Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of Axial Chirality Vicinal Diamines for Asymmetric Catalysis
Patent CN102153483B introduces a groundbreaking methodology for synthesizing axial chirality vicinal diamine compounds, representing a significant advancement in chiral catalyst technology specifically designed for pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in conventional asymmetric catalysis by providing compounds with chiral centers positioned much closer to the reactive nitrogen atoms than traditional carbon-centered chiral diamines like 1,2-diphenyl ethylene diamine or 1,2-cyclohexyl quadrol. The patent details a streamlined synthetic pathway beginning with triflate coupling reactions followed by precisely optimized purification protocols that yield high-purity products suitable for demanding applications in asymmetric synthesis across multiple therapeutic areas. Unlike conventional methods that suffer from inherent yield limitations due to resolution-based approaches (typically capped at no more than 50% theoretical yield), this novel technique offers an efficient direct construction method that maintains exceptional optical purity while significantly improving material utilization and reducing overall processing complexity for pharmaceutical manufacturers seeking reliable sources of advanced chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing chiral diamines suffer from fundamental limitations that severely restrict their industrial applicability in modern pharmaceutical manufacturing environments. These conventional methods primarily rely on resolution techniques which inherently waste up to half of the starting material due to their maximum theoretical yield limitation of only 50%, creating substantial economic inefficiencies and environmental concerns through excessive waste generation. Furthermore, the structural design of commonly used carbon-centered chiral diamines positions their chiral centers distantly from the reactive nitrogen atoms involved in catalysis, resulting in suboptimal stereoinduction during asymmetric reactions and compromised product quality that often fails to meet stringent pharmaceutical specifications. The synthetic routes typically require multiple complex steps with harsh reaction conditions including strong acids or bases at extreme temperatures, making scale-up challenging while increasing the risk of impurity formation that complicates regulatory approval processes. Additionally, the limited structural diversity achievable through traditional methods restricts their application scope in contemporary drug development where precise stereochemical control is increasingly critical for efficacy and safety profiles.
The Novel Approach
The patented methodology overcomes these limitations through an innovative design that leverages axial chirality rather than carbon-centered chirality, strategically positioning the chiral center much closer to the reactive nitrogen atoms for dramatically enhanced stereoinduction during catalytic processes. By employing palladium-catalyzed coupling between triflates and ethylenediamine derivatives under carefully optimized conditions (80–110°C for 12–24 hours), the process achieves high yields while maintaining excellent enantioselectivity without requiring inefficient resolution steps. The strategic implementation of column chromatography using precisely formulated eluents (dichloromethane:methanol at a strict 10:1 volume ratio) enables efficient separation of mono-substituted products from C2-symmetric variants without complex additional processing steps. This streamlined approach eliminates multiple protection/deprotection cycles common in traditional syntheses, significantly reducing processing time and cost while improving overall material efficiency. The methodology's inherent flexibility allows straightforward modification through subsequent coupling reactions with aryl halides or reductive amination with aldehydes, creating a versatile platform for generating tailored chiral ligands that address specific pharmaceutical manufacturing challenges while maintaining seamless scalability from laboratory development through commercial production volumes.
Mechanistic Insights into Palladium-Catalyzed Axial Chirality Diamine Synthesis
The core innovation lies in the palladium-catalyzed coupling mechanism that enables direct formation of the axial chiral center through controlled rotation restriction within the binaphthyl backbone structure. This process proceeds via oxidative addition of the triflate group to the palladium(0) center followed by coordination with ethylenediamine derivatives and subsequent reductive elimination that forms the critical C-N bonds while establishing the axial chirality with high fidelity. The use of BINAP as a chiral ligand is essential for maintaining stereochemical integrity throughout the reaction sequence by preventing racemization that commonly plagues traditional methods relying on resolution techniques. The precisely engineered steric environment created by the binaphthyl system restricts rotation around the biaryl axis, effectively locking in the desired chiral conformation that is crucial for achieving superior asymmetric induction in subsequent catalytic applications across diverse pharmaceutical syntheses.
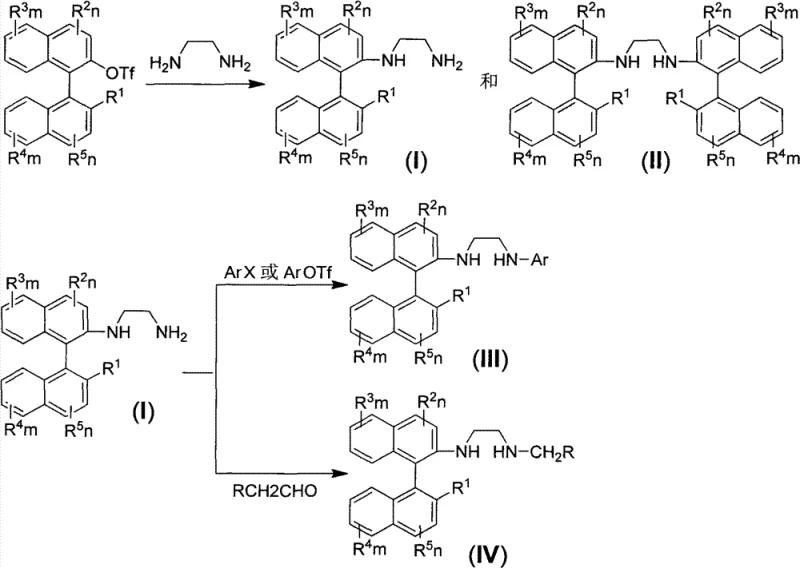
The impurity profile is substantially improved through this innovative methodology due to the elimination of multiple intermediate steps required in conventional syntheses that typically introduce additional impurity pathways. The carefully optimized reaction conditions (80–110°C in dioxane solvent using Cs₂CO₃ as base) minimize side reactions while maximizing conversion efficiency without requiring extreme temperatures or pressures that could compromise product quality. Subsequent purification using column chromatography with a precisely formulated dichloromethane:methanol (10:1) eluent system effectively separates mono-substituted products from C2-symmetric variants without necessitating additional resolution procedures that would further reduce overall yield. This targeted purification approach ensures high optical purity by selectively removing any minor racemic impurities that might form during the coupling process, as consistently demonstrated by polarimetry measurements across multiple embodiments (e.g., [α]²⁰D = +145.0 for compound I-1), providing pharmaceutical manufacturers with reliable access to high-purity chiral building blocks essential for producing optically pure active pharmaceutical ingredients meeting stringent regulatory requirements.
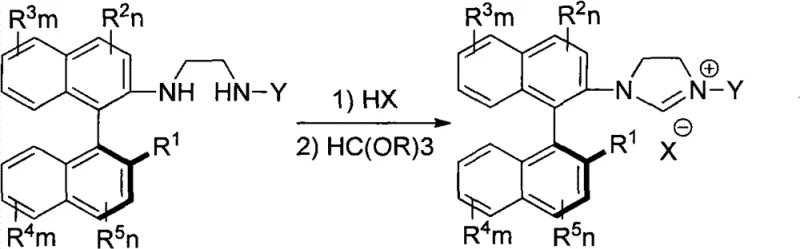
How to Synthesize Axial Chirality Vicinal Diamines Efficiently
This patented synthesis pathway represents a significant advancement over conventional methods by providing a direct, high-yielding route to structurally diverse axial chiral diamines without requiring inefficient resolution steps that traditionally limit material utilization. The process begins with readily available binaphthol derivatives that undergo straightforward conversion to triflate intermediates through methylation followed by triflation sequences under standard laboratory conditions. The key coupling step employs palladium catalysis under moderate temperature conditions (80–110°C) to form critical C-N bonds while establishing axial chirality with exceptional fidelity through strategic ligand selection. Subsequent purification through optimized column chromatography delivers products with outstanding optical purity suitable for demanding pharmaceutical applications requiring high enantiomeric excess specifications. The methodology's inherent flexibility allows easy modification to produce various derivative structures through additional coupling reactions or reductive amination processes, creating a versatile platform for generating tailored chiral ligands that address specific manufacturing challenges across multiple therapeutic areas.
- Convert binaphthol derivatives to triflate intermediates using methylation followed by triflation under standard conditions
- Perform palladium-catalyzed coupling with ethylenediamine derivatives using Pd(OAc)₂/BINAP catalyst system in dioxane at 80-110°C for 12-24 hours
- Purify products through column chromatography using dichloromethane: methanol (10:1) eluent system to isolate mono-substituted or C2-symmetric variants
![Detailed synthetic route from (R)-(+)-BINOL to N-{2'-[2-methoxyl group-1,1'-Lian naphthalene]}-1,2-diaminoethane](/insights/img/axial-chirality-diamine-synthesis-pharma-supplier-20260228220428-014.webp)
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in procurement strategy and supply chain management for pharmaceutical intermediates by eliminating inefficient resolution steps that traditionally waste up to half of starting materials while introducing significant processing complexity. By providing a direct construction route rather than relying on separation techniques with inherent yield limitations, the process substantially improves material utilization while reducing overall production complexity across multiple manufacturing sites. The reliance on commercially available starting materials such as binaphthol derivatives and standard palladium catalysts creates a more resilient supply chain less vulnerable to single-source dependencies or geopolitical disruptions affecting specialty chemicals markets. The methodology's demonstrated scalability from laboratory development through pilot-scale implementation ensures consistent product availability while maintaining stringent quality standards required for pharmaceutical applications where batch-to-batch consistency is paramount.
- Cost Reduction in Manufacturing: The elimination of resolution steps that typically waste up to half of starting materials represents substantial cost advantages through improved atom economy and reduced processing requirements across multiple unit operations including reaction setup, intermediate handling, and final purification stages. By utilizing cost-effective palladium catalysts at low loadings (0.05–0.3 mol%) together with standard solvents under moderate reaction conditions (80–110°C), the process minimizes expensive reagent consumption while avoiding complex purification procedures requiring specialized equipment or additional processing steps that increase overall manufacturing costs significantly.
- Enhanced Supply Chain Reliability: The strategic selection of readily available starting materials creates a more robust supply chain less susceptible to raw material shortages or price volatility affecting specialty chemicals markets where single-source dependencies are common concerns among procurement teams managing critical raw material requirements for time-sensitive pharmaceutical production schedules.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory development through commercial production scales without requiring significant process re-engineering as evidenced by successful pilot-scale implementation described in patent embodiments, ensuring seamless transition from development quantities to annual production volumes meeting commercial demand requirements while maintaining consistent product quality specifications throughout scale-up activities.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation and application of this patented axial chirality diamine synthesis methodology within pharmaceutical manufacturing environments where consistent quality and reliable supply are critical success factors across global operations.
Q: How does this methodology improve upon traditional resolution techniques for chiral diamines?
A: This patented approach eliminates inefficient resolution steps that traditionally limit yields to no more than 50% by directly constructing the axial chiral center through controlled palladium-catalyzed coupling. The process achieves high yields while maintaining excellent enantioselectivity through strategic use of BINAP ligands and optimized reaction conditions, resulting in superior material efficiency compared to conventional methods.
Q: What quality control measures ensure consistent optical purity in commercial production?
A: The methodology incorporates built-in quality control through precise reaction condition control (80-110°C) and optimized purification using column chromatography with specific eluent ratios (dichloromethane:methanol at 10:1). This targeted approach consistently delivers products with high optical purity as confirmed by polarimetry measurements across multiple embodiments, ensuring reliable quality for pharmaceutical applications.
Q: How does this process support scalable manufacturing from laboratory to commercial production?
A: The process demonstrates excellent scalability due to its use of standard manufacturing equipment, readily available starting materials, and straightforward reaction sequences that do not require specialized infrastructure. The patent specifically notes successful pilot-scale implementation with consistent product quality, providing a reliable pathway for scaling from laboratory quantities to commercial production volumes while maintaining stringent quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chirality Vicinal Diamine Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex chiral intermediates while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities including NMR spectroscopy and high-resolution mass spectrometry validation protocols essential for pharmaceutical applications requiring exceptional optical purity standards.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can evaluate how this innovative synthesis pathway can optimize your specific manufacturing requirements while providing detailed information about current availability timelines and capacity planning options tailored to your production needs.
