Advanced Synthesis of 2-Iminothiazoline Derivatives for High-Value Agrochemical and Pharmaceutical Intermediates
Introduction to Novel 2-Iminothiazoline Synthesis Technology
The chemical landscape for producing high-value heterocyclic compounds has been significantly advanced by the methodologies disclosed in patent CN1097007A, which outlines a robust process for preparing 2-iminothiazoline derivatives. These specific derivatives serve as critical building blocks in the synthesis of complex herbicidal compounds and pharmaceutical agents, addressing a growing demand for efficient agrochemical intermediate supplier solutions. The core innovation lies in the direct acid treatment of specific thiourea derivatives, a strategy that bypasses the limitations of earlier synthetic routes. By leveraging this technology, manufacturers can achieve superior control over the molecular architecture of the final product, ensuring high purity and consistent quality essential for downstream applications in crop protection and medicinal chemistry.
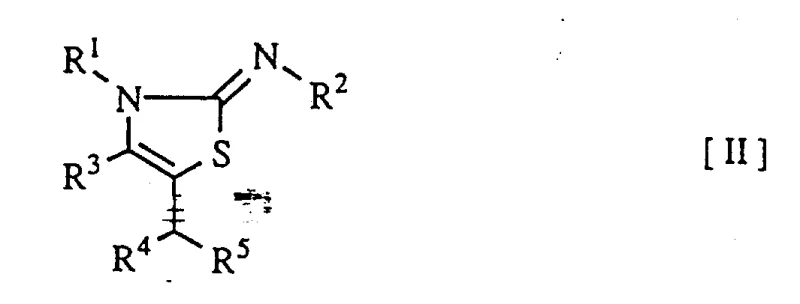
This patent specifically targets the synthesis of compounds defined by general formula (II), where the structural variability allows for extensive customization of biological activity. The ability to introduce diverse substituents at the R1, R2, R3, R4, and R5 positions provides chemists with a powerful toolkit for optimizing the physicochemical properties of the final active ingredients. Furthermore, the process is designed to be compatible with a wide range of functional groups, including halogens, alkoxyl groups, and trifluoromethyl moieties, which are frequently encountered in modern drug design. As a reliable agrochemical intermediate supplier, understanding these structural nuances is paramount for delivering materials that meet the rigorous specifications of global R&D teams seeking cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-iminothiazoline derivatives has been plagued by significant challenges regarding regioselectivity and yield, as exemplified by the methods described in literature such as J. Chem. Soc. Perkin Trans. I. The conventional pathways often involve multi-step sequences that fail to consistently direct the cyclization to the desired position on the thiazoline ring. This lack of precision results in complex mixtures of isomers that are difficult and expensive to separate, thereby inflating the overall cost of goods and reducing the effective throughput of the manufacturing line. Such inefficiencies are particularly detrimental when scaling up for commercial production, where even minor impurities can compromise the efficacy of the final herbicidal or pharmaceutical product.
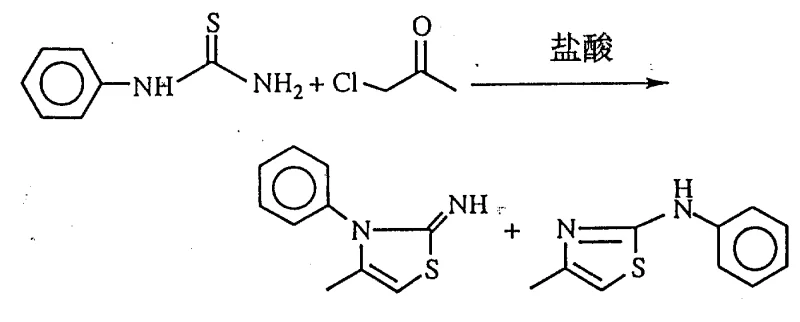
Moreover, the traditional methods often require harsh reaction conditions or specialized reagents that are not readily available on an industrial scale, creating bottlenecks in the supply chain. The inability to broadly apply these older techniques to various substituted substrates limits the chemical space that can be explored by researchers. Consequently, there has been a persistent need for a more versatile and reliable synthetic route that can accommodate the structural diversity required by modern agrochemical and pharmaceutical pipelines without sacrificing yield or purity. The shortcomings of these legacy processes underscore the importance of adopting the innovative acid-catalyzed approach detailed in the present patent data.
The Novel Approach
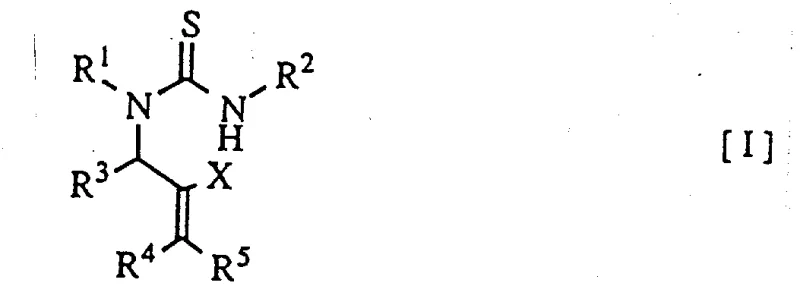
The breakthrough presented in this patent involves a streamlined acid treatment of thiourea derivatives of general formula (I), which directly yields the desired 2-iminothiazoline derivatives with exceptional efficiency. This novel approach eliminates the regioselectivity issues inherent in previous methods by utilizing specific protonic or Lewis acids to drive the cyclization mechanism precisely. The reaction conditions are remarkably flexible, allowing for the use of common mineral acids like sulfuric acid or organic sulfonic acids, as well as metal salts such as zinc chloride and tin chloride. This versatility empowers process chemists to tailor the reaction environment to the specific sensitivity of their substrate, ensuring optimal conversion rates and minimizing the formation of by-products.
By shifting the paradigm to this acid-mediated cyclization, manufacturers can achieve a drastic simplification of the workflow, moving from complex multi-step sequences to a more direct transformation. The compatibility of this method with a broad spectrum of solvents, including aliphatic hydrocarbons, aromatic solvents, and halogenated hydrocarbons, further enhances its industrial applicability. This adaptability is crucial for facilitating the commercial scale-up of complex polymer additives and fine chemicals, as it allows for the utilization of existing infrastructure without the need for exotic equipment. Ultimately, this novel approach represents a significant leap forward in process chemistry, offering a pathway to high-purity intermediates that are essential for the next generation of agricultural and therapeutic solutions.
Mechanistic Insights into Acid-Catalyzed Cyclization
The mechanistic foundation of this synthesis relies on the activation of the thiourea moiety through protonation or coordination with a Lewis acid, which facilitates the intramolecular nucleophilic attack necessary for ring closure. When a strongly-acid protonic acid is employed, the sulfur or nitrogen atoms of the thiourea derivative become protonated, increasing the electrophilicity of the adjacent carbon centers and promoting the cyclization event. Alternatively, Lewis acid metal salts function by coordinating with the electron-rich centers of the molecule, effectively lowering the energy barrier for the formation of the thiazoline ring. This dual capability ensures that the reaction can proceed efficiently across a wide range of electronic environments, accommodating both electron-deficient and electron-rich aromatic systems.
Impurity control is inherently built into this mechanism due to the high specificity of the acid-catalyzed pathway. Unlike radical-based or thermal cyclizations that may generate random fragmentation products, the acid-mediated process directs the reaction through a well-defined transition state. The use of controlled temperatures, typically ranging from 20°C to 120°C, further suppresses side reactions such as polymerization or decomposition of sensitive functional groups. Post-reaction workup involving neutralization and extraction effectively removes residual acids and metal salts, resulting in a crude product of high purity that often requires minimal further purification. This mechanistic clarity provides R&D directors with the confidence that the process is robust and reproducible, key factors for maintaining stringent quality standards in regulated industries.
How to Synthesize 2-Iminothiazoline Derivatives Efficiently
To implement this synthesis effectively, one must begin with the preparation of the requisite thiourea precursor, which can be obtained through the reaction of N-arylcyanamide derivatives with allylic halides followed by sulfuration. The detailed standardized synthesis steps for converting these precursors into the final 2-iminothiazoline products are outlined below, providing a clear roadmap for laboratory and pilot-scale execution. The process emphasizes the careful selection of acid catalysts and solvents to maximize yield while maintaining operational safety and environmental compliance. Adhering to these protocols ensures that the resulting intermediates meet the high purity specifications demanded by downstream applications in herbicide and pharmaceutical manufacturing.
- Prepare the thiourea derivative of general formula (I) by reacting N-arylcyanamide with allylic halide derivatives followed by sulfuration.
- Treat the thiourea derivative with a strongly-acid protonic acid (e.g., sulfuric acid) or a Lewis acid metal salt (e.g., zinc chloride) in a suitable solvent at 20-120°C.
- Neutralize the reaction mixture with alkali, extract with organic solvent, and purify via chromatography or crystallization to obtain the target 2-iminothiazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this acid-catalyzed synthesis route offers profound commercial advantages, particularly in terms of cost optimization and supply chain resilience. By utilizing widely available and inexpensive reagents such as sulfuric acid and zinc chloride, the raw material costs are significantly reduced compared to processes requiring precious metal catalysts or exotic organometallic reagents. This shift not only lowers the direct cost of manufacturing but also mitigates the risk associated with the volatility of specialty chemical markets. Furthermore, the simplified workup procedure reduces the consumption of solvents and energy, contributing to a more sustainable and economically viable production model that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity acids drastically lower the input costs for producing these valuable intermediates. The high yields reported in the patent examples indicate that less raw material is wasted, directly improving the overall material efficiency of the process. Additionally, the ability to use common industrial solvents like methyl isobutyl ketone and toluene avoids the premium pricing associated with specialized aprotic solvents, further enhancing the economic profile of the synthesis.
- Enhanced Supply Chain Reliability: Since the key reagents and solvents are globally sourced commodities, the risk of supply disruption is minimized, ensuring continuous production capabilities. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures. This reliability is critical for maintaining steady inventory levels and meeting the just-in-time delivery requirements of major agrochemical and pharmaceutical clients.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, making it easily scalable from kilogram to multi-ton quantities without significant engineering hurdles. The waste streams generated are primarily aqueous acidic or saline solutions, which are easier to treat and dispose of compared to heavy metal-contaminated waste. This ease of waste management simplifies regulatory compliance and reduces the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-iminothiazoline derivatives, based on the detailed data provided in the patent documentation. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their supply chains. Understanding these specifics is essential for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of the acid-catalyzed cyclization method over conventional routes?
A: The acid-catalyzed method described in CN1097007A offers superior regioselectivity compared to conventional methods, which often suffer from poor selectivity and limited application scenarios. It allows for the use of diverse substituents and achieves high purity levels suitable for commercial scale-up.
Q: Which acids are suitable for this cyclization process?
A: The process is versatile, accommodating both strongly-acid protonic acids like sulfuric acid (50-100% concentration) and trifluoromethanesulfonic acid, as well as Lewis acid metal salts such as zinc chloride, tin chloride, and aluminum chloride. This flexibility allows manufacturers to optimize based on cost and availability.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process utilizes common industrial solvents like methyl isobutyl ketone (MIBK), toluene, and dimethylbenzene, and operates at moderate temperatures (20-120°C). The use of commercially available reagents and standard workup procedures (neutralization, extraction) makes it highly amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Iminothiazoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value heterocyclic intermediates like 2-iminothiazoline derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for agrochemical and pharmaceutical applications. Our commitment to technical excellence allows us to deliver consistent quality, supporting our partners in their quest for innovation and market leadership.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments, enabling you to make data-driven decisions that optimize your production costs and enhance your competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
