Revolutionizing N-Methylformanilide Production: A Deep Dive into Boron-Mediated Dehydration Technology
The landscape of organic intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A pivotal advancement in this domain is documented in patent CN101475501B, which introduces a novel preparation method for N-methylformanilide (MFA), a critical precursor in the Vilsmeier-Haack reaction for synthesizing aromatic and heterocyclic aldehydes. Traditional approaches to MFA synthesis have long been plagued by thermodynamic limitations and hazardous reagent profiles, creating significant bottlenecks for large-scale pharmaceutical and agrochemical manufacturing. This patent discloses a transformative strategy that utilizes metaboric anhydride as an in-situ chemical dehydrating agent, effectively breaking the equilibrium barrier of the esterification reaction between N-methylaniline and formic acid. By integrating this boron-mediated dehydration cycle, the process achieves rapid and thorough conversion rates exceeding 99%, while simultaneously allowing for the use of lower-grade, cost-effective raw materials such as 88% aqueous formic acid. For R&D directors and procurement specialists seeking a reliable pharma intermediate supplier, understanding the mechanistic depth and operational simplicity of this technology is essential for optimizing supply chains and reducing overall production expenditures in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-methylformanilide has relied on pathways that present severe economic and environmental drawbacks, limiting their viability for modern sustainable manufacturing. One conventional route involves the methylation of formanilide using dimethyl sulfate, a reagent notorious for its high toxicity and carcinogenic properties, which necessitates stringent safety protocols and expensive waste treatment infrastructure that drives up operational costs significantly. Another common method employs the oxidation of dimethylaniline, which suffers from the use of costly oxidizing agents and generates substantial amounts of hazardous three-waste pollutants, creating a heavy burden on environmental compliance teams. Even the direct formylation of N-methylaniline with formic acid, while chemically straightforward, is fundamentally constrained by being a reversible equilibrium reaction that produces water as a byproduct. In traditional setups, driving this reaction to completion requires the continuous physical removal of water, often through energy-intensive azeotropic distillation or the use of a large excess of expensive anhydrous formic acid, both of which negatively impact the atom economy and final product cost structure.
The Novel Approach
The methodology outlined in patent CN101475501B represents a paradigm shift by replacing physical water removal with a sophisticated chemical dehydration strategy using metaboric anhydride. Instead of struggling against the equilibrium with heat and vacuum, this novel approach introduces boric acid into the reaction system, which is first dehydrated via reflux in a water-immiscible solvent to generate metaboric anhydride in situ. This anhydride species acts as a potent water scavenger, chemically binding the water molecules released during the formylation of N-methylaniline, thereby irreversibly shifting the reaction equilibrium towards the product side. This innovation eliminates the need for continuous water-carrying distillation during the synthesis step itself and drastically reduces the required molar excess of formic acid. Furthermore, the process is robust enough to tolerate the presence of water in the feedstock, enabling the use of commercially available 88% aqueous formic acid rather than premium anhydrous grades, which translates directly into substantial cost reduction in pharmaceutical intermediates manufacturing without compromising on yield or purity standards.
Mechanistic Insights into Boron-Mediated Dehydration Cycle
To fully appreciate the technical elegance of this synthesis, one must examine the catalytic cycle involving the boron species, which serves as the engine driving the high conversion efficiency observed in this patent. The process initiates with the thermal dehydration of boric acid (H3BO3) in a solvent such as toluene or xylene at temperatures ranging from 70 to 130°C, resulting in the formation of a cyclic metaboric anhydride structure. This anhydride is not merely a passive spectator but an active chemical participant that reacts stoichiometrically with the water generated during the subsequent formylation step. As N-methylaniline reacts with formic acid to produce N-methylformanilide and water, the metaboric anhydride immediately captures the water, reverting back to boric acid. This regeneration of boric acid is crucial, as it allows the solid byproduct to be filtered off post-reaction and potentially recycled, creating a semi-closed loop for the dehydrating agent. The ability of the boron species to toggle between its acidic and anhydride forms under the specific thermal conditions provided (60-90°C for reaction, higher for dehydration) ensures that the reaction medium remains sufficiently anhydrous to favor product formation, effectively solving the thermodynamic bottleneck that plagues conventional esterification processes.
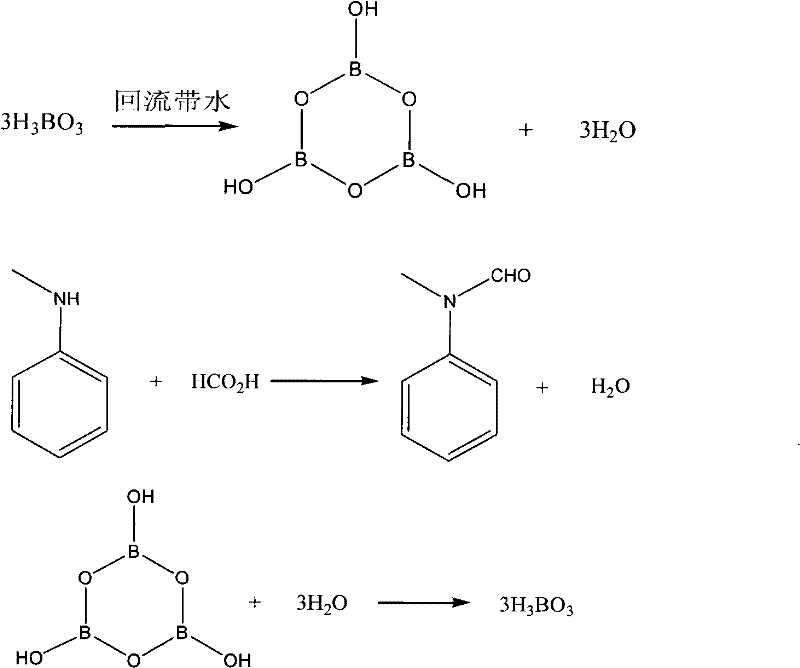
Beyond the primary dehydration mechanism, the impurity profile control in this system is inherently superior due to the mild reaction conditions and the specific selectivity of the boron-mediated pathway. Unlike oxidative methods that can lead to over-oxidation byproducts or methylating agents that introduce sulfur-containing impurities difficult to remove, this formylation route is clean and atom-economical. The use of metaboric anhydride prevents the hydrolysis of the formed amide bond, a common side reaction in aqueous or wet environments, by keeping the local water concentration near zero through chemical sequestration. This results in a crude product with exceptionally high purity, often exceeding 99% as confirmed by gas chromatography analysis in the patent examples, which significantly reduces the burden on downstream purification units such as distillation columns or crystallization tanks. For quality assurance teams, this means a more consistent impurity spectrum and a lower risk of batch failure, ensuring that the high-purity pharma intermediate meets the rigorous specifications required for downstream API synthesis, particularly for sensitive Vilsmeier reactions where trace water or impurities can derail the entire synthetic sequence.
How to Synthesize N-Methylformanilide Efficiently
Implementing this synthesis route in a pilot or production plant requires precise control over the thermal profile and addition rates to maximize the efficiency of the boron cycle. The process begins with the activation of the dehydrating agent, followed by the controlled introduction of reactants to manage exotherms, and concludes with a straightforward workup that facilitates reagent recovery. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and agitation speeds are critical for reproducibility and safety.
- Dehydrate boric acid in a water-immiscible solvent (e.g., toluene) via reflux to generate metaboric anhydride in situ.
- Cool the mixture to 60-90°C and add N-methylaniline and formic acid (anhydrous or 88% aqueous) dropwise.
- Maintain reaction temperature at 90-100°C until conversion is complete, then filter to recover boric acid for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of the technology described in CN101475501B offers compelling advantages that extend far beyond simple chemical yield improvements, addressing core pain points related to raw material volatility and waste management. The ability to utilize 88% aqueous formic acid instead of anhydrous grades is a significant logistical win, as aqueous formic acid is a bulk commodity chemical with a more stable supply chain and lower price point compared to its anhydrous counterpart, which often requires specialized storage and handling. Moreover, the elimination of toxic reagents like dimethyl sulfate removes a major regulatory hurdle and reduces the insurance and safety equipment costs associated with handling highly hazardous substances, thereby improving the overall EHS (Environment, Health, and Safety) profile of the manufacturing site. The recyclability of the boric acid further enhances the economic model, as the solid byproduct can be recovered via simple filtration and reused, minimizing the consumption of auxiliary chemicals and reducing the volume of solid waste requiring disposal. These factors combine to create a manufacturing process that is not only chemically robust but also commercially resilient against market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive anhydrous formic acid with cheaper aqueous alternatives and the reduction of solvent usage due to the elimination of continuous azeotropic distillation. By chemically scavenging water rather than physically distilling it, the process saves significant energy costs associated with heating and cooling large volumes of solvent over extended periods. Additionally, the high conversion rate (>99%) minimizes the loss of the valuable N-methylaniline starting material, ensuring that nearly every mole of input is converted to saleable product, which directly improves the gross margin per kilogram of output. The avoidance of sulfur-based reagents also eliminates the need for expensive desulfurization steps or specialized scrubbing systems, further lowering the capital and operational expenditure required for facility compliance.
- Enhanced Supply Chain Reliability: Relying on bulk industrial commodities like boric acid and aqueous formic acid insulates the supply chain from the shortages often seen with specialized or highly regulated reagents. Since these materials are produced on a massive scale for various industries, their availability is generally high, reducing the risk of production stoppages due to raw material stockouts. The simplified process flow, which avoids complex multi-step purifications or hazardous intermediate isolations, also shortens the overall manufacturing cycle time, allowing for faster turnaround on customer orders. This agility is crucial for maintaining a reliable pharma intermediate supplier status, as it enables the manufacturer to respond quickly to demand spikes from downstream API producers without the lead times associated with more cumbersome synthetic routes.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the boron-mediated route make it ideally suited for commercial scale-up of complex pharmaceutical intermediates, as it avoids the thermal runaway risks associated with strong oxidizers or highly exothermic methylations. The process generates minimal hazardous waste, with the primary byproduct being recoverable boric acid, aligning perfectly with modern green chemistry principles and increasingly strict environmental regulations. This reduced environmental footprint simplifies the permitting process for new production lines and lowers the long-term liability associated with waste disposal. Furthermore, the robustness of the reaction conditions (90-100°C) is easily manageable in standard glass-lined or stainless steel reactors, meaning that existing infrastructure can often be adapted for this process without requiring significant capital investment in specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented method is vital for stakeholders evaluating its integration into their supply networks. The following questions address common inquiries regarding the practical application, safety, and scalability of this boron-catalyzed formylation technology.
Q: Why is metaboric anhydride superior to traditional azeotropic distillation for this reaction?
A: Metaboric anhydride acts as a chemical dehydrating agent that reacts directly with the water byproduct, shifting the equilibrium completely without requiring continuous physical removal of water via complex distillation setups, thus simplifying the process.
Q: Can aqueous formic acid be used instead of expensive anhydrous formic acid?
A: Yes, the patent explicitly demonstrates that 88% aqueous formic acid can be used effectively because the metaboric anhydride scavenges the additional water introduced, significantly lowering raw material costs.
Q: Is the boric acid catalyst recoverable for industrial scale-up?
A: Absolutely. The process allows for the filtration and recovery of solid boric acid after the reaction, which can be recycled back into the dehydration step, minimizing waste and enhancing sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methylformanilide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to industrial reality requires deep technical expertise and robust process engineering capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the boron-mediated dehydration process are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced GC-MS and NMR instrumentation to guarantee that every batch of N-methylformanilide meets the exacting standards required for Vilsmeier-Haack applications. Our commitment to quality ensures that the impurity profiles are tightly controlled, providing our partners with a consistent and reliable feedstock for their critical synthetic steps.
We invite global procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging this advanced synthesis technology, we can help you optimize your bill of materials and reduce your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative approach to fine chemical manufacturing can become a cornerstone of your supply chain strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
