Revolutionizing Alkyl Borate Production: Titanium-Catalyzed Direct Borylation for Commercial Scale
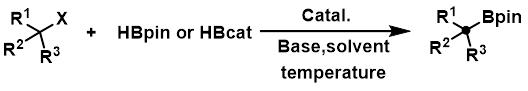
The landscape of organoboron chemistry is undergoing a significant transformation driven by the need for more sustainable and cost-effective synthetic routes. Patent CN112645971B introduces a groundbreaking method for the direct preparation of alkyl borate compounds from alkyl halides, utilizing a titanium metal catalyst system. This innovation addresses critical bottlenecks in the synthesis of pharmaceutical intermediates and fine chemicals, offering a robust alternative to traditional transmetallation or hydroboration protocols. By leveraging earth-abundant titanium instead of precious metals, this technology not only reduces raw material costs but also simplifies the purification process, thereby enhancing the overall efficiency of the supply chain for high-value chemical building blocks.
Alkyl boronic acid pinacol esters are indispensable intermediates in modern organic synthesis, particularly for Suzuki-Miyaura cross-coupling reactions which are foundational in drug discovery. However, conventional methods often rely on expensive catalysts or harsh conditions that limit substrate scope. The disclosed technology operates under mild conditions, typically between 35°C and 100°C, and demonstrates remarkable compatibility with a wide array of functional groups. For procurement managers and supply chain directors, this represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering complex molecules with greater consistency and lower environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkyl borate compounds has relied heavily on the use of reactive metal reagents such as alkyl lithium or alkyl magnesium species through transmetallation reactions. While effective for simple substrates, these methods suffer from narrow substrate applicability and poor functional group tolerance, often necessitating cumbersome protection and deprotection steps. Additionally, olefin hydroboration, another common pathway, is plagued by regioselectivity challenges due to multiple reaction sites on the olefin and the potential for isomerization. These limitations result in lower yields, increased waste generation, and higher production costs, creating significant friction in the manufacturing of high-purity OLED material or agrochemical intermediate precursors.
The Novel Approach
In stark contrast, the titanium-catalyzed direct borylation method described in the patent offers a streamlined solution that bypasses these historical constraints. By employing a titanium metal catalyst in conjunction with a base and a borate compound, alkyl halides or sulfonates are directly converted into alkyl boronic acid pinacol esters with high efficiency. This approach eliminates the need for pre-formed organometallic reagents and avoids the regioselectivity pitfalls of hydroboration. As illustrated in the reaction scheme below, the process is operationally simple and scalable, providing a clear pathway for cost reduction in electronic chemical manufacturing and other high-demand sectors.
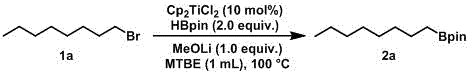
The mechanistic underpinnings of this titanium-catalyzed transformation reveal why it is superior for complex molecule synthesis. The catalytic cycle likely involves the activation of the alkyl halide by the low-valent titanium species, generating a radical intermediate that subsequently reacts with the boron source. This radical pathway allows for the activation of unactivated alkyl halides, which are typically inert under noble metal catalysis. Furthermore, the specific choice of ligands and bases, such as bis(cyclopentadienyl)titanium dichloride and lithium methoxide, fine-tunes the reactivity to prevent side reactions like beta-hydride elimination, ensuring high selectivity for the desired boronated product.
Impurity control is another critical aspect where this methodology excels. Traditional methods often leave behind trace metals that are difficult to remove and can poison downstream catalytic processes. The titanium system described here facilitates easier removal of metal residues, leading to a cleaner impurity profile. This is paramount for R&D directors focusing on the purity and杂质谱 (impurity profile) of API intermediates. The ability to tolerate diverse functional groups, including esters, ethers, and heterocycles, means that complex scaffolds can be constructed without compromising the integrity of sensitive moieties, thereby accelerating the timeline from benchtop discovery to commercial production.
How to Synthesize Alkyl Boronic Acid Pinacol Ester Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize yield and purity. The process begins with the preparation of the reaction mixture under an inert atmosphere, followed by controlled heating and stirring. The versatility of the method allows for the use of various solvents and bases, enabling customization based on specific substrate requirements. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Preparation of Reaction Mixture: Mix titanium metal catalyst, alkali compound, borate compound, and alkyl halide in an organic solvent under a protective nitrogen or argon atmosphere.
- Catalytic Borylation Reaction: Heat the mixture to a temperature between 35°C and 100°C and stir for 8 to 24 hours to facilitate the direct conversion of the halide to the boronic ester.
- Workup and Purification: Upon completion, purify the resulting alkyl boronic acid pinacol ester system using column chromatography or standard extraction techniques to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this titanium-catalyzed technology offers profound benefits for procurement and supply chain management. The shift from precious metal catalysts to abundant titanium significantly lowers the raw material cost base, which is a critical factor in maintaining competitive pricing for bulk chemical orders. Moreover, the use of readily available alkyl halides as starting materials ensures a stable supply chain, reducing the risk of disruptions associated with specialized reagents. This stability is essential for maintaining continuous production schedules and meeting the rigorous delivery timelines expected by global pharmaceutical and agrochemical companies.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or rhodium catalysts with cost-effective titanium complexes results in substantial cost savings per kilogram of product. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing process. By eliminating the need for cryogenic conditions often required for organolithium reagents, energy consumption is also drastically reduced, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: Utilizing common alkyl halides and sulfonates as feedstocks mitigates supply chain risks associated with scarce or geopolitically sensitive materials. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or pressure, ensuring consistent output quality. This reliability allows supply chain heads to plan inventory more effectively and reduce lead time for high-purity alkyl borates, fostering stronger partnerships with downstream customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild reaction temperatures and atmospheric pressure conditions make this process inherently safer and easier to scale from laboratory to industrial reactors. The reduced toxicity of the catalyst system aligns with increasingly stringent environmental regulations, minimizing the burden of hazardous waste treatment. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key differentiator in winning contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented borylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on its practical application and advantages over existing methods.
Q: What are the primary advantages of using titanium catalysts over traditional transition metals for borylation?
A: Titanium catalysts offer significant cost reductions compared to precious metals like palladium or rhodium. Furthermore, this method utilizes abundant alkyl halides as starting materials and demonstrates superior functional group tolerance, avoiding the regioselectivity issues common in olefin hydroboration.
Q: Does this synthesis method support complex substrates with sensitive functional groups?
A: Yes, the patented process exhibits excellent functional group tolerance. It successfully converts various alkyl halides and sulfonates, including those containing esters, ethers, and heterocycles, without requiring extensive protecting group strategies.
Q: How does this method impact the scalability of organoboron compound production?
A: The reaction operates under mild temperatures (35°C to 100°C) and standard atmospheric pressure, making it highly amenable to commercial scale-up. The use of readily available reagents and simple workup procedures further enhances its viability for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Boronic Acid Pinacol Ester Supplier
The technological potential of this titanium-catalyzed borylation method is immense, offering a pathway to more efficient and sustainable chemical manufacturing. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of alkyl borate meets the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this advanced synthesis method can enhance your product portfolio and operational efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
