Advanced Manufacturing of 4-Methyl-5-Alkoxyl Oxazole for Global Vitamin B6 Supply Chains
Advanced Manufacturing of 4-Methyl-5-Alkoxyl Oxazole for Global Vitamin B6 Supply Chains
The global demand for high-purity vitamin precursors necessitates robust, scalable, and environmentally compliant synthetic routes. Patent CN102964311A introduces a transformative methodology for the synthesis of 4-methyl-5-alkoxyl oxazole, a critical intermediate in the industrial production of Vitamin B6 (Pyridoxine). This technical insight report analyzes the proprietary two-step sequence that converts readily available 1,1-dialkoxy acetone into the target oxazole derivative via a novel acetyl halide mediated pathway. By eliminating the reliance on toxic heavy metal catalysts and complex multi-step protections found in legacy technologies, this process offers a streamlined approach to heterocyclic chemistry. For R&D directors and procurement strategists, understanding the mechanistic nuances and operational simplicity of this route is essential for optimizing supply chain resilience and reducing the total cost of ownership for fine chemical intermediates.
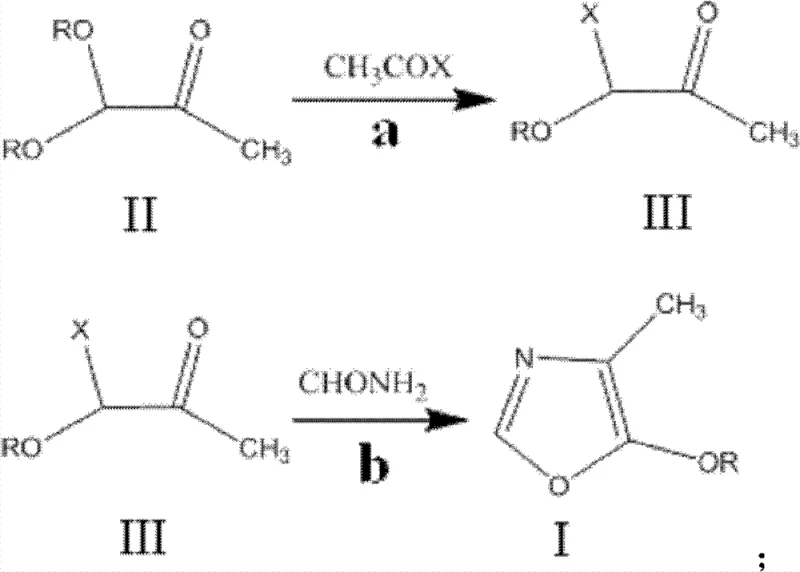
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 4-methyl-5-alkoxyl oxazole has been plagued by significant technical and environmental bottlenecks that hinder efficient large-scale manufacturing. Traditional pathways often rely on the isomerization of alkyl isocyanates, a process that requires harsh heating conditions or strong base catalysis, leading to poor reaction selectivity and difficult-to-remove byproducts. Alternative routes utilizing N-alkyloxy oxalyl alanine alkyl esters involve energy-intensive evaporation steps that drive up utility costs and carbon footprints. Furthermore, methods employing Vanadium Pentoxide (V2O5) as a dehydration condensation agent introduce heavy metal contamination risks, necessitating expensive purification protocols to meet stringent pharmaceutical impurity specifications. Other prior art involving bis(trichloromethyl) carbonate generates substantial hazardous waste streams, creating severe disposal challenges and regulatory compliance burdens for modern chemical facilities aiming for green chemistry certification.
The Novel Approach
In stark contrast to these legacy inefficiencies, the methodology disclosed in CN102964311A utilizes a direct and atom-economical strategy centered on the reactivity of acetyl halides. The process initiates with the reaction of 1,1-dialkoxy acetone with acetyl halides to generate a reactive 1-alkoxy-1-halogenated acetone intermediate, which subsequently undergoes a clean cyclization with formamide. This approach fundamentally shifts the paradigm from complex, waste-generating condensations to a straightforward substitution-cyclization sequence. The ability to conduct these reactions under solvent-free conditions represents a major breakthrough in process intensification, drastically reducing the volume of organic waste and eliminating the need for solvent recovery systems. By leveraging commodity chemicals like acetyl chloride or bromide and formamide, the route ensures raw material security while delivering superior yields and purity profiles that are essential for downstream vitamin synthesis.
Mechanistic Insights into Acetyl Halide Mediated Cyclization
The core of this synthetic innovation lies in the precise control of the electrophilic substitution and subsequent nucleophilic attack. In the first stage, the acetyl halide acts as a source of the acyl group and the halogen, effectively replacing one of the alkoxy groups on the 1,1-dialkoxy acetone substrate. This transformation generates the key 1-alkoxy-1-haloacetone intermediate (Compound III), which possesses the necessary electronic configuration for ring closure. The reaction temperature is critically maintained between 40°C and 80°C to balance reaction kinetics with the stability of the halogenated intermediate, preventing premature decomposition or polymerization. The choice of halogen (Cl or Br) influences the leaving group ability in the subsequent step, allowing process chemists to tune the reactivity based on specific equipment constraints and safety protocols regarding halogenated byproducts.
A critical aspect of the second stage involves the cyclization with formamide to form the oxazole ring, where impurity control is paramount. As illustrated in the mechanistic warning below, excessive formamide or uncontrolled thermal conditions can lead to the formation of imidazole derivatives, a structurally similar impurity that is notoriously difficult to separate from the target oxazole. 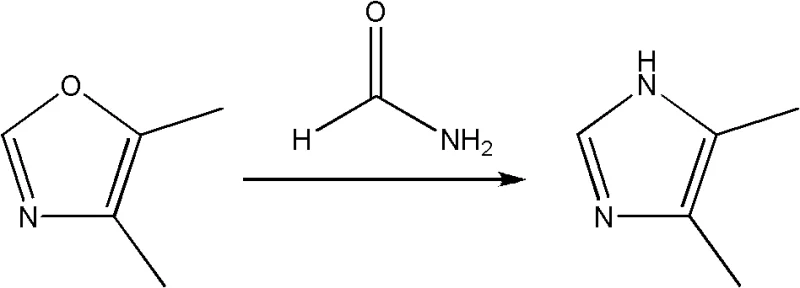 To mitigate this risk, the patent specifies a strict molar ratio of intermediate to formamide, preferably 1:1, and mandates a dropwise addition protocol. This kinetic control ensures that the formamide concentration remains low enough to favor the formation of the five-membered oxazole ring over the thermodynamically stable but undesirable imidazole byproduct. Maintaining the reaction temperature between 100°C and 130°C provides sufficient energy for cyclization without triggering the secondary reactions that degrade product quality.
To mitigate this risk, the patent specifies a strict molar ratio of intermediate to formamide, preferably 1:1, and mandates a dropwise addition protocol. This kinetic control ensures that the formamide concentration remains low enough to favor the formation of the five-membered oxazole ring over the thermodynamically stable but undesirable imidazole byproduct. Maintaining the reaction temperature between 100°C and 130°C provides sufficient energy for cyclization without triggering the secondary reactions that degrade product quality.
How to Synthesize 4-Methyl-5-Alkoxyl Oxazole Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to maximize yield and minimize impurities. The process is designed for scalability, moving seamlessly from laboratory glassware to industrial reactors with minimal modification. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety.
- React 1,1-dialkoxy acetone with acetyl halide at 40-80°C to generate 1-alkoxy-1-haloacetone intermediate.
- Heat the intermediate to 100-130°C and slowly add formamide dropwise to initiate cyclization.
- Perform vacuum distillation to isolate the final 4-methyl-5-alkoxyl oxazole with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of transition metal catalysts like vanadium removes a significant cost center associated with catalyst procurement, usage, and the subsequent removal of trace metals to meet regulatory limits. Furthermore, the solvent-free nature of the reaction dramatically reduces the volume of hazardous waste requiring treatment, leading to substantial cost savings in environmental compliance and waste disposal fees. The use of widely available commodity feedstocks such as acetyl halides and formamide insulates the supply chain from the volatility often seen with specialized reagents, ensuring consistent availability and pricing stability for long-term production contracts.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by operating without organic solvents, which eliminates the capital expenditure and operational costs associated with solvent recovery distillation columns and storage tanks. By removing the need for expensive heavy metal catalysts and complex purification steps required to remove them, the overall cost of goods sold (COGS) is drastically lowered. The high reaction yields reported in the patent examples indicate excellent atom economy, meaning less raw material is wasted, directly translating to improved margin potential for high-volume manufacturing campaigns.
- Enhanced Supply Chain Reliability: The reliance on basic chemical building blocks like acetyl chloride, acetyl bromide, and formamide ensures a robust supply chain that is less susceptible to disruptions compared to routes requiring niche intermediates. These raw materials are produced globally at massive scales, guaranteeing continuity of supply even during market fluctuations. Additionally, the simplified process flow reduces the number of unit operations, shortening the overall production cycle time and allowing for faster turnaround on customer orders, thereby enhancing the agility of the supply chain response.
- Scalability and Environmental Compliance: The solvent-free design inherently aligns with green chemistry principles, making it easier to obtain environmental permits and maintain compliance with increasingly strict global regulations on VOC emissions. The exothermic nature of the reactions is manageable through controlled dropwise addition, facilitating safe scale-up from pilot plants to multi-ton commercial reactors without the need for specialized high-pressure or cryogenic equipment. This operational simplicity reduces the barrier to entry for contract manufacturing organizations looking to onboard this technology for their clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions within the patent documentation to provide accurate guidance for technical teams evaluating this route for potential licensing or production.
Q: What are the critical parameters to prevent imidazole formation during oxazole synthesis?
A: To prevent the formation of imidazole byproducts, the molar ratio of 1-alkoxy-1-haloacetone to formamide must be strictly controlled between 1:1 and 1:1.5. Additionally, formamide should be added dropwise rather than all at once, and reaction temperatures should be maintained between 100-130°C to ensure selective cyclization without over-reaction.
Q: Does this synthesis method require hazardous solvents?
A: No, the patented process described in CN102964311A is specifically designed to operate under solvent-free conditions. Both the initial halogenation step and the subsequent cyclization step can be performed without organic solvents, which significantly reduces VOC emissions and simplifies the post-reaction workup and waste treatment processes.
Q: What is the expected purity profile for the final oxazole product?
A: Using the optimized vacuum distillation protocol described in the patent examples, the final 4-methyl-5-alkoxyl oxazole product consistently achieves a GC purity exceeding 98%. This high purity level minimizes the need for extensive recrystallization or chromatographic purification in downstream Vitamin B6 manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-5-Alkoxyl Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the uninterrupted production of essential vitamins and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 4-methyl-5-alkoxyl oxazole meets the exacting standards required for Vitamin B6 synthesis, providing our partners with the confidence needed for their own regulatory filings.
We invite forward-thinking organizations to collaborate with us on optimizing their supply chains through advanced chemical manufacturing. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
