Advanced Synthesis of NDHPI-Modified Polycarboxylic Ligands for High-Performance MOF Catalysts
Advanced Synthesis of NDHPI-Modified Polycarboxylic Ligands for High-Performance MOF Catalysts
The landscape of heterogeneous catalysis is undergoing a significant transformation with the introduction of novel organic ligands designed for Metal-Organic Frameworks (MOFs). Patent CN109734718B discloses a breakthrough class of polycarboxylic acid organic ligands modified based on N,N'-dihydroxypyromellitic diimide (NDHPI). Unlike traditional small-molecule catalysts that suffer from recovery difficulties and active site degradation, these new ligands facilitate the construction of robust, three-dimensional porous materials. The core innovation lies in the direct covalent integration of the NDHPI functional group, which retains dual active N-hydroxyl sites, into a rigid polycarboxylic backbone. This structural integrity ensures that when coordinated with metal nodes, the resulting MOFs exhibit exceptional catalytic efficiency in aerobic oxidations, with reported efficiencies ranging from 94% to 99%. For procurement and R&D teams seeking reliable fine chemical intermediates suppliers, this technology represents a pivotal shift towards sustainable, high-performance catalytic systems.
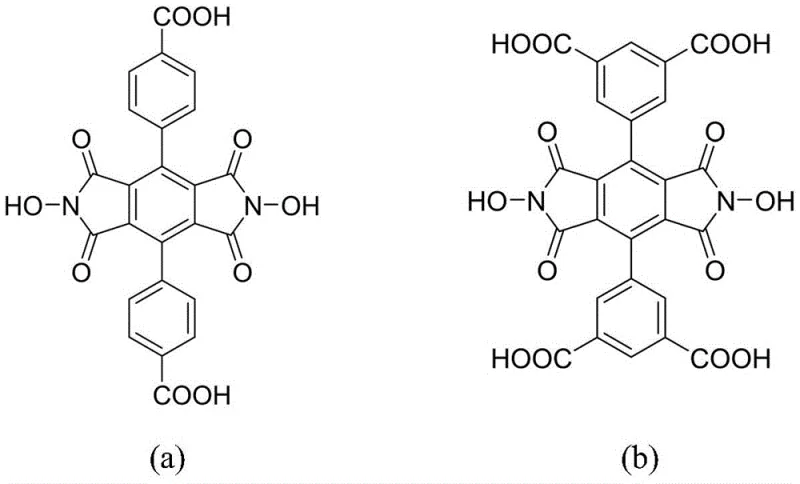
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the application of N-hydroxyphthalimide (NHPI) and its analogues in industrial oxidation has been hampered by their homogeneous nature. While effective, these small molecules are difficult to separate from reaction mixtures, leading to increased downstream processing costs and potential product contamination. Attempts to immobilize these catalysts on solid supports, such as SBA-15 silica, often involve grafting through one of the N-hydroxyl groups. This approach inadvertently destroys 50% of the catalytic active sites, significantly diminishing the overall activity of the material. Furthermore, physical encapsulation methods, where the catalyst is merely trapped within MOF pores without covalent bonding, suffer from severe leaching issues. During continuous flow or batch operations, the active species gradually escape the framework, resulting in a rapid decline in catalytic performance and raising concerns about heavy metal or organic residue in the final pharmaceutical or agrochemical products.
The Novel Approach
The synthetic strategy outlined in the patent data offers a definitive solution to these structural and functional deficiencies. By designing a ligand that inherently contains the NDHPI motif flanked by multiple carboxylic acid groups, the invention enables the formation of MOFs where the catalytic center is an integral part of the framework struts. This covalent integration guarantees that the active sites remain fixed within the pore channels, preventing leaching while maintaining full accessibility to substrates. Crucially, the NDHPI unit preserves both N-hydroxyl groups, effectively doubling the radical generation capacity compared to single-site NHPI analogues. This design not only enhances the turnover frequency of the catalyst but also allows for a substantial reduction in catalyst loading—experimental data suggests that half the amount of this new catalyst can achieve the same conversion rates as traditional NHPI-functionalized materials, driving significant cost reduction in fine chemical manufacturing.
Mechanistic Insights into the Three-Step Synthetic Route
The synthesis of these advanced ligands relies on a highly efficient three-step sequence that maximizes atom economy and minimizes purification burdens. The process initiates with a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. Specifically, 3,6-dibromotetratoluene is coupled with either 4-methylphenylboronic acid or 3,5-dimethylphenylboronic acid using Pd(PPh3)4 as the catalyst and sodium carbonate as the base in a dioxane-water system. This step constructs the rigid terphenyl backbone with high precision, achieving isolated yields of approximately 91% to 93%. The second stage involves the exhaustive oxidation of the peripheral methyl groups to carboxylic acids. Utilizing potassium permanganate (KMnO4) in a pyridine-water mixture at temperatures between 90°C and 110°C, the methyl groups are converted to carboxylates. This oxidation is critical for generating the coordination sites required for MOF assembly and proceeds with impressive efficiency, yielding the polycarboxylic acid intermediate in up to 95% yield after acidification and filtration.
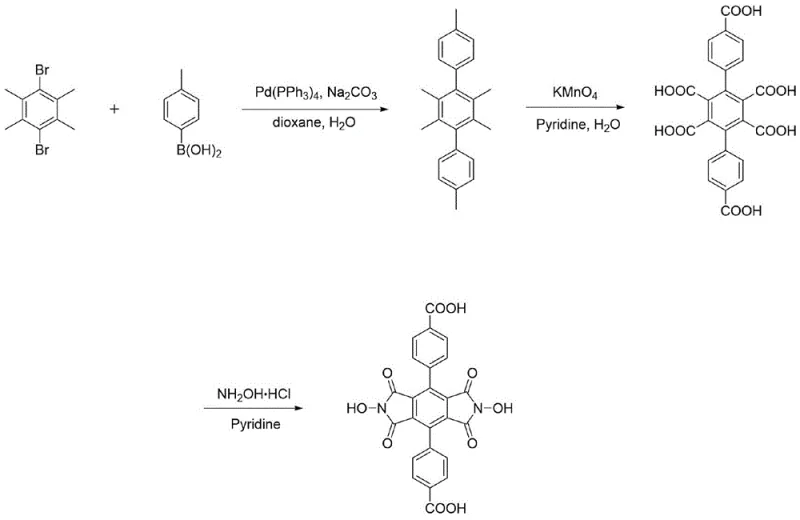
The final and most innovative step is the cyclization to form the imide ring. Traditional methods often require the prior conversion of carboxylic acids to anhydrides, a tedious and moisture-sensitive process. In contrast, this patent describes a direct condensation of the polycarboxylic acid with hydroxylamine hydrochloride in pyridine at 100°C. This streamlined approach bypasses the anhydride formation entirely, reducing reaction time and eliminating the need for dehydrating agents. The result is the direct formation of the N,N'-dihydroxy imide structure with exceptional purity, reported at 97% for the final product. This direct cyclization mechanism is particularly advantageous for scaling up, as it simplifies the operational protocol and reduces the generation of hazardous waste associated with anhydride synthesis, aligning perfectly with modern green chemistry principles.
How to Synthesize NDHPI-Modified Polycarboxylic Ligand Efficiently
The synthesis protocol described above offers a robust pathway for producing high-purity ligands suitable for immediate MOF fabrication. The process is characterized by mild reaction conditions, commercially available starting materials, and straightforward workup procedures involving simple filtration and acidification. For research and development teams aiming to replicate or scale this chemistry, the key lies in maintaining strict control over the oxidation stoichiometry and the final cyclization temperature to ensure complete imide formation without degrading the sensitive N-oxygen bonds. The detailed standardized operating procedures, including specific molar ratios and purification techniques, are essential for achieving the reported high yields and purity levels necessary for catalytic applications.
- Perform palladium-catalyzed Suzuki coupling of 3,6-dibromotetratoluene with methylphenylboronic acid to form the polymethyl terphenyl backbone.
- Oxidize the methyl groups to carboxylic acids using potassium permanganate in a pyridine-water solvent system at 90-110°C.
- React the resulting polycarboxylic acid directly with hydroxylamine hydrochloride in pyridine at 100°C to form the final NDHPI imide structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this NDHPI-based ligand synthesis offers compelling economic and logistical benefits. The elimination of complex protection-deprotection sequences and the avoidance of anhydride intermediates drastically simplify the manufacturing workflow. This simplification translates directly into reduced operational expenditures (OPEX) by lowering energy consumption and minimizing the requirement for specialized drying equipment. Furthermore, the high yields observed at every stage of the synthesis—consistently exceeding 83% and reaching up to 97%—ensure that raw material utilization is optimized. For a reliable fine chemical intermediates supplier, this efficiency means a more stable supply of the final ligand with less batch-to-batch variability, securing the continuity of downstream MOF production.
- Cost Reduction in Manufacturing: The direct reaction of polycarboxylic acids with hydroxylamine hydrochloride removes the need for expensive dehydrating agents and the energy-intensive steps required to form acid anhydrides. This process intensification significantly lowers the cost of goods sold (COGS). Additionally, the dual-active-site nature of the NDHPI ligand means that end-users can achieve target reaction conversions with approximately half the catalyst loading compared to single-site alternatives. This reduction in catalyst demand creates a multiplier effect on cost savings, making the overall catalytic process far more economically viable for large-scale industrial applications such as pharmaceutical intermediate synthesis.
- Enhanced Supply Chain Reliability: The synthetic route utilizes commodity chemicals such as bromotoluenes, phenylboronic acids, and potassium permanganate, all of which are readily available in the global chemical market. There is no reliance on exotic or geopolitically restricted reagents, which mitigates supply chain risk. The robustness of the Suzuki coupling and the subsequent oxidation steps ensures that production can be scaled from kilogram to multi-ton quantities without encountering significant bottlenecks. This scalability is crucial for commercial scale-up of complex polymer additives and catalytic materials, ensuring that procurement managers can secure long-term contracts with confidence in delivery timelines.
- Scalability and Environmental Compliance: The process is inherently greener due to the high atom economy of the coupling reaction and the avoidance of chlorinating agents often used in anhydride synthesis. The workup procedures primarily involve filtration and acidification, generating solid waste (such as manganese dioxide) that can be managed and recycled more easily than complex organic solvent streams. The high purity of the final product (up to 97%) reduces the need for extensive recrystallization or chromatographic purification, thereby minimizing solvent waste. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for companies aiming to reduce their carbon footprint and comply with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NDHPI ligand technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear picture of the material's performance profile. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this catalyst into existing production lines.
Q: What is the primary advantage of NDHPI ligands over traditional NHPI ligands in MOF synthesis?
A: NDHPI ligands possess two active N-hydroxyl sites per molecule compared to the single site in NHPI, effectively doubling the catalytic density within the MOF structure and allowing for reduced catalyst loading in oxidation reactions.
Q: How does this synthesis method improve product purity?
A: The process eliminates the traditional dehydration step to form anhydrides, reacting the polycarboxylic acid directly with hydroxylamine hydrochloride. This simplification reduces side reactions and achieves product purity up to 97%.
Q: Can these ligands be used for large-scale industrial catalysis?
A: Yes, the ligands are designed to form Metal-Organic Frameworks (MOFs) via covalent bonding, which prevents the leaching of active sites common in physically encapsulated catalysts, ensuring long-term stability and reusability in industrial processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NDHPI Ligand Supplier
The synthesis of NDHPI-modified polycarboxylic ligands represents a sophisticated intersection of organic synthesis and materials science, requiring precise control over reaction parameters to achieve the dual active-site configuration. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facility is equipped with rigorous QC labs capable of verifying the structural integrity and purity specifications of these complex ligands, guaranteeing that every batch meets the stringent requirements necessary for high-performance MOF fabrication.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how this technology can optimize your catalytic processes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in catalyst loading and waste disposal costs specific to your operation. We encourage you to contact us for specific COA data and route feasibility assessments to determine the optimal integration strategy for your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
