Optimizing Mebhydrolin Napadisylate Production: A Technical Breakthrough for Global Pharma Supply Chains
Optimizing Mebhydrolin Napadisylate Production: A Technical Breakthrough for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for established antihistamine agents like Mebhydrolin Napadisylate. The recent disclosure in patent CN116410190A introduces a transformative methodology that addresses long-standing stability and purification challenges associated with this active pharmaceutical ingredient (API) intermediate. This technical insight report analyzes the strategic advantages of this novel route, which replaces unstable precursors with a streamlined sequence yielding products with HPLC purity exceeding 99%. For R&D directors and procurement specialists, understanding this shift is critical for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality standards while optimizing manufacturing costs.
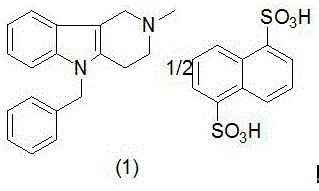
The core innovation lies in the re-engineering of the synthetic backbone to bypass the formation of problematic sulfate salts and unstable hydrazine derivatives. By utilizing a direct salt formation strategy with sodium 1,5-naphthalene disulfonate, the process ensures a white crystalline final product, eliminating the yellow discoloration often seen in legacy methods. This level of control over the impurity profile is essential for downstream formulation, reducing the burden on quality control laboratories and ensuring batch-to-batch consistency. As global demand for effective allergy treatments rises, the ability to produce high-purity antihistamine intermediates efficiently becomes a decisive competitive advantage for any chemical manufacturer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mebhydrolin Napadisylate, as described in earlier patents like CN1590387A, relied heavily on the preparation of benzylphenylhydrazine as a key building block. This intermediate is notoriously unstable, prone to rapid decomposition upon storage, which leads to significant variability in reaction outcomes and increased levels of complex impurities. Furthermore, the conventional route involves the formation of a Mebhydrolin sulfate salt intermediate, which subsequently requires a difficult conversion to the desired naphthalene disulfonate salt. This extra step not only complicates the workflow but often results in incomplete conversion, leaving residual sulfate ions that are difficult to remove and cause the final product to appear slightly yellow. These technical bottlenecks create substantial inefficiencies, increasing purification costs and reducing overall yield, which negatively impacts the cost reduction in API manufacturing for producers relying on outdated chemistry.
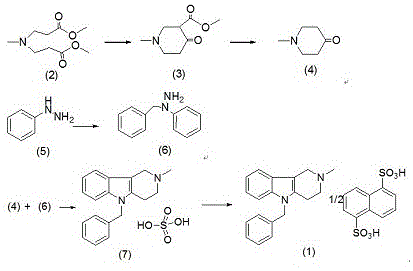
The Novel Approach
In stark contrast, the methodology outlined in CN116410190A fundamentally redesigns the reaction sequence to enhance stability and simplify purification. Instead of generating unstable benzylphenylhydrazine externally, the new process generates the pyrido-indole core directly from N-methyl-4-piperidone hydrochloride and phenylhydrazine under controlled heating. This intermediate, 2-methyl-2,3,4,5-tetrahydro-1H-pyrido[4,3-b]indole, is chemically stable and easy to isolate, providing a solid foundation for the subsequent quaternization step. Crucially, the process eliminates the sulfate salt detour entirely; the quaternary ammonium base reacts directly with sodium 1,5-naphthalene disulfonate to form the final salt. This direct approach minimizes side reactions, ensures complete ion exchange, and guarantees a high-quality white powder, effectively solving the color and purity issues that plagued previous generations of this synthesis.
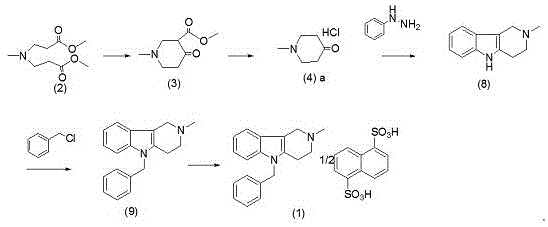
Mechanistic Insights into Pyrido-Indole Cyclization and Quaternization
The heart of this improved synthesis is a modified Fischer indole synthesis variant coupled with a robust quaternization protocol. The process begins with the cyclization of methylimine-N,N-dipropionate methyl ester using sodium methoxide, followed by acid-mediated decarboxylation to yield N-methyl-4-piperidone hydrochloride. This ketone then undergoes condensation with phenylhydrazine, where the hydrazone intermediate cyclizes to form the rigid tricyclic pyrido[4,3-b]indole system. The stability of this core structure is paramount, as it resists the degradation pathways common to open-chain hydrazines. Following isolation, the secondary amine nitrogen within the ring system acts as a nucleophile in the presence of benzyl chloride under alkaline conditions. This quaternization step is highly efficient, creating the cationic center necessary for the final salt formation without generating excessive thermal byproducts.
From an impurity control perspective, the mechanistic elegance of this route lies in its avoidance of competing acid-base equilibria that complicate sulfate-to-disulfonate exchanges. In the old method, the strong binding of sulfate ions often required aggressive washing or recrystallization, leading to product loss. The new method leverages the solubility differences between the organic quaternary base and the inorganic sodium salt byproducts in ethanol-water mixtures. By adjusting the pH to acidic conditions (pH ≤ 1) during the final salt formation, the process drives the equilibrium completely toward the Mebhydrolin Napadisylate precipitate. This precise control over protonation and ion pairing ensures that sulfate contaminants are excluded from the crystal lattice from the outset, resulting in a product that meets rigorous pharmacopeial standards with minimal post-reaction processing.
How to Synthesize Mebhydrolin Napadisylate Efficiently
The implementation of this patented route requires careful attention to solvent selection and temperature profiles to maximize the stability of the intermediates. The process is divided into three distinct operational phases: the preparation of the piperidone precursor, the construction of the heterocyclic core, and the final quaternization and salting-out steps. Each phase has been optimized to use commercially available reagents like toluene, phenylhydrazine, and benzyl chloride, ensuring that the supply chain remains resilient against raw material fluctuations. The detailed standardized synthesis steps below outline the specific conditions required to achieve the reported yields and purity levels, serving as a guideline for process chemists aiming to replicate this high-efficiency workflow.
- Preparation of N-methyl-4-piperidone hydrochloride via decarboxylation of methylimine-N,N-dipropionate methyl ester.
- Cyclization with phenylhydrazine to form the stable 2-methyl-2,3,4,5-tetrahydro-1H-pyrido[4,3-b]indole core.
- Quaternization with benzyl chloride followed by direct salt formation with sodium 1,5-naphthalene disulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route represents a significant opportunity to optimize operational expenditures and mitigate supply risks. The elimination of unstable intermediates like benzylphenylhydrazine removes a major source of batch failure and rework, which historically drove up the effective cost of goods sold. By stabilizing the supply of key precursors and simplifying the reaction sequence, manufacturers can achieve a more predictable production schedule, reducing the lead time for high-purity pharmaceutical intermediates. Furthermore, the removal of the sulfate salt conversion step reduces the consumption of auxiliary chemicals and water, aligning production with modern environmental sustainability goals while lowering waste disposal costs.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive purification steps required to remove sulfate ions and decomposition byproducts found in legacy methods. By avoiding the isolation of unstable hydrazine derivatives, the process reduces material loss due to degradation, thereby improving the overall mass balance and yield. This efficiency translates directly into lower unit production costs, allowing for more competitive pricing strategies in the global market without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates ensures that production can be paused and resumed without the risk of raw material spoilage, a common issue with sensitive hydrazine compounds. This stability enhances inventory management capabilities, allowing suppliers to maintain safety stock of intermediates rather than just finished goods. Consequently, this leads to improved delivery reliability and the ability to respond rapidly to sudden spikes in market demand for antihistamine medications.
- Scalability and Environmental Compliance: The use of common organic solvents like toluene and ethanol, combined with the avoidance of harsh sulfate conversion conditions, makes this process highly scalable from pilot plant to multi-ton commercial production. The simplified workup procedures reduce the volume of aqueous waste streams containing high concentrations of inorganic salts, facilitating easier wastewater treatment and compliance with increasingly strict environmental regulations. This green chemistry approach future-proofs the manufacturing asset against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Mebhydrolin Napadisylate using this advanced methodology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on how this route compares to traditional synthesis methods. Understanding these nuances is vital for technical teams evaluating potential partners for long-term supply agreements.
Q: Why is the new synthesis route for Mebhydrolin Napadisylate superior to previous methods?
A: The new route eliminates the use of unstable benzylphenylhydrazine and avoids the formation of sulfate salt intermediates, resulting in higher purity (>99% HPLC) and better product color compared to traditional methods.
Q: What are the key impurities controlled in this patented process?
A: The process specifically addresses sulfate ion contamination and decomposition products from unstable hydrazine intermediates, ensuring a white crystalline powder with minimal degradation byproducts.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent highlights mild reaction conditions, stable intermediates, and simplified purification steps, which are critical factors for successful commercial scale-up and energy efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mebhydrolin Napadisylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like CN116410190A is essential for maintaining competitiveness in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in large-scale manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of Mebhydrolin Napadisylate meets the >99% HPLC purity benchmark, guaranteeing performance consistency for your downstream formulations.
We invite global partners to collaborate with us on optimizing their supply chains through the adoption of this superior technology. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
