Advanced Hydrolysis and Extraction Technology for Vitamin B1 Intermediate Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to enhance the purity and yield of critical intermediates, particularly those serving as foundational building blocks for essential vitamins. A significant breakthrough in this domain is documented in patent CN115850045A, which outlines a sophisticated production method for gamma-chloro-gamma-acetopropanol. This compound serves as a pivotal intermediate in the synthetic route of Vitamin B1 (Thiamine), a nutrient of paramount importance for human health and metabolic function. The traditional challenges associated with synthesizing this molecule often revolve around its high solubility in water, which complicates isolation and leads to significant product loss during workup. The novel approach detailed in the patent addresses these inefficiencies by integrating a controlled acidic hydrolysis step followed by a rigorous multi-stage solvent extraction protocol. This strategic combination not only resolves the issue of aqueous solubility but also ensures that the final product meets stringent purity specifications required for pharmaceutical applications. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable gamma-chloro-gamma-acetopropanol supplier capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gamma-chloro-gamma-acetopropanol has been plagued by inherent inefficiencies stemming from the physicochemical properties of the molecule itself. In conventional processes, the hydrolysis of the precursor lactone typically results in a product that remains heavily dissolved in the aqueous reaction medium. This high water solubility creates a formidable barrier to effective isolation, often necessitating energy-intensive evaporation processes or complex salting-out procedures that can degrade the sensitive chloro-acetyl functionality. Furthermore, traditional methods frequently suffer from poor selectivity, leading to the formation of various by-products that contaminate the final stream. These impurities not only reduce the overall yield but also impose a heavy burden on downstream purification steps, increasing both the time and cost associated with manufacturing. For supply chain managers, these limitations translate into unpredictable lead times and higher raw material costs, as significant quantities of starting materials are lost to the aqueous waste stream. The inability to efficiently separate the product from water without compromising its integrity has long been a bottleneck in the cost reduction in vitamin B1 intermediate manufacturing.
The Novel Approach
The methodology presented in the patent offers a transformative solution by re-engineering the separation phase of the synthesis. Instead of relying on brute-force evaporation, the process utilizes a targeted liquid-liquid extraction strategy immediately following the hydrolysis and neutralization steps. By carefully selecting immiscible organic solvents with high partition coefficients for gamma-chloro-gamma-acetopropanol, the method effectively pulls the product out of the aqueous phase while leaving behind water-soluble impurities and inorganic salts. The innovation lies in the optimization of the extraction parameters, including the number of extraction cycles and the mass ratio of the solvent to the reactant. Experimental data indicates that performing multiple extractions, preferably three times, significantly enhances the recovery rate compared to single-step methods. Additionally, the process incorporates a distillation step under reduced pressure to recover the solvent, which can then be recycled back into the system. This closed-loop approach not only minimizes waste generation but also drastically reduces the consumption of fresh solvents, thereby offering substantial economic advantages. The result is a streamlined workflow that delivers high-purity material suitable for immediate use in subsequent synthetic steps without extensive further purification.
Mechanistic Insights into Acidic Hydrolysis and Solvent Extraction
To fully appreciate the technical merit of this production method, one must examine the underlying chemical transformations and separation mechanics. The process initiates with the hydrolysis of alpha-chloro-alpha-acetyl-gamma-butyrolactone under acidic conditions. This reaction involves the nucleophilic attack of water on the carbonyl carbon of the lactone ring, facilitated by the presence of hydrochloric acid. The ring-opening reaction is exothermic and requires precise temperature control, typically maintained between 85°C and 100°C, to ensure complete conversion while preventing the decomposition of the thermally sensitive chloro-ketone moiety. The acidic environment protonates the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon and accelerating the cleavage of the ester bond. Once the ring is opened, the resulting gamma-chloro-gamma-acetyl propanol exists primarily in its hydrated form within the aqueous matrix. Following the reaction, the mixture is neutralized to a pH of approximately 6.0 using sodium bicarbonate. This step is crucial as it quenches the acid catalyst and converts any remaining acidic species into their salt forms, which remain in the aqueous layer, thereby simplifying the subsequent organic extraction.
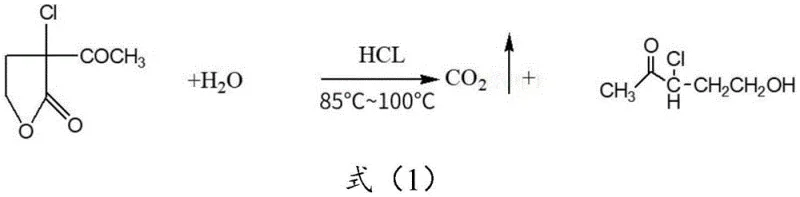
The core of the purification mechanism relies on the principles of solubility and partitioning. Gamma-chloro-gamma-acetopropanol possesses both hydrophilic (hydroxyl group) and hydrophobic (chloro-acetyl chain) characteristics, making it amphiphilic. However, by introducing a water-immiscible organic solvent, the equilibrium shifts towards the organic phase. The patent highlights several effective solvents, including methyl acetate, toluene, hexane, diethyl ether, and dichloromethane. Each solvent interacts differently with the solute based on polarity and hydrogen bonding capabilities. For instance, methyl acetate offers a balance of polarity that effectively solvates the hydroxyl group while maintaining immiscibility with water. The efficiency of this transfer is quantified by the distribution coefficient, which is maximized by performing the extraction in multiple stages. By splitting the total volume of solvent into three separate portions rather than using it all at once, the concentration gradient driving the mass transfer is maintained at a higher level throughout the process. This ensures that even the trace amounts of product remaining in the water after the first pull are recovered in subsequent stages. Finally, the removal of the solvent via vacuum distillation at temperatures ranging from 20°C to 90°C (depending on the solvent's boiling point) yields the pure product, free from the bulk of the aqueous contaminants.
How to Synthesize Gamma-Chloro-Gamma-Acetopropanol Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the acidic hydrolysis medium, where water and hydrochloric acid are heated to the target temperature before the slow addition of the lactone precursor. Controlling the addition rate is vital to manage the exotherm and prevent localized overheating. Once the reaction is complete and the mixture is neutralized, the extraction phase commences. It is recommended to use a separatory funnel for small-scale trials or a continuous extractor for larger batches to ensure thorough mixing of the phases. The choice of solvent should be dictated by availability, cost, and ease of recovery, with methyl acetate and toluene being prime candidates due to their favorable boiling points and extraction efficiencies. After separating the organic layers, they are combined and subjected to distillation. Care must be taken during distillation to avoid thermal degradation of the product; thus, operating under reduced pressure is essential to lower the boiling point of the solvent.
- Hydrolyze alpha-chloro-alpha-acetyl-gamma-butyrolactone in an acidic aqueous solution at 85°C to 100°C to obtain the crude product solution.
- Neutralize the reaction mixture to a pH of approximately 6.0 using sodium bicarbonate after cooling.
- Perform multiple extractions (2 to 5 times) using a selected organic solvent such as methyl acetate or toluene to isolate the product from the aqueous phase.
- Distill the combined organic extracts under reduced pressure to recover the solvent and obtain the final high-purity gamma-chloro-gamma-acetopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the significant optimization of resource utilization. By enabling the recycling of extraction solvents, the process dramatically lowers the recurring cost of raw materials. In traditional linear processes, solvents are often consumed or disposed of after a single use, representing a major line item in the operating budget. Here, the ability to condense and reuse solvents like toluene or methyl acetate creates a circular economy within the manufacturing plant, leading to substantial cost savings over the lifecycle of the production campaign. Furthermore, the high purity of the output, consistently exceeding 97%, reduces the need for expensive downstream purification steps such as recrystallization or column chromatography. This simplification of the workflow shortens the overall production cycle time, allowing facilities to increase throughput without expanding their physical footprint. For a reliable gamma-chloro-gamma-acetopropanol supplier, this efficiency means more stable pricing and the ability to fulfill large orders with shorter lead times.
- Cost Reduction in Manufacturing: The elimination of complex purification stages and the implementation of solvent recycling directly impact the bottom line. By avoiding the use of transition metal catalysts or exotic reagents, the process relies on commodity chemicals that are readily available in the global market. This reduces exposure to price volatility and supply disruptions. Additionally, the high selectivity of the extraction minimizes the generation of hazardous waste, lowering disposal costs and environmental compliance burdens. The qualitative improvement in process efficiency ensures that every kilogram of starting material is converted into valuable product with minimal loss, maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route contributes to a more resilient supply chain. Because the method tolerates a range of solvents and operates under relatively mild conditions, it is less prone to batch failures caused by minor fluctuations in utility supplies or reagent quality. This reliability is crucial for maintaining continuous production schedules, especially when supplying critical intermediates for the pharmaceutical industry where interruptions can halt downstream API manufacturing. The scalability of the process from gram to ton scale ensures that suppliers can ramp up production quickly in response to surges in demand for Vitamin B1, providing a buffer against market shortages.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles. The use of recyclable solvents and the avoidance of heavy metals reduce the ecological footprint of the manufacturing operation. The aqueous waste stream, having been stripped of the organic product, is easier to treat and discharge in compliance with local regulations. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer. The ability to scale this process commercially without generating prohibitive amounts of waste makes it an attractive option for large-scale industrial applications, ensuring long-term viability and sustainability.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this production method is key for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding purity, solvent selection, and scalability, drawing directly from the experimental data and claims within the patent documentation. These insights are intended to provide clarity on how the process performs under various conditions and what expectations buyers should have regarding the quality of the final intermediate.
Q: What is the achievable purity of gamma-chloro-gamma-acetopropanol using this method?
A: The patented production method consistently achieves a purity of greater than or equal to 97%, with optimized examples reaching up to 99.2%. This high purity is critical for ensuring the quality of downstream Vitamin B1 synthesis.
Q: Which extraction solvents are most effective for this process?
A: The process allows for flexibility in solvent selection, including methyl acetate, toluene, hexane, diethyl ether, and dichloromethane. Methyl acetate and toluene have shown particularly high yields and selectivity in experimental trials.
Q: Can this process be scaled for industrial production?
A: Yes, the method is designed for large-scale popularization. The use of recyclable solvents and standard unit operations like hydrolysis and distillation makes it highly suitable for commercial scale-up from pilot plants to multi-ton facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Chloro-Gamma-Acetopropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a theoretical process; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity gamma-chloro-gamma-acetopropanol that meets stringent purity specifications, leveraging advanced analytical techniques in our rigorous QC labs to verify every batch. Our facility is equipped to handle the specific thermal and separation requirements of this hydrolysis-extraction process, guaranteeing a consistent supply of material that supports your Vitamin B1 synthesis needs without interruption.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to see firsthand how our implementation of this patented technology can drive value for your organization. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
