Advanced Catalytic Epoxidation for High-Purity Alicyclic Epoxy Resin Manufacturing
The global demand for high-performance electronic materials continues to surge, driven by the rapid expansion of the LED industry and advanced coating technologies. In this context, Patent CN111138383A introduces a transformative approach to synthesizing alicyclic epoxy resin, a critical component known for its exceptional electrical insulation and ultraviolet resistance. This patent details a sophisticated catalytic oxidation method that overcomes the longstanding limitations of traditional peroxyacid oxidation and earlier catalytic systems. By leveraging organic heteropolyacid salts combined with a novel alkali metal carbonate post-treatment, the invention achieves a level of purity and color stability previously difficult to attain. This technical breakthrough is particularly relevant for manufacturers seeking a reliable electronic chemical supplier capable of delivering materials that meet the rigorous standards of optoelectronic applications.
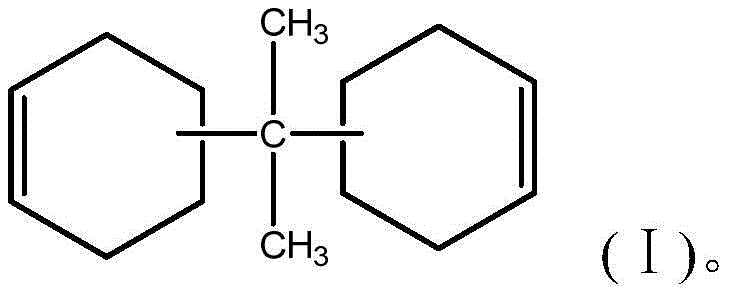
The core innovation lies in the precise management of the catalyst throughout the reaction lifecycle. Unlike conventional methods where catalyst removal is inefficient, leading to product degradation during purification, this process ensures the catalyst is rendered water-soluble and easily separable. The result is an alicyclic epoxy resin with superior optical properties, making it an ideal candidate for UV-resistant coatings and encapsulation materials in sensitive electronic devices. For R&D teams focusing on material performance, this patent offers a robust pathway to high-quality intermediates without the compromise of thermal degradation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of alicyclic epoxy resins has been plagued by significant technical and safety challenges. The traditional peroxyacid oxidation method relies on anhydrous peroxyacetic acid or high-concentration peroxyorganic acids as oxidants. These reagents are inherently unstable and present severe safety hazards, including the risk of explosion, necessitating extremely harsh production conditions and specialized containment infrastructure. Furthermore, the catalytic oxidation methods that emerged as alternatives often utilized metal compounds that were difficult to separate completely from the organic phase. Residual catalysts in the product would frequently cause discoloration or induce polymerization into high molecular weight byproducts during the rectification stage. This not only compromised the visual quality of the resin but also necessitated high-temperature distillation, which further degraded the active epoxy groups and reduced overall selectivity.
The Novel Approach
The methodology described in Patent CN111138383A fundamentally re-engineers the post-reaction processing to eliminate these defects. Instead of relying on reducing agents like sodium sulfite or sodium thiosulfate to neutralize excess hydrogen peroxide, this novel approach employs alkali metal carbonates, such as sodium carbonate or potassium carbonate. This alkaline treatment serves a dual purpose: it promotes the thorough decomposition of residual hydrogen peroxide and reacts with the remaining heteropolyacid catalyst to form water-soluble salts. This chemical transformation allows the catalyst to be effectively washed away from the oil phase containing the alicyclic epoxy resin. Additionally, the integration of adsorbents like activated carbon or clay further scavenges impurities. Consequently, the rectification process can be conducted at significantly lower temperatures, preserving the integrity of the epoxy ring and ensuring the final product maintains excellent color and luster.
Mechanistic Insights into Heteropolyacid-Catalyzed Epoxidation
The catalytic mechanism employed in this synthesis is a masterclass in phase-transfer efficiency and selective oxidation. The organic heteropolyacid salt catalyst exhibits unique solubility characteristics; it is initially insoluble in the organic solvent but forms a soluble species in the presence of hydrogen peroxide. This allows the reaction to proceed homogeneously, maximizing the contact between the catalyst and the alicyclic olefin substrate, which leads to high conversion rates and selectivity. The specific structure of the heteropolyacid, often containing tungsten or molybdenum atoms coordinated with organic cations, facilitates the transfer of oxygen from the peroxide to the double bond of the alicyclic ring. This selective epoxidation is crucial for maintaining the structural integrity of the complex alicyclic framework, ensuring that side reactions such as ring-opening or over-oxidation are minimized.
Impurity control is achieved through a sophisticated chemical switch triggered during the workup phase. Upon completion of the oxidation, the addition of alkali metal carbonate alters the chemical environment of the catalyst. The basic conditions cause the organic heteropolyacid salt to revert to a state where it forms water-soluble inorganic salts or precipitates, effectively breaking its association with the organic product phase. This mechanism prevents the catalyst from acting as a Lewis acid during the subsequent heating stages, which is the primary cause of discoloration and polymerization in conventional processes. By adsorbing any remaining trace metals or organic impurities onto the added clay or activated carbon, the process ensures that the organic phase entering the rectification column is exceptionally clean, thereby guaranteeing the high purity required for electronic grade applications.
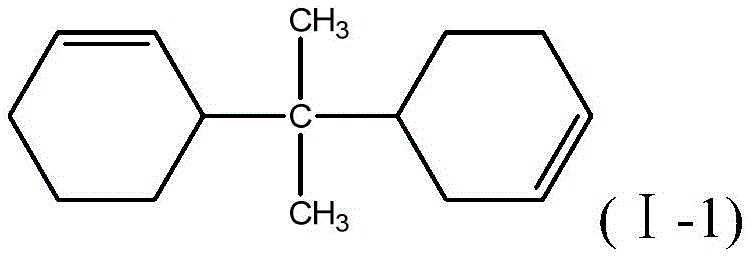
How to Synthesize Alicyclic Epoxy Resin Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing high-quality alicyclic epoxy resin. The process begins with the preparation of a reaction mixture containing the alicyclic olefin, the specific organic heteropolyacid catalyst, an adsorbent, and a suitable organic solvent such as dichloroethane or acetonitrile. Hydrogen peroxide is then introduced dropwise at controlled temperatures between 40°C and 60°C to initiate the epoxidation. Following the reaction, the critical post-treatment step involves the addition of an aqueous alkali metal carbonate solution and additional adsorbent to scrub the organic phase. This detailed procedure ensures that the catalyst is deactivated and removed before the final purification step, which involves vacuum rectification at mild temperatures to isolate the pure resin. For a comprehensive guide on the specific molar ratios and operational parameters, please refer to the standardized synthesis steps provided below.
- Mix alicyclic olefin compounds, organic heteropolyacid salt catalyst, adsorbent, and organic solvent to form a homogeneous mixture ready for oxidation.
- Add hydrogen peroxide to the mixture at controlled temperatures (40-60°C) to facilitate selective epoxidation, followed by solid-liquid separation to isolate the organic phase.
- Treat the organic phase with alkali metal carbonate and water to decompose residual peroxide and convert the catalyst into water-soluble salts for effective removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical performance. The shift from hazardous peroxyacids to stable hydrogen peroxide significantly lowers the barrier for safe manufacturing, reducing the need for expensive safety infrastructure and insurance costs associated with explosive reagents. Furthermore, the enhanced efficiency of catalyst removal translates directly into cost reduction in electronic chemical manufacturing. By eliminating the need for complex heavy metal scavenging procedures or high-energy high-temperature distillation to remove stubborn impurities, the overall production cost is drastically simplified. The ability to produce resin with superior color without extensive downstream purification means fewer processing steps, lower energy consumption, and reduced waste generation, all of which contribute to a more sustainable and economically viable supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous peroxyacid reagents in favor of hydrogen peroxide represents a direct material cost saving. Moreover, the efficient removal of the catalyst via alkali carbonate treatment removes the necessity for costly transition metal scavengers often required in traditional catalytic processes. This streamlined purification workflow reduces the consumption of auxiliary chemicals and minimizes the loss of product during multiple washing or filtration cycles, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The use of mild reaction conditions (40-60°C) and stable reagents enhances the robustness of the production process, reducing the likelihood of batch failures due to thermal runaway or reagent instability. This reliability ensures consistent delivery schedules and reduces the risk of supply disruptions caused by safety incidents or equipment downtime. Additionally, the raw materials required, such as hydrogen peroxide and common alkali carbonates, are commodity chemicals with stable global supply chains, further securing the continuity of production for high-purity alicyclic epoxy resins.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory bench scale to multi-ton commercial production without significant re-engineering. The reduction in hazardous waste, particularly the avoidance of sulfur-containing byproducts from traditional reducing agents, simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the marketability of the final product to environmentally conscious downstream customers in the electronics and coatings sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alicyclic epoxy resin synthesis technology. These insights are derived directly from the experimental data and technical disclosures within Patent CN111138383A, providing clarity on how this method resolves historical pain points in resin manufacturing. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this high-purity intermediate into their existing supply chains.
Q: How does the new post-treatment method improve product color compared to traditional reduction methods?
A: Traditional methods use reducing agents like sodium sulfite which often fail to completely remove the heteropolyacid catalyst, leading to discoloration during high-temperature rectification. The novel method uses alkali metal carbonate to convert the catalyst into water-soluble salts, ensuring thorough removal and preventing polymerization or discoloration.
Q: What are the safety advantages of this catalytic oxidation process over peroxyacid methods?
A: Conventional peroxyacid oxidation requires anhydrous peroxyacetic acid or high-concentration peroxyorganic acids, which pose significant explosion risks and require harsh production conditions. This patented method utilizes hydrogen peroxide under mild conditions (40-60°C), drastically reducing potential safety hazards in the manufacturing environment.
Q: Can this synthesis route be scaled for industrial production of electronic grade resins?
A: Yes, the process is designed for scalability. It avoids high-temperature rectification which can degrade sensitive epoxy groups, and the efficient catalyst removal system ensures consistent high purity and low color number, which are critical specifications for electronic grade alicyclic epoxy resins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alicyclic Epoxy Resin Supplier
As the electronics industry demands ever-higher purity standards for materials like alicyclic epoxy resin, partnering with an experienced CDMO is crucial for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of resin meets the exacting requirements for optical clarity and electrical performance needed in LED and semiconductor applications.
We invite you to collaborate with us to optimize your material sourcing strategy. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
