Scalable Production of High-Purity Pyraclostrobin via Advanced Gas-Phase Methylation
Scalable Production of High-Purity Pyraclostrobin via Advanced Gas-Phase Methylation
The global demand for high-efficacy strobilurin fungicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for market-leading compounds like pyraclostrobin. Patent CN111655668A introduces a transformative approach to synthesizing this critical active ingredient, addressing long-standing challenges in yield, purity, and environmental compliance. This technical insight report analyzes the novel gas-phase methylation strategy that converts [2-({[1-(4-chlorophenyl)-1H-pyrazol-3-yl]oxy}methyl)phenyl]hydroxycarbamate directly into the final active structure. By shifting away from traditional liquid alkylating agents, this methodology offers a robust pathway for reliable agrochemical intermediate supplier networks seeking to optimize their production capabilities.
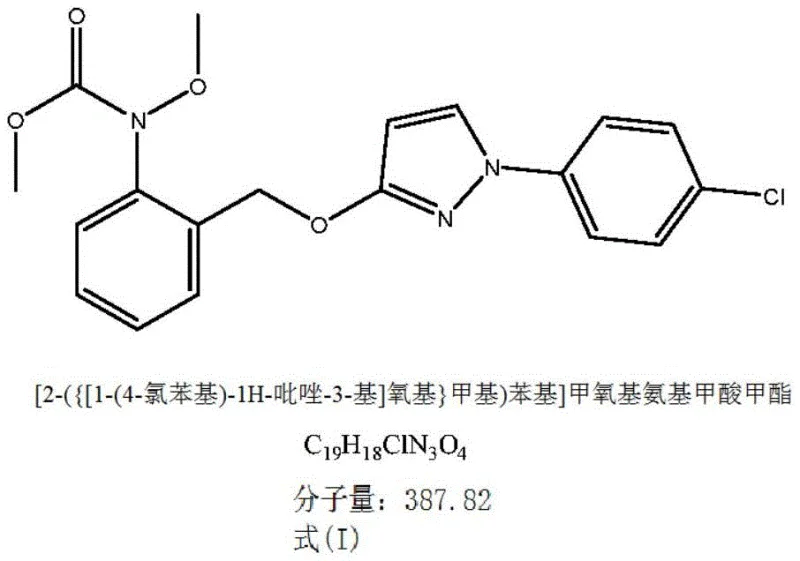
The core innovation lies in the precise manipulation of the methylation step, which is often the bottleneck in pyrazole-based fungicide synthesis. Conventional routes frequently struggle with the handling of toxic reagents and the generation of difficult-to-remove impurities. The disclosed technology leverages gaseous methylating agents—specifically methyl chloride or methyl bromide—under controlled autogenous pressure. This shift not only streamlines the reaction kinetics but also fundamentally alters the impurity profile, resulting in a product that meets stringent quality specifications required by major crop protection companies. The ability to achieve conversions exceeding 99% with minimal downstream processing represents a significant leap forward in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pyraclostrobin has relied heavily on liquid methylating agents such as dimethyl sulfate or methyl iodide, each presenting distinct operational and economic drawbacks. The use of dimethyl sulfate, while effective, generates substantial quantities of acidic sulfate wastewater, imposing a heavy burden on effluent treatment plants and increasing the overall environmental footprint of the manufacturing facility. Furthermore, controlling the reaction exotherm and preventing over-alkylation or side reactions with liquid reagents requires meticulous temperature management and often results in lower selectivity. Alternative routes utilizing methyl iodide suffer from prohibitive raw material costs, rendering them economically unviable for large-scale commercial production. Additionally, previous methods involving the reduction of nitro groups to hydroxylamines often faced difficulties in stopping the reaction at the desired stage, leading to complex impurity profiles that are arduous to purify.
The Novel Approach
In stark contrast, the improved method detailed in the patent utilizes gaseous methyl chloride or methyl bromide in a closed-loop system, effectively circumventing the waste generation issues associated with sulfate esters. This gas-phase approach allows for superior stoichiometric control; the reaction pressure serves as a real-time indicator of reagent consumption, enabling automatic feed cutoff when the system reaches equilibrium. The implementation of a phase transfer catalyst, such as tetrabutylammonium bromide or polyethylene glycols, further accelerates the reaction rate in heterogeneous mixtures, ensuring complete conversion of the hydroxycarbamate precursor. This methodology not only simplifies the work-up procedure by eliminating aqueous washes required to remove sulfate salts but also facilitates the recovery and recycling of unreacted alkylating gas, contributing to substantial cost reduction in fungicide manufacturing.
Mechanistic Insights into Gas-Phase Nucleophilic Substitution
The chemical transformation centers on an SN2-type nucleophilic substitution where the nitrogen atom of the hydroxycarbamate moiety acts as the nucleophile. In the presence of a mild inorganic base like potassium carbonate, the hydroxyl proton is abstracted to form a reactive anionic species. The introduction of methyl chloride gas into the headspace of the reactor creates a dynamic equilibrium where the gas dissolves into the solvent phase to encounter the nucleophile. The addition of a catalytic amount of potassium iodide plays a crucial role by generating methyl iodide in situ, which is a more potent alkylating agent due to the superior leaving group ability of the iodide ion. This catalytic cycle significantly lowers the activation energy of the methylation step, allowing the reaction to proceed efficiently at moderate temperatures between 30°C and 50°C.
Impurity control is inherently built into this mechanistic pathway. Unlike liquid reagents that can cause localized high concentrations leading to side reactions, the gradual dissolution of the gaseous reagent ensures a uniform concentration throughout the reaction medium. This homogeneity minimizes the formation of di-alkylated byproducts or degradation of the sensitive pyrazole ring. Furthermore, the use of non-protic solvents such as acetone, methyl isobutyl ketone, or dichloroethane provides an optimal dielectric environment that stabilizes the transition state without participating in unwanted side reactions. The result is a clean reaction profile where the primary impurity is simply unreacted starting material, which is easily removed during the subsequent crystallization step, ensuring the delivery of high-purity agrochemical intermediates.
How to Synthesize Pyraclostrobin Efficiently
Executing this synthesis requires careful attention to pressure management and gas handling protocols to ensure both safety and efficiency. The process begins with the preparation of a slurry containing the hydroxycarbamate precursor, a suitable organic solvent, and the base-catalyst system. Once the system is sealed and evacuated to remove oxygen and moisture, the alkylating gas is introduced. The reaction is self-regulating to an extent; as the gas is consumed, the pressure drops, triggering the feed valve to open, and as the reaction nears completion, the pressure rises, shutting off the feed. This elegant control mechanism removes the need for complex gravimetric dosing systems. Following the reaction, the mixture is heated to ensure complete conversion before cooling to precipitate inorganic salts.
- Charge the reactor with the hydroxycarbamate precursor, solvent (e.g., acetone or MIBK), potassium carbonate base, and potassium iodide catalyst.
- Evacuate the system and introduce methyl chloride or methyl bromide gas while maintaining temperature between 30-50°C and pressure below 6.0 Kg/cm².
- Upon completion, purge excess gas into a dissolution vessel, filter salts, and crystallize the crude product from alcohol/hexane mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gas-phase methylation technology translates into tangible strategic benefits beyond simple yield improvements. The elimination of dimethyl sulfate removes a highly regulated and hazardous material from the supply chain, reducing regulatory compliance costs and insurance premiums associated with storage and transport. The simplified work-up procedure, which avoids extensive aqueous washing and salt disposal, leads to a drastic reduction in utility consumption and waste treatment fees. Moreover, the ability to recycle unreacted methyl chloride or bromide gas creates a closed-loop material flow, insulating the production process from volatility in raw material pricing and ensuring consistent availability of key reagents.
- Cost Reduction in Manufacturing: The replacement of expensive methyl iodide with commodity-grade methyl chloride or bromide gas results in direct raw material savings. Additionally, the removal of the wastewater treatment burden associated with sulfate byproducts significantly lowers the operational expenditure per kilogram of produced API. The high conversion rates minimize the loss of valuable intermediate, maximizing the throughput of existing reactor assets without the need for capital-intensive expansion.
- Enhanced Supply Chain Reliability: By utilizing gaseous reagents that can be sourced from broad industrial chemical suppliers, the dependency on niche specialty chemical vendors is reduced. The robust nature of the reaction, which tolerates slight variations in temperature and pressure without compromising quality, ensures consistent batch-to-batch performance. This reliability is critical for maintaining uninterrupted supply to downstream formulators and meeting the rigorous delivery schedules of global agrochemical contracts.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales with consistent results. The closed-vessel design aligns perfectly with modern environmental, health, and safety (EHS) standards by containing volatile organic compounds and preventing fugitive emissions. This compliance facilitates easier permitting for capacity expansions and strengthens the sustainability profile of the manufacturing site, a key metric for ESG-focused partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved pyraclostrobin synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfers and procurement teams negotiating supply agreements.
Q: What are the advantages of using methyl chloride over dimethyl sulfate in this synthesis?
A: Using methyl chloride gas eliminates the generation of acidic sulfate wastewater associated with dimethyl sulfate, significantly reducing environmental treatment costs and simplifying the purification process.
Q: How is the reaction pressure controlled during the methylation step?
A: The process utilizes a closed-vessel system where gas feed is automatically interrupted when pressure exceeds approximately 6.0 Kg/cm², ensuring safety and stoichiometric efficiency without complex weighing systems.
Q: What purity levels can be achieved with this improved method?
A: The patented method consistently yields pyraclostrobin with purity levels ranging from 94% to 99%, with specific examples demonstrating purities exceeding 98% after crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyraclostrobin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this gas-phase methylation process are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of pyraclostrobin intermediate meets the exacting standards of the global agrochemical industry.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this greener, more efficient process. We are prepared to provide specific COA data and route feasibility assessments tailored to your production requirements, helping you secure a competitive advantage in the fungicide market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
