Advanced Synthesis of 2,6-Diiodo BODIPY Derivatives for High-Performance Photodynamic Therapy Applications
Advanced Synthesis of 2,6-Diiodo BODIPY Derivatives for High-Performance Photodynamic Therapy Applications
The landscape of photodynamic therapy (PDT) and advanced nanomaterial functionalization is undergoing a significant transformation driven by innovations in fluorescent probe chemistry. Patent CN108892683B introduces a breakthrough methodology for the preparation of 2,6-diiodo BODIPY derivatives, specifically targeting the synthesis of Compound III. This technology addresses long-standing challenges in the field of organic synthetic chemistry, particularly regarding the modification of boron-dipyrromethene (BODIPY) cores for biomedical and nanotechnological applications. By leveraging a novel reduction strategy, this patent enables the conversion of nitro-functionalized precursors into highly reactive amino-functionalized derivatives, which serve as critical intermediates for surface modification of covalent organic frameworks (COFs) and metal-organic frameworks (MOFs). For R&D directors and procurement specialists in the pharmaceutical and electronic materials sectors, this represents a pivotal opportunity to access high-purity intermediates that offer superior photophysical properties, including enhanced intersystem crossing efficiency and prolonged triplet state lifetimes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for BODIPY derivatives often rely on the direct condensation of benzaldehyde and pyrrole, a method fraught with significant operational inefficiencies and chemical limitations. This classical approach frequently suffers from low reaction yields and necessitates complex, labor-intensive separation processes that drive up production costs and extend lead times. Furthermore, the harsh conditions often required for these condensations are incompatible with sensitive functional groups, such as amino or iodine moieties, which are essential for downstream applications in photodynamic therapy and nanomaterial coupling. The inability to tolerate these active groups severely restricts the structural diversity of BODIPY molecules that can be practically manufactured, limiting their utility in advanced drug delivery systems and imaging agents. Consequently, the industry has faced a bottleneck in scaling up the production of specialized BODIPY derivatives that possess both heavy atom enhancements and reactive handles for bioconjugation.
The Novel Approach
The innovative process disclosed in patent CN108892683B circumvents these historical barriers by employing a post-synthetic modification strategy centered on selective reduction. Instead of attempting to introduce sensitive groups during the initial ring formation, the method first constructs a stable nitro-functionalized BODIPY core and subsequently reduces the nitro group to an amino group under meticulously controlled conditions. This approach utilizes specific reducing agents—such as ammonium formate, stannous chloride, or hydrazine hydrate—that are potent enough to effect the transformation yet mild enough to preserve the integrity of the delicate BODIPY fluorophore and the iodine substituents. 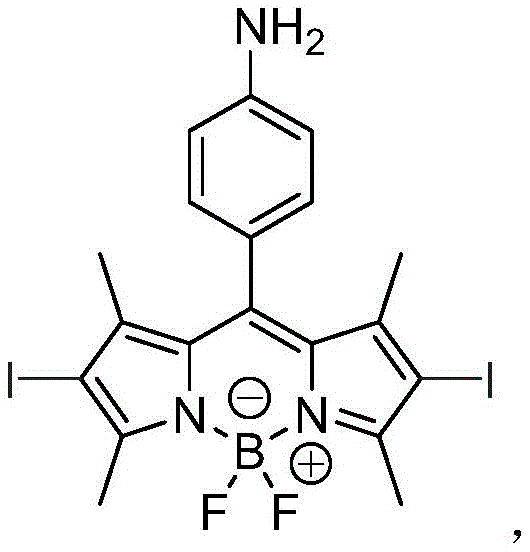 This strategic decoupling of core synthesis and functional group installation allows for yields reaching up to 42%, a substantial improvement over many traditional routes for complex fluorophores. The resulting Compound III exhibits excellent characteristics for easy expansion and modification, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to support next-generation therapeutic developments.
This strategic decoupling of core synthesis and functional group installation allows for yields reaching up to 42%, a substantial improvement over many traditional routes for complex fluorophores. The resulting Compound III exhibits excellent characteristics for easy expansion and modification, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to support next-generation therapeutic developments.
Mechanistic Insights into Selective Nitro-Reduction and Heavy Atom Enhancement
The core chemical innovation lies in the precise management of the reduction potential to convert the nitro group of Compound II into the amino group of Compound III without compromising the 2,6-diiodo substitution pattern. The presence of iodine atoms is not merely structural; it serves a critical photophysical function known as the heavy atom effect. This effect facilitates spin-orbit coupling, which dramatically enhances the rate of intersystem crossing from the singlet excited state to the triplet excited state. 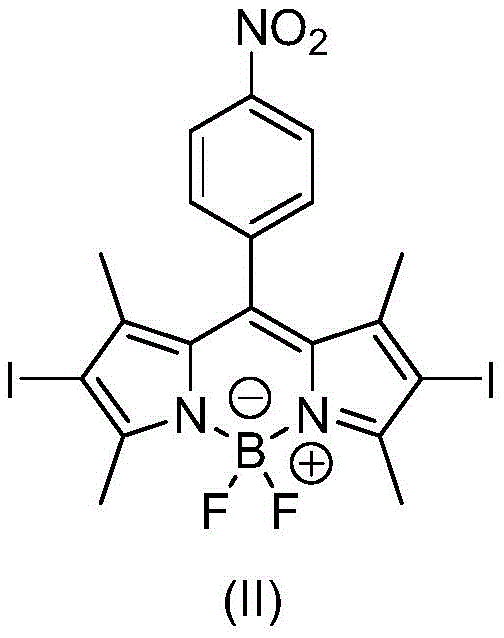 By prolonging the lifetime of the triplet state, the molecule becomes a far more efficient photosensitizer, capable of generating cytotoxic singlet oxygen species upon light irradiation, which is the fundamental mechanism of action in photodynamic therapy. The mechanistic pathway involves the interaction of the reducing agent with the nitro group, where reagents like stannous chloride in the presence of hydrochloric acid or ammonium formate with palladium on carbon provide the necessary electrons and protons. The patent highlights that the choice of solvent and temperature is paramount; for instance, using a ternary mixed solvent of dichloromethane, methanol, and isopropanol with stannous chloride ensures solubility and reaction homogeneity, preventing the precipitation of intermediates that could lead to side reactions or incomplete conversion.
By prolonging the lifetime of the triplet state, the molecule becomes a far more efficient photosensitizer, capable of generating cytotoxic singlet oxygen species upon light irradiation, which is the fundamental mechanism of action in photodynamic therapy. The mechanistic pathway involves the interaction of the reducing agent with the nitro group, where reagents like stannous chloride in the presence of hydrochloric acid or ammonium formate with palladium on carbon provide the necessary electrons and protons. The patent highlights that the choice of solvent and temperature is paramount; for instance, using a ternary mixed solvent of dichloromethane, methanol, and isopropanol with stannous chloride ensures solubility and reaction homogeneity, preventing the precipitation of intermediates that could lead to side reactions or incomplete conversion.
Furthermore, the impurity profile of the final product is tightly controlled by the specificity of these reducing agents. Unlike stronger, non-selective reducing agents such as sodium borohydride or iron powder, which the patent notes failed to produce the target compound or resulted in no reaction even after extended periods, the selected reagents (ammonium formate, stannous chloride, hydrazine hydrate) exhibit a unique chemoselectivity. They reduce the aromatic nitro group while leaving the BODIPY core and the carbon-iodine bonds intact. This selectivity is crucial for maintaining the fluorescence quantum yield and the heavy atom effect simultaneously. For quality control teams, this means a cleaner crude reaction mixture that requires less aggressive purification, thereby preserving the overall mass balance and reducing the environmental footprint associated with solvent-intensive chromatographic separations. The ability to tune reaction parameters, such as maintaining temperatures between 25°C and 100°C depending on the reductant used, provides process engineers with the flexibility to optimize for either speed or maximum yield in a commercial setting.
How to Synthesize 2,6-Diiodo BODIPY Derivative Efficiently
The synthesis of this high-value intermediate follows a logical three-stage progression designed to maximize purity and structural integrity. Initially, the BODIPY core is assembled via the condensation of 4-nitrobenzoyl chloride and 2,4-dimethylpyrrole, followed by complexation with boron trifluoride etherate to lock the structure. Subsequently, electrophilic iodination introduces the heavy atoms at the 2 and 6 positions using iodine and iodic acid. The final and most critical stage is the reduction of the nitro group to the amino group, which activates the molecule for nanomaterial coupling. The detailed standardized synthesis steps, including specific molar ratios, solvent systems, and thermal profiles for each reducing agent option, are outlined in the guide below to ensure reproducibility and safety in your pilot plant operations.
- Condense 4-nitrobenzoyl chloride with 2,4-dimethylpyrrole using an organic weak base and boron trifluoride etherate to form the core BODIPY structure (Compound I).
- Perform electrophilic substitution using iodine and iodic acid aqueous solution to introduce iodine atoms at the 2,6-positions, yielding Compound II.
- Execute selective reduction of the nitro group in Compound II using ammonium formate, stannous chloride, or hydrazine hydrate under controlled temperatures to obtain the final amino-functionalized Compound III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift towards using readily available and cost-effective reducing agents like ammonium formate and stannous chloride represents a significant departure from reliance on expensive, specialized catalysts or high-pressure hydrogenation equipment. This simplification of the reagent profile directly translates to cost reduction in pharmaceutical intermediate manufacturing by lowering raw material procurement costs and minimizing the capital expenditure required for specialized reaction vessels. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, enhances supply chain reliability by reducing the risk of batch failures due to minor fluctuations in process parameters. This resilience is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream partners in the biotech and nanomaterial sectors.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step protection and deprotection strategies typically required for amino-functionalized fluorophores streamlines the production workflow. By utilizing a direct reduction of a stable nitro-precursor, the process avoids the need for expensive protecting groups and the additional reagents required to install and remove them. This consolidation of steps significantly lowers the overall cost of goods sold (COGS) and reduces the volume of chemical waste generated per kilogram of product. Additionally, the high selectivity of the reduction minimizes the formation of difficult-to-remove byproducts, which reduces the load on purification resources and solvent consumption, further driving down operational expenses.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 2,4-dimethylpyrrole, 4-nitrobenzoyl chloride, and common reducing agents, are commodity chemicals with established global supply chains. This abundance ensures that production is not vulnerable to the bottlenecks often associated with exotic or proprietary reagents. The flexibility to choose between three different reducing systems (ammonium formate, stannous chloride, or hydrazine hydrate) provides a built-in contingency plan; if one reagent faces supply constraints, the process can be switched to an alternative pathway without requiring a complete redesign of the manufacturing protocol. This adaptability is a key factor in reducing lead time for high-purity photodynamic agents and securing long-term supply continuity.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, operating effectively at temperatures ranging from ambient to 100°C and pressures that do not require specialized high-pressure infrastructure. This facilitates the commercial scale-up of complex fluorescent probes from gram-scale laboratory batches to multi-kilogram or ton-scale commercial production with minimal engineering hurdles. Furthermore, the ability to achieve high conversion rates with defined stoichiometry reduces the environmental burden of unreacted starting materials. The use of standard organic solvents that can be readily recovered and recycled aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising on product quality or yield.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN108892683B, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this intermediate into their own drug discovery pipelines or material science projects.
Q: Why is the selective reduction of the nitro group critical in this BODIPY synthesis?
A: The nitro group in the precursor (Compound II) has low activity for coupling with nanomaterials. Converting it to an amino group (Compound III) significantly enhances reactivity, allowing for effective surface modification of covalent organic frameworks (COFs) and metal-organic frameworks (MOFs).
Q: What are the optimal reducing agents for preparing 2,6-diiodo BODIPY derivatives?
A: The patent identifies ammonium formate (with Pd/C), anhydrous stannous chloride (with HCl), and hydrazine hydrate (with Pd/C) as effective reducing agents. These agents prevent over-reduction or structural degradation, achieving yields up to 42%.
Q: How does the introduction of iodine atoms affect the photodynamic properties?
A: The heavy atom effect induced by the iodine atoms at the 2,6-positions greatly enhances intersystem crossing efficiency. This prolongs the triplet state life of the molecule, thereby significantly improving its capacity to generate singlet oxygen for photodynamic therapy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Diiodo BODIPY Derivative Supplier
The technological potential of 2,6-diiodo BODIPY derivatives in advancing photodynamic therapy and nanomaterial engineering is immense, yet realizing this potential requires a manufacturing partner with deep expertise in complex organic synthesis. NINGBO INNO PHARMCHEM stands ready to bridge the gap between patent innovation and commercial reality. As a premier CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Compound III meets the exacting standards required for biomedical applications. We understand that the integrity of your final therapeutic or diagnostic product depends on the quality of the intermediates, which is why we prioritize process optimization and impurity control at every stage of manufacturing.
We invite you to collaborate with us to optimize your supply chain for these advanced materials. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to our wealth of process knowledge and the ability to rapidly scale production as your clinical or commercial needs evolve. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
