Optimizing Desmopressin Acetate Production: A Technical Breakthrough in Solid-Phase Synthesis and Liquid-Phase Oxidation
Optimizing Desmopressin Acetate Production: A Technical Breakthrough in Solid-Phase Synthesis and Liquid-Phase Oxidation
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for complex peptide hormones like Desmopressin Acetate. A pivotal advancement in this domain is detailed in patent CN103102395A, which discloses a sophisticated preparation method combining the precision of solid-phase synthesis with the controllability of liquid-phase oxidation. This hybrid approach addresses longstanding challenges in peptide manufacturing, specifically targeting the issues of low yield and difficult purification associated with traditional methods. By integrating reversed-phase high-performance liquid chromatography (RP-HPLC) purification prior to the critical oxidation step, the process ensures that only high-quality linear peptides undergo cyclization. This technical insight is crucial for R&D Directors and Procurement Managers looking for a reliable Desmopressin Acetate supplier who can guarantee consistent quality and supply continuity for central diabetes insipidus and hemophilia treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Desmopressin has been plagued by inefficiencies inherent in both traditional liquid-phase and early solid-phase methodologies. Conventional liquid-phase synthesis, as referenced in older literature like U.S. Pat. No. 3497491, requires a stepwise coupling of amino acids under alkaline conditions, necessitating purification after every single coupling step. This iterative process is not only time-consuming but also operationally cumbersome, leading to significantly low overall yields that are economically unviable for large-scale industrial production. Furthermore, alternative solid-phase methods often struggle with the oxidation step; when using iodine for solid-phase oxidation to form the disulfide bond, the substitution degree of the resin must be kept low to prevent intermolecular cross-linking. This limitation restricts the loading capacity of the resin, thereby increasing the volume of solvents and reagents required per unit of product, which drastically escalates production costs and environmental waste burdens for manufacturing facilities.
The Novel Approach
The innovative method described in the patent data overcomes these bottlenecks by decoupling the synthesis and oxidation phases. Instead of attempting oxidation on the resin, this protocol synthesizes the linear desmopressin peptide resin, cleaves it to obtain the crude linear peptide, and critically, purifies this linear intermediate via RP-HPLC before any oxidation occurs. This strategic intervention removes insoluble impurities and deletion sequences that would otherwise hinder the oxidation reaction. By performing the oxidation in the liquid phase using agents like hydrogen peroxide or potassium ferricyanide at a controlled pH of 7.0-8.0, the process avoids the steric hindrance and intermolecular bonding issues of solid-phase oxidation. This allows for the use of resins with higher substitution degrees, theoretically reducing the cost of goods sold (COGS) while simultaneously enhancing the purity profile of the final Active Pharmaceutical Ingredient (API).
Mechanistic Insights into Fmoc/tBu Solid-Phase Synthesis and Liquid-Phase Oxidation
The core of this synthesis relies on the Fmoc/tBu orthogonal protection strategy, utilizing Sieber Amide resin as the solid support. The process begins with the preparation of Fmoc-Gly-Sieber Amide resin, followed by the sequential coupling of protected amino acids including Fmoc-D-Arg(Pbf)-OH, Fmoc-Pro-OH, and Fmoc-Cys(Trt)-OH. The precise arrangement of these residues is critical for the biological activity of the final hormone. The use of Fmoc chemistry allows for mild deprotection conditions using piperidine, which preserves the acid-labile side-chain protecting groups until the final cleavage step. This meticulous control over the peptide elongation cycle minimizes the formation of racemization byproducts and ensures the correct stereochemistry at the 8-D-arginine position, which is essential for the antidiuretic potency of Desmopressin compared to native vasopressin.
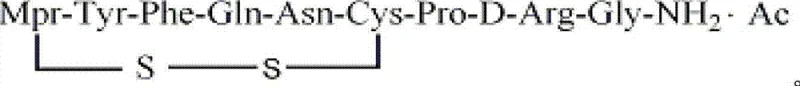
Following the assembly of the linear peptide resin, the cleavage step employs a cocktail of trifluoroacetic acid, triisopropylsilane, 1,2-ethanedithiol, and water to release the crude peptide. Unlike prior art methods that oxidize immediately, this protocol introduces a purification stage where the crude linear peptide is subjected to RP-HPLC. The mobile phase typically consists of acidic aqueous solutions like 0.1v/v% trifluoroacetic acid and acetonitrile. This step is mechanistically vital because it removes hydrophobic impurities and truncated sequences that could compete during the oxidation phase. The subsequent liquid-phase oxidation utilizes the free thiol groups of the cysteine residues to form the intramolecular disulfide bond. By controlling the pH between 7.0 and 8.0 and using mild oxidants, the reaction favors intramolecular cyclization over intermolecular polymerization, resulting in a cyclic peptide with superior chromatographic purity ready for salt conversion.
How to Synthesize Desmopressin Acetate Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding resin substitution and purification gradients to ensure reproducibility at scale. The process begins with swelling the Sieber Amide resin and coupling the first amino acid, followed by iterative deprotection and coupling cycles monitored by the Kaiser test to ensure completeness. Once the linear sequence is assembled, cleavage and precipitation yield the crude linear peptide, which must be dissolved in acidic media for chromatographic purification. The purified linear peptide is then collected, lyophilized, and redissolved for the oxidation step where pH control is paramount to prevent over-oxidation or sulfone formation. Detailed standardized synthesis steps see the guide below.
- Synthesize linear desmopressin peptide resin using Fmoc/tBu strategy on Sieber Amide resin with controlled substitution degrees.
- Cleave the resin to obtain crude linear peptide, followed by RP-HPLC purification to remove insoluble impurities before oxidation.
- Perform liquid-phase oxidation at pH 7.0-8.0 to form the disulfide bond, followed by salt conversion to yield Desmopressin Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hybrid synthesis method represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing without compromising quality standards. The ability to use resins with higher substitution degrees directly translates to a reduction in the volume of solid support required per batch, which lowers raw material costs and reduces the physical footprint of the reaction vessels needed for production. Furthermore, by purifying the linear peptide before oxidation, the process minimizes the formation of difficult-to-remove oxidative byproducts, thereby reducing the burden on downstream purification processes and decreasing the consumption of expensive chromatography solvents. This efficiency gain is critical for maintaining competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of complex solid-phase oxidation controls and the ability to use higher loading resins significantly streamline the production workflow. By removing the need for multiple purification steps after every amino acid coupling as seen in liquid-phase synthesis, and avoiding the low yields of traditional solid-phase oxidation, the overall process efficiency is drastically improved. This qualitative improvement in yield and material utilization leads to substantial cost savings in reagent consumption and waste disposal, making the final Desmopressin Acetate more economically viable for large-scale procurement.
- Enhanced Supply Chain Reliability: The robustness of the Fmoc/tBu strategy combined with liquid-phase oxidation ensures a more consistent production output, reducing the risk of batch failures that can disrupt supply chains. Since the oxidation step is performed in solution where parameters like pH and temperature are easier to monitor and control than on a solid support, the process offers greater reproducibility. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug product manufacturers receive their active ingredients on schedule without quality-related delays.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex peptide hormones, as evidenced by the successful transition from gram-scale examples to potential metric-ton production. The reduction in waste liquid generation, achieved by removing impurities prior to oxidation and increasing sample concentration, aligns with modern environmental compliance standards. This reduced environmental footprint simplifies the regulatory approval process for manufacturing sites and ensures long-term sustainability of the supply chain for this critical medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Desmopressin Acetate using this advanced hybrid methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and long-term supply agreements for this high-value therapeutic peptide.
Q: Why is liquid-phase oxidation preferred over solid-phase oxidation for Desmopressin?
A: Liquid-phase oxidation avoids the intermolecular disulfide bond formation often seen in solid-phase oxidation due to high resin substitution. This results in higher yields and easier purification of the cyclic peptide.
Q: What is the advantage of purifying the linear peptide before oxidation?
A: Purifying the linear peptide via RP-HPLC prior to oxidation removes insoluble impurities and deletion sequences. This increases the effective concentration of the reactant during oxidation, significantly improving reaction efficiency and final purity.
Q: How does this method impact commercial scalability?
A: By allowing the use of resins with higher substitution degrees and simplifying the oxidation control, this method reduces raw material costs and waste generation, making it highly suitable for large-scale industrial production ranging from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desmopressin Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the rigorous demands of the global pharmaceutical market. Our technical team has extensively evaluated the hybrid solid-phase and liquid-phase oxidation route described in CN103102395A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Desmopressin Acetate that meets stringent purity specifications through our rigorous QC labs, ensuring that every batch complies with international pharmacopoeia standards. Our facility is equipped to handle the specific chromatography and oxidation requirements of this process, guaranteeing a supply of high-quality API that supports your drug development and commercialization goals.
We invite you to collaborate with us to optimize your supply chain for this essential medication. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to verify our capability to deliver cost-effective and reliable Desmopressin Acetate solutions. Let us partner to ensure the continuous availability of this life-saving hormone for patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
