Revolutionizing Allene Production: Room-Temperature Gold Carbene Catalysis for Scalable Pharmaceutical Intermediates
The chemical synthesis landscape is constantly evolving, driven by the need for more sustainable, efficient, and versatile methodologies to construct complex molecular architectures. A significant breakthrough in this domain is documented in Chinese Patent CN115872825A, which discloses a novel preparation method for 1,3-disubstituted allene compounds based on a gold carbene catalytic system operating at room temperature. This technology represents a paradigm shift from the traditional Allenation of Terminal Alkynes (ATA) reactions, which historically demanded harsh thermal conditions and often suffered from poor catalyst turnover due to water sensitivity. By leveraging a specific gold carbene catalyst, such as Au3d, in conjunction with molecular sieves and fluorinated alcohol solvents, this invention enables the one-step synthesis of valuable allene intermediates from readily available terminal alkynes, aldehydes, and amines. For R&D directors and process chemists, this development offers a powerful tool for accessing diverse chemical space with unprecedented mildness, while supply chain managers will appreciate the implications for safer, more energy-efficient manufacturing protocols.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
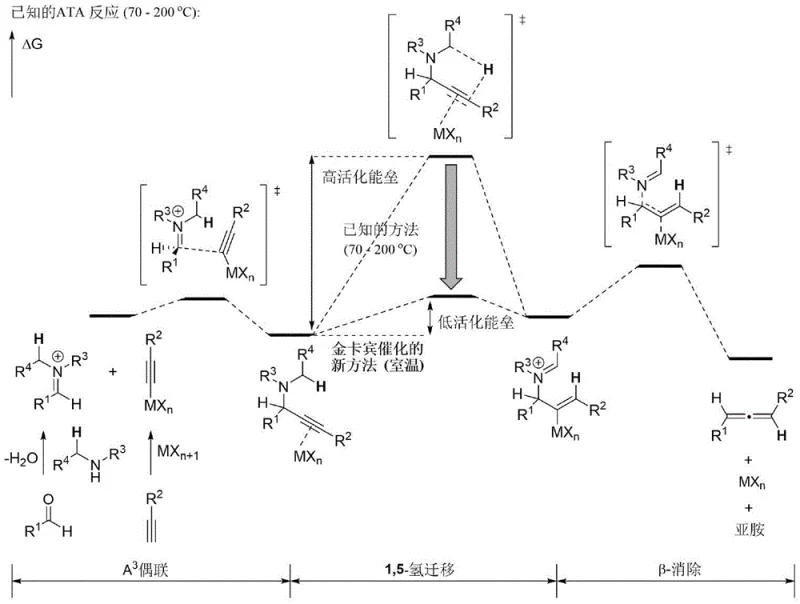
Historically, the synthesis of allenes via the ATA reaction has been plagued by significant thermodynamic and kinetic barriers that hinder its widespread adoption in sensitive pharmaceutical manufacturing. The conventional pathway typically involves an initial A³-coupling to form a propargylamine, followed by a critical 1,5-hydrogen migration and subsequent beta-elimination. The 1,5-hydrogen transfer step is notoriously difficult, possessing a very high activation energy barrier that necessitates reaction temperatures ranging from 70°C to 200°C. Such elevated temperatures not only escalate energy consumption and operational costs but also introduce severe safety hazards, particularly when dealing with thermally unstable substrates or intermediates. Furthermore, the in-situ generation of water and imine species during the reaction often poisons traditional metal catalysts, forcing the use of stoichiometric amounts of metal reagents rather than catalytic quantities. This leads to increased metal waste, complicated purification processes to remove heavy metal residues, and ultimately, a higher environmental footprint that conflicts with modern green chemistry principles.
The Novel Approach
In stark contrast, the methodology described in patent CN115872825A overcomes these entrenched limitations through the strategic application of a gold carbene catalytic system. By utilizing specialized catalysts like Au3d, the reaction successfully lowers the activation energy barrier for the rate-determining 1,5-hydrogen migration step, allowing the transformation to proceed smoothly at room temperature (25-60°C). This dramatic reduction in thermal requirement eliminates the need for energy-intensive heating and mitigates safety risks associated with high-temperature operations. Additionally, the inclusion of molecular sieves effectively scavenges the water produced during the condensation of aldehydes and amines, protecting the catalytic species from deactivation. This innovation ensures that the gold catalyst remains active throughout the cycle, enabling true catalytic turnover with loadings as low as 5 mol%. The result is a robust, operationally simple protocol that tolerates a wide range of functional groups, including halogens, esters, and heterocycles, making it an ideal candidate for the late-stage functionalization of complex drug candidates.
Mechanistic Insights into Gold Carbene-Catalyzed Allene Formation
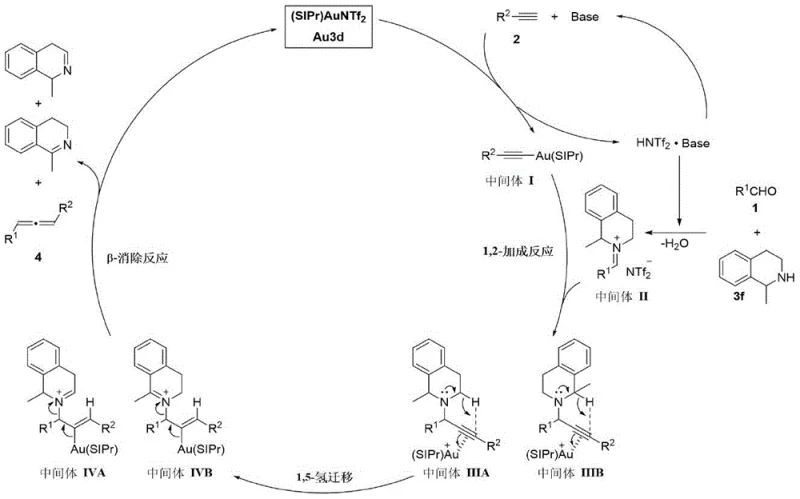
To fully appreciate the technical superiority of this room-temperature protocol, one must examine the underlying mechanistic pathway that differentiates it from thermal methods. The reaction initiates with the formation of an alkynyl gold intermediate (Intermediate I) through the interaction of the terminal alkyne with the gold carbene catalyst in the presence of a base. Simultaneously, the aldehyde and amine undergo dehydration to generate an imine intermediate (Intermediate II). The pivotal step involves the 1,2-addition of the alkynyl gold species to the imine, forming gold-carbene-coordinated propargyl amine intermediates (IIIA and IIIB). Unlike thermal processes where hydrogen migration is sluggish, the gold center facilitates a rapid 1,5-hydrogen transfer from the propargylic position to the adjacent carbon. This step, which is the bottleneck in conventional synthesis, is significantly accelerated by the electronic properties of the gold carbene complex, effectively bypassing the high-energy transition state that typically requires heat.
Following the hydrogen migration, the resulting alkenyl gold intermediates (IVA and IVB) undergo a beta-elimination reaction. This final step releases the desired 1,3-disubstituted allene product and regenerates the active gold carbene catalyst species, closing the catalytic cycle. The elegance of this mechanism lies in its ability to orchestrate multiple bond-forming and bond-breaking events under mild conditions without compromising yield or selectivity. The use of 2,2,2-trifluoroethanol (TFE) as the solvent further enhances the reaction efficiency by stabilizing charged intermediates and facilitating proton transfer processes. This detailed understanding of the catalytic cycle provides R&D teams with the confidence to adapt this chemistry for a broad spectrum of substrates, knowing that the fundamental energetics favor product formation even at ambient temperatures.
How to Synthesize 1,3-Disubstituted Allenes Efficiently
Implementing this advanced synthetic route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The process is designed to be user-friendly, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The standard procedure involves charging a dried reaction vessel with molecular sieves, the gold catalyst, and the fluorinated solvent under an inert atmosphere, followed by the sequential addition of the aldehyde, amine, and alkyne components. The mixture is then stirred at room temperature for a period ranging from 24 to 72 hours, depending on the steric and electronic nature of the substrates. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel under argon atmosphere with molecular sieves, gold carbene catalyst (e.g., Au3d), and organic solvent such as 2,2,2-trifluoroethanol.
- Add aldehyde, amine (e.g., 1-methyl-1,2,3,4-tetrahydroisoquinoline), and terminal alkyne sequentially with stirring at room temperature.
- React for 24-72 hours, then filter through silica gel, concentrate, and purify via flash column chromatography to obtain the allene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from high-temperature thermal methods to this room-temperature gold-catalyzed process offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of high-temperature heating requirements translates directly into substantial cost reduction in fine chemical manufacturing by lowering utility consumption and reducing the wear and tear on reactor vessels. Furthermore, the shift from stoichiometric metal usage to a truly catalytic system drastically reduces the amount of expensive precious metal required per batch, optimizing raw material costs and simplifying the downstream removal of metal impurities to meet stringent regulatory standards for pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the drastic simplification of the energy profile and reagent consumption. By operating at room temperature, facilities can avoid the capital expenditure associated with high-temperature reactors and the ongoing operational costs of maintaining elevated thermal conditions. Moreover, the catalytic nature of the gold complex means that significantly less metal is consumed compared to traditional stoichiometric methods, leading to direct savings on raw material procurement. The simplified workup procedure, which avoids complex quenching steps often needed for reactive metal species, further reduces labor and waste disposal costs, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, the mild reaction conditions enhance the reliability of the supply chain by minimizing the risk of batch failures due to thermal runaway or equipment malfunction. The broad substrate scope demonstrated in the patent, which includes sensitive functional groups like halides, nitriles, and esters, allows for the sourcing of a wider variety of commercially available starting materials without the need for extensive protection-deprotection strategies. This flexibility ensures that procurement teams can maintain continuity of supply even if specific raw material vendors face disruptions, as alternative substrates can often be accommodated within the robust catalytic framework without re-optimizing the entire process.
- Scalability and Environmental Compliance: The environmental and scalability advantages of this method align perfectly with modern sustainability goals and regulatory pressures. The ability to run reactions at ambient temperature inherently reduces the carbon footprint of the manufacturing process, supporting corporate ESG (Environmental, Social, and Governance) initiatives. Additionally, the patent data confirms successful scale-up to at least 50 mmol (11.0 g), demonstrating that the kinetics and heat transfer characteristics remain favorable as the reaction volume increases. This scalability ensures that the transition from lab-scale discovery to commercial production is smooth, reducing the time-to-market for new drug candidates while maintaining compliance with increasingly strict environmental regulations regarding solvent use and metal waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-catalyzed allene synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for industrial adoption.
Q: What is the primary advantage of the gold carbene catalytic system over traditional ATA reactions?
A: The primary advantage is the ability to operate at room temperature (25-60°C) instead of the harsh 70-200°C required by conventional methods. This is achieved by significantly lowering the activation energy barrier for the rate-determining 1,5-hydrogen migration step, thereby reducing energy consumption and safety risks while improving functional group compatibility.
Q: How does this method handle the water generated during the reaction?
A: Traditional metal catalysts are often poisoned by water and imine byproducts. This novel method utilizes molecular sieves (such as 5Å molecular sieves) in conjunction with a robust gold carbene catalyst (like Au3d), which maintains high catalytic activity even in the presence of in-situ generated water, allowing for true catalytic turnover rather than stoichiometric usage.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent data demonstrates successful scalability. The method has been validated on a 50 mmol scale (producing 11.0 g of product) with maintained yields, indicating that the mild reaction conditions and simple workup procedures are suitable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Disubstituted Allenes Supplier
The technological advancements detailed in patent CN115872825A underscore the immense potential of gold carbene catalysis in modern organic synthesis, particularly for the production of high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent literature into robust, commercial-grade manufacturing processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this room-temperature methodology are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,3-disubstituted allenes delivered meets the exacting standards required for drug substance manufacturing.
We invite global pharmaceutical and fine chemical companies to collaborate with us to leverage this cutting-edge synthetic route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates how switching to this catalytic system can optimize your current supply chain economics. We encourage you to reach out today to discuss your requirements,索取 specific COA data for related intermediates, and obtain comprehensive route feasibility assessments tailored to your target molecules. Let us help you accelerate your development timeline with safer, greener, and more efficient chemical solutions.
