Scalable Manufacturing of GT-1061 Hydrochloride: A Novel Nitration Strategy for Alzheimer’s Research
Scalable Manufacturing of GT-1061 Hydrochloride: A Novel Nitration Strategy for Alzheimer’s Research
The development of effective therapeutics for neurodegenerative disorders remains a paramount challenge in modern medicinal chemistry, driving intense demand for high-quality intermediates such as GT-1061. Patent CN108239040B introduces a groundbreaking preparation method for 2-(4-methylthiazol-5-yl) ethyl nitrate hydrochloride, a critical compound investigated for its potential in treating Alzheimer's disease. This technical disclosure addresses long-standing inefficiencies in synthesizing thiazole-based nitrate esters, offering a pathway that combines operational simplicity with exceptional safety profiles. By shifting away from hazardous concentration steps and complex purification protocols, this innovation provides a robust foundation for reliable pharmaceutical intermediate supplier networks aiming to support clinical and commercial demands. The following analysis details how this proprietary chemistry transforms the manufacturing landscape for this specific molecular scaffold.
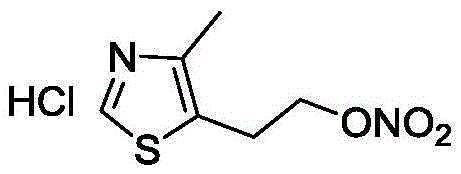
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrate esters on heterocyclic scaffolds has been plagued by severe safety hazards and poor atom economy. Prior art, including documents WO2000054756 and US6310052, typically relied on harsh nitration conditions utilizing mixtures of concentrated nitric and sulfuric acids or urea-based systems. As illustrated in the comparative reaction schemes, these traditional routes often resulted in incomplete conversion of the starting material, 4-methyl-5-hydroxyethyl thiazole, necessitating tedious column chromatography for purification. Validation experiments cited in the background technology revealed that even optimized literature procedures yielded merely 45% to 56% of the desired product, with some analogues producing yields as low as 23%. Furthermore, the requirement to concentrate the free base nitrate ester prior to salt formation introduced significant explosion risks, rendering these methods unsuitable for cost reduction in API manufacturing on an industrial scale.
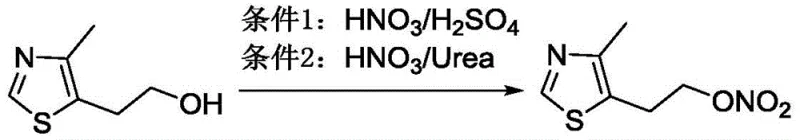
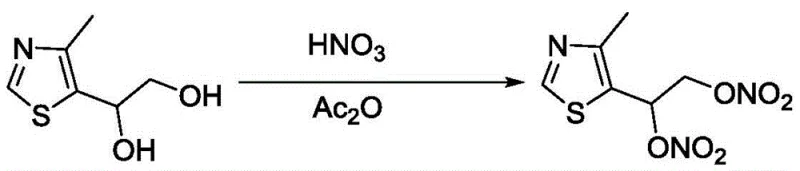
The Novel Approach
In stark contrast, the methodology disclosed in CN108239040B employs a streamlined two-stage sequence that bypasses the pitfalls of legacy chemistry. The core innovation lies in the use of fuming nitric acid activated by acetic anhydride under strictly controlled low-temperature conditions, typically between -10°C and 5°C. This specific reagent combination facilitates rapid and complete nitration without the need for strong mineral acids like sulfuric acid, thereby minimizing side reactions and degradation. Crucially, the process eliminates the dangerous concentration step; the crude nitrate ester is reacted directly in the solvent system to form the hydrochloride salt. This telescoped approach not only enhances safety by avoiding the isolation of unstable energetic intermediates but also drastically simplifies the workflow, allowing for direct crystallization of the final product with high purity. The result is a commercially viable route that supports the commercial scale-up of complex pharmaceutical intermediates with unprecedented efficiency.
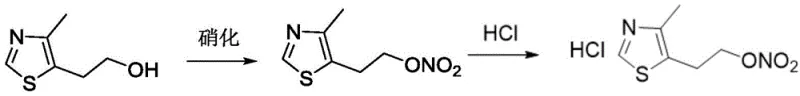
Mechanistic Insights into Acetic Anhydride-Activated Nitration
The mechanistic superiority of this process stems from the in situ generation of acetyl nitrate, a potent nitrating agent that reacts selectively with the primary hydroxyl group of the thiazole side chain. By maintaining the reaction temperature below 5°C, the kinetic energy of the system is carefully managed to prevent the decomposition of the newly formed nitrate ester bond, which is thermally sensitive. The use of dichloromethane or ethyl acetate as solvents provides an ideal medium for heat dissipation and solubility control, ensuring that the exothermic nitration event does not lead to thermal runaway. This precise thermal management is critical for maintaining the integrity of the thiazole ring, which can be susceptible to acid-catalyzed ring opening under more aggressive conditions found in sulfuric acid-mediated nitrations. Consequently, the impurity profile is significantly cleaner, reducing the burden on downstream purification units.
Furthermore, the direct salt formation step leverages the nucleophilicity of the chloride ion in the presence of an acid source, such as acetyl chloride in ethanol or hydrochloric acid in ethyl acetate. This step proceeds efficiently at 0-5°C, causing the hydrochloride salt to precipitate directly from the solution due to its lower solubility compared to the free base. This precipitation acts as a driving force for the reaction equilibrium and serves as an initial purification step, excluding soluble organic impurities that remain in the mother liquor. The subsequent recrystallization from solvents like absolute ethanol further refines the crystal lattice, removing trace colored impurities and residual solvents. This multi-layered purification strategy ensures that the final active pharmaceutical ingredient meets stringent purity specifications required for clinical trials without the need for preparative HPLC or silica gel chromatography.
How to Synthesize 2-(4-Methylthiazol-5-yl) Ethyl Nitrate Hydrochloride Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for laboratory and pilot plant operations. The process begins with the dissolution of 4-methyl-5-(2-hydroxyethyl)thiazole in dichloromethane, followed by the controlled addition of acetic anhydride and fuming nitric acid while maintaining an internal temperature below 5°C. After the nitration is complete, the mixture is quenched with water and neutralized to isolate the nitrate ester. Instead of isolating this intermediate, it is immediately subjected to salt formation by adding a hydrochloric acid source, leading to the precipitation of the target hydrochloride salt. Detailed standard operating procedures regarding stoichiometry, addition rates, and specific agitation speeds are critical for maximizing yield and safety. For the complete step-by-step technical guide and specific parameter settings used in successful validation batches, please refer to the standardized synthesis protocol below.
- Nitration: React 4-methyl-5-(2-hydroxyethyl)thiazole with fuming nitric acid and acetic anhydride at -10°C to 5°C to form the nitrate ester.
- Salt Formation: React the crude nitrate ester directly with a hydrochloric acid source (e.g., acetyl chloride/ethanol) at 0-5°C to precipitate the hydrochloride salt.
- Purification: Recrystallize the crude hydrochloride salt in solvents like absolute ethanol or ethyl acetate at 0-30°C to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel manufacturing route represents a strategic opportunity to optimize sourcing strategies for Alzheimer's research materials. The elimination of column chromatography, a batch-process bottleneck that limits throughput and increases solvent consumption, translates directly into substantial cost savings in manufacturing operations. By replacing complex purification with simple crystallization, the process reduces the consumption of silica gel and large volumes of elution solvents, thereby lowering both material costs and waste disposal fees. Additionally, the avoidance of concentrated sulfuric acid removes the need for specialized corrosion-resistant equipment and extensive neutralization protocols, further streamlining the capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The high yield of the nitration step, consistently exceeding 90% in optimized examples, ensures maximum utilization of the expensive thiazole starting material. Unlike prior art methods that suffered from yields below 50%, this efficiency minimizes raw material waste. Furthermore, the telescoped nature of the reaction—where the intermediate is not isolated but converted directly to the salt—reduces unit operations, labor hours, and energy consumption associated with drying and re-dissolving intermediates. These cumulative efficiencies drive down the cost of goods sold (COGS), making the final intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic anhydride, fuming nitric acid, and common organic solvents ensures a stable supply of reagents, mitigating the risk of shortages associated with specialized catalysts. The robustness of the reaction conditions, which tolerate slight variations in temperature and time without significant yield loss, enhances batch-to-batch consistency. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for pharmaceutical clients who require just-in-time inventory management for their drug development pipelines.
- Scalability and Environmental Compliance: Safety is a paramount concern in the production of nitrate esters, and this process addresses it by avoiding the concentration of energetic intermediates. This inherent safety feature facilitates easier regulatory approval for plant operations and reduces insurance premiums. Moreover, the simplified workup generates less hazardous waste compared to traditional methods involving heavy metal catalysts or large volumes of acidic aqueous waste. The ability to scale this process from gram quantities to multi-kilogram batches, as demonstrated in Example 10 with a 57.2g input, confirms its readiness for commercial scale-up of complex pharmaceutical intermediates without requiring fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of GT-1061 hydrochloride. These answers are derived directly from the experimental data and technical disclosures within patent CN108239040B, providing transparency for potential partners evaluating this technology. Understanding these nuances is essential for assessing the feasibility of integrating this intermediate into broader drug substance manufacturing workflows.
Q: Why is the new nitration method safer than conventional concentrated sulfuric acid methods?
A: The novel process avoids the use of concentrated sulfuric acid and eliminates the need for concentrating the nitrate ester intermediate, significantly reducing the risk of explosion associated with unstable nitro compounds.
Q: What is the expected yield improvement compared to literature methods?
A: While literature methods reported yields as low as 23% to 56% requiring complex chromatography, this patented route achieves nitration yields exceeding 90% and overall isolated yields over 66% without column purification.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes mild temperatures (-10°C to 10°C) and common solvents like dichloromethane and ethyl acetate, making it highly suitable for large-scale industrial manufacturing up to 100 MT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Methylthiazol-5-yl) Ethyl Nitrate Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating the development of life-saving therapies. Our technical team has thoroughly analyzed the pathways described in CN108239040B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and state-of-the-art reactors capable of handling the precise temperature controls required for safe nitration chemistry. Our commitment to delivering products with stringent purity specifications ensures that your research and production timelines remain uninterrupted by quality issues or supply delays.
We invite you to collaborate with us to leverage this advanced synthesis technology for your project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for this vital Alzheimer's intermediate is secure, compliant, and economically optimized for the long term.
