practicality of this route, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
. The reaction is facilitated by a cooperative catalytic system where a chiral phosphoric acid works in tandem with a metal catalyst to control both reactivity and stereochemistry. This eliminates the need for intermediate isolation and protection group manipulation, drastically simplifying the workflow. The use of mild conditions and commercially available starting materials further enhances the practicality of this route, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Dual-Catalyzed Asymmetric Synthesis
The success of this transformation lies in the intricate interplay between the organocatalyst and the metal catalyst, which orchestrate the formation of key reactive intermediates with high precision. The mechanism begins with the condensation of the amine and aldehyde to form an imine, which is activated by the chiral phosphoric acid through hydrogen bonding interactions. Simultaneously, the metal catalyst (such as Rh2(OAc)4) decomposes the diazo compound to generate a reactive metal carbene species. This carbene subsequently reacts with the alcohol component to form an oxonium ylide intermediate. 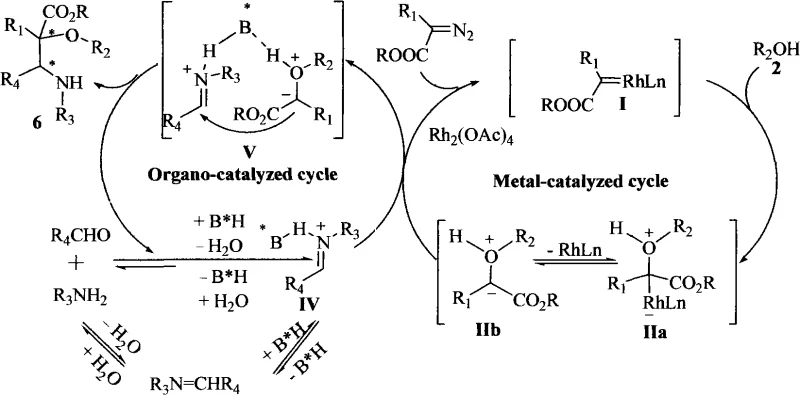 . The chiral environment provided by the phosphoric acid ensures that the nucleophilic attack of the imine on the oxonium ylide occurs with strict stereocontrol, leading to the formation of the desired α-hydroxyl-β-amino acid with high enantiomeric excess. This dual-activation mode effectively lowers the energy barrier for the reaction while maintaining a rigid transition state geometry.
. The chiral environment provided by the phosphoric acid ensures that the nucleophilic attack of the imine on the oxonium ylide occurs with strict stereocontrol, leading to the formation of the desired α-hydroxyl-β-amino acid with high enantiomeric excess. This dual-activation mode effectively lowers the energy barrier for the reaction while maintaining a rigid transition state geometry.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise approaches. Because the reaction proceeds through a concerted pathway under mild temperatures (typically ranging from -20°C to 20°C), the formation of side products associated with thermal decomposition or over-reaction is minimized. The use of molecular sieves and magnesium sulfate as water scavengers drives the equilibrium towards product formation by removing the water generated during imine formation, thereby preventing hydrolysis of sensitive intermediates. The high diastereoselectivity (dr values often greater than 99:1) observed in the examples indicates that the catalyst system effectively discriminates between competing transition states, ensuring that the final product profile is clean and easy to purify. This level of control is critical for meeting the stringent purity specifications required for API intermediates.
How to Synthesize Optically Active Alpha-Hydroxy-Beta-Amino Acids Efficiently
The experimental protocol outlined in the patent provides a clear roadmap for implementing this synthesis in a laboratory or pilot plant setting. The process begins with the preparation of the catalytic mixture, where the amine, aldehyde, and chiral phosphoric acid are dissolved in a suitable organic solvent such as dichloromethane or toluene. Drying agents like 4Å molecular sieves and MgSO4 are added to the reaction vessel to maintain anhydrous conditions, which are essential for the stability of the metal carbene and the efficiency of the imine formation. After stirring at room temperature to allow for imine generation, the metal catalyst and alcohol are introduced, and the mixture is cooled to the optimal reaction temperature. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction system by dissolving amine, aldehyde, and chiral phosphoric acid in an organic solvent, then add molecular sieves and MgSO4 as water scavengers.
- Introduce the metal catalyst (rhodium carboxylate or copper complex) and alcohol to the mixture, cooling the system to a controlled temperature between -20°C and 20°C.
- Slowly add the diazo compound solution over one hour, stir for three hours, quench with saturated NaHCO3, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift from multi-step linear synthesis to a one-step convergent process fundamentally alters the cost structure of producing these valuable intermediates. By reducing the number of unit operations, manufacturers can significantly lower labor costs, energy consumption, and solvent usage, all of which contribute to a more sustainable and cost-efficient production model. The ability to source raw materials from a broad range of commercially available aldehydes and amines also mitigates supply risk, ensuring continuity of supply even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherent in traditional methods leads to a drastic simplification of the manufacturing process. Without the need for expensive chiral starting materials or complex protection-deprotection sequences, the raw material costs are substantially optimized. Furthermore, the high yields and selectivity reported in the patent examples mean that less material is wasted, maximizing the output per batch and reducing the cost of goods sold. This efficiency allows for competitive pricing strategies without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aromatic aldehydes, anilines, and simple alcohols ensures a robust and diversified supply base. Unlike methods dependent on scarce natural extracts or specialized chiral auxiliaries, this four-component reaction can be scaled using widely available feedstocks. This flexibility reduces lead times for high-purity pharmaceutical intermediates and minimizes the risk of production delays caused by raw material shortages. The straightforward workup procedure also facilitates faster turnaround times from synthesis to final delivery.
- Scalability and Environmental Compliance: The operational simplicity of this reaction makes it highly amenable to scale-up from gram to ton quantities. The use of standard organic solvents and the absence of hazardous reagents simplify waste management and regulatory compliance. The high atom economy of the multicomponent reaction aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This sustainability advantage is increasingly important for meeting the corporate social responsibility goals of major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity on the feasibility and advantages of this synthetic route for potential partners and licensees.
Q: What are the primary advantages of this four-component reaction over traditional multi-step synthesis?
A: This method offers superior atom economy and operational simplicity by constructing the complex chiral skeleton in a single step, eliminating the need for tedious protection-deprotection sequences and intermediate isolations found in conventional routes.
Q: How does the dual-catalyst system ensure high enantioselectivity?
A: The synergy between the chiral phosphoric acid, which activates the imine via hydrogen bonding, and the metal catalyst, which generates the reactive oxonium ylide from the diazo compound and alcohol, creates a highly organized transition state that strictly controls stereochemistry.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the use of readily available starting materials like aromatic aldehydes and amines, combined with a simple workup procedure involving solvent removal and chromatography, makes this route highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy-Beta-Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this one-step synthesis can be fully realized at an industrial level. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can accelerate your path to market while optimizing your supply chain efficiency.
