Advanced Synthesis of Thiazole Derivatives: A Strategic Upgrade for Global Supply Chains
The global demand for high-purity heterocyclic building blocks continues to surge, driven by the relentless pace of innovation in both pharmaceutical and agrochemical sectors. Within this landscape, the synthesis of aminomethyl thiazoles stands out as a critical yet historically challenging transformation. Patent CN1968938B introduces a paradigm-shifting methodology that fundamentally reimagines the aminomethylation of thiazole derivatives. Unlike traditional approaches that struggle with selectivity and yield, this invention leverages a novel hexahydrotriazine intermediate to achieve unprecedented purity levels. By strategically utilizing inexpensive reagents such as ammonia and formaldehyde, the process circumvents the thermodynamic pitfalls of direct substitution. This technical breakthrough not only enhances the efficiency of producing key intermediates like 2-chloro-5-(aminomethyl)thiazole but also establishes a robust framework for scalable manufacturing. For industry leaders seeking reliable sources of complex heterocycles, understanding the mechanistic advantages of this patent is essential for optimizing supply chain resilience and cost structures.
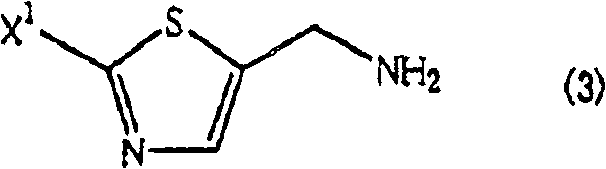
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aminomethyl thiazoles has been fraught with significant inefficiencies that hinder large-scale production. Conventional methods, such as the direct reaction of halomethyl thiazoles with ammonia, often suffer from poor selectivity due to the high nucleophilicity of the resulting primary amine product. Once formed, the primary amine competes with ammonia for the remaining alkyl halide, leading to the pervasive formation of secondary amine byproducts. As illustrated in the structural representation of the problematic impurity, this bis-substitution creates a difficult-to-separate contaminant that drastically lowers the overall yield of the desired mono-amine. Furthermore, alternative routes involving phthalimide or hexamethylenetetramine introduce additional synthetic steps, requiring harsh hydrazinolysis or hydrolysis conditions that generate substantial waste streams. These legacy processes are not only economically burdensome due to low atom economy but also pose environmental compliance challenges that modern manufacturing facilities strive to avoid.
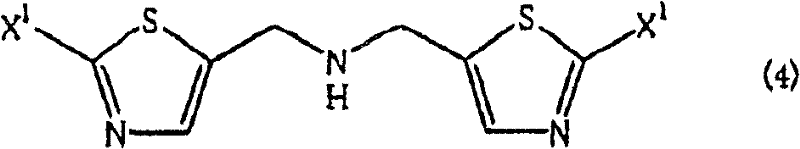
The Novel Approach
In stark contrast to these outdated techniques, the methodology disclosed in CN1968938B employs a clever two-step sequence that effectively masks the reactive amine functionality during the critical substitution phase. By reacting the halomethyl thiazole precursor with a combination of ammonia and formaldehyde, the process generates a stable hexahydrotriazine derivative rather than the free amine. This cyclic intermediate acts as a protected form of the amine, sterically preventing further alkylation and thus eliminating the formation of the troublesome secondary amine byproducts. Subsequent hydrolysis of this triazine ring under mild acidic conditions cleanly releases the target aminomethyl thiazole. This approach transforms a low-yielding, messy reaction into a high-efficiency process with yields exceeding 90% in optimized examples. The ability to utilize cheap, commodity chemicals like formaldehyde and ammonia while achieving pharmaceutical-grade purity represents a significant leap forward in process chemistry, offering a viable path for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Hexahydrotriazine-Mediated Aminomethylation
The success of this novel synthetic route lies in the intricate interplay between kinetics and thermodynamics during the initial aminomethylation step. It is postulated that the reaction proceeds through the in situ formation of an unstable methyleneimine species, generated from the condensation of ammonia and formaldehyde. This highly reactive intermediate rapidly undergoes trimerization to form the thermodynamically stable 1,3,5-hexahydrotriazine ring system. This trimerization effectively sequesters the nitrogen atoms, preventing them from acting as nucleophiles towards the thiazole substrate until the specific hydrolysis step is initiated. The structural integrity of this hexahydrotriazine intermediate is crucial, as it serves as a temporary reservoir for the amine equivalents. By controlling the stoichiometry of formaldehyde relative to ammonia, manufacturers can fine-tune the equilibrium to favor the formation of the triazine over other oligomers. This mechanistic control ensures that when the halomethyl thiazole is introduced, the substitution occurs selectively on the triazine nitrogen or via a concerted mechanism that preserves the mono-substitution pattern, thereby guaranteeing high selectivity.
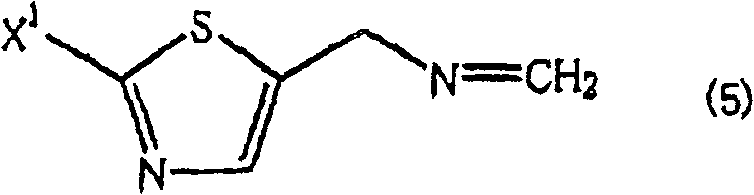
Furthermore, the hydrolysis step offers a unique advantage in terms of impurity management and process control. During the acid-catalyzed cleavage of the hexahydrotriazine ring, formaldehyde is released as a byproduct. The patent discloses a sophisticated strategy to manage this byproduct by conducting the hydrolysis in the presence of lower alcohols such as methanol or ethanol. These alcohols react with the liberated formaldehyde to form acetals, effectively driving the hydrolysis equilibrium towards the completion of the reaction according to Le Chatelier's principle. This in situ scavenging of formaldehyde not only improves the conversion rate but also simplifies the downstream purification process by converting a volatile, toxic aldehyde into a stable, non-interfering acetal. Additionally, the use of aqueous acid solutions allows for the direct formation of the amine salt, which can be easily isolated via crystallization or extraction. This level of mechanistic sophistication ensures that the final product meets stringent purity specifications required for active pharmaceutical ingredient (API) synthesis, minimizing the risk of genotoxic impurities associated with alkylating agents.
How to Synthesize 2-Chloro-5-(aminomethyl)thiazole Efficiently
Implementing this advanced synthesis protocol requires precise control over reaction parameters to maximize the benefits of the hexahydrotriazine pathway. The process begins with the careful selection of solvents and reagents, where methanol is often preferred for its ability to dissolve both the ammonia source and the organic substrates. Operators must maintain the reaction temperature within the optimal range of 20-90°C to ensure rapid formation of the triazine intermediate without promoting decomposition. Following the aminomethylation, the subsequent hydrolysis step demands careful pH management and temperature control to ensure complete ring opening while preserving the integrity of the thiazole ring. Detailed standardized operating procedures are essential to replicate the high yields reported in the patent literature consistently. For a comprehensive guide on the specific molar ratios, addition rates, and workup procedures, please refer to the structured synthesis protocol below.
- React a halomethyl thiazole compound with ammonia and formaldehyde in a solvent like methanol at 15-100°C to form a hexahydrotriazine intermediate.
- Isolate the hexahydrotriazine compound or proceed directly with the reaction mixture containing the intermediate.
- Hydrolyze the hexahydrotriazine intermediate using an aqueous acid solution (e.g., hydrochloric acid) at 10-100°C to obtain the target aminomethyl thiazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this hexahydrotriazine-based synthesis route offers compelling advantages that extend beyond mere technical feasibility. The primary driver for cost optimization lies in the dramatic improvement in reaction yield. By suppressing the formation of secondary amine byproducts, the process significantly increases the amount of usable product obtained per unit of starting material. This enhancement in atom economy directly translates to substantial cost savings in raw material consumption, as less expensive halomethyl thiazole precursor is wasted on unwanted side reactions. Moreover, the reliance on commodity chemicals like ammonia and formaldehyde, which are globally available and priced competitively, reduces exposure to volatile specialty reagent markets. The simplified purification workflow, facilitated by the formation of stable salts and the removal of formaldehyde as acetals, further reduces the operational expenditure associated with chromatography or complex distillation setups. These factors collectively contribute to a more predictable and lower cost of goods sold (COGS), enabling procurement managers to negotiate more favorable long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups and the reduction of waste generation lead to significant operational efficiencies. By avoiding the multi-step sequences required in older methods, such as phthalimide protection and hydrazine deprotection, manufacturers can reduce utility consumption and labor hours. The high selectivity of the reaction minimizes the need for extensive recycling of unreacted starting materials, streamlining the production cycle. Consequently, the overall manufacturing footprint is reduced, allowing for higher throughput in existing facilities without the need for major capital investment in new equipment. This efficiency is critical for maintaining competitiveness in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing dependency on scarce or specialized reagents. Ammonia and formaldehyde are produced at massive scales globally, ensuring a consistent and reliable supply even during market fluctuations. The tolerance of the reaction to various solvent systems and the ability to operate at moderate pressures reduce the risk of production delays caused by equipment failures or safety incidents. Furthermore, the high purity of the crude product simplifies quality control testing, accelerating the release of batches for shipment. This reliability is paramount for pharmaceutical companies that require just-in-time delivery of critical intermediates to maintain their own production schedules for final drug products.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The use of aqueous workups and the conversion of toxic formaldehyde into benign acetals align with green chemistry principles, reducing the environmental burden of the manufacturing process. This compliance with increasingly stringent environmental regulations mitigates the risk of regulatory fines and shutdowns. Additionally, the reduced volume of organic waste solvents lowers disposal costs and simplifies waste management logistics. For supply chain heads, this means a more sustainable and resilient sourcing strategy that aligns with corporate social responsibility goals and future-proofs the supply base against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiazole synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers cover aspects ranging from reaction optimization to impurity profiles, ensuring a comprehensive understanding of the technology's capabilities and limitations.
Q: How does the hexahydrotriazine route improve yield compared to direct ammonolysis?
A: The hexahydrotriazine route significantly suppresses the formation of secondary amine byproducts (bis-thiazole amines) which typically plague direct ammonolysis methods, thereby increasing the yield of the primary amine target from roughly 40% to over 90%.
Q: What are the critical reaction conditions for the aminomethylation step?
A: The reaction typically requires temperatures between 15-100°C (preferably 20-90°C) and pressures not exceeding 0.5 MPa. Using ammonia dissolved in organic solvents like methanol is preferred for operational ease and yield optimization.
Q: Can the hexahydrotriazine intermediate be isolated before hydrolysis?
A: Yes, the intermediate can be isolated via concentration or crystallization, but the patent also discloses a telescoped process where the crude reaction mixture is directly hydrolyzed, simplifying the workflow and reducing processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-(aminomethyl)thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the hexahydrotriazine route are fully realized in practice. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your downstream synthesis processes. Whether you require custom synthesis services or bulk supply of established intermediates, our facility is designed to meet the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this high-yield methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to establish a long-term partnership that drives mutual growth through innovation and reliability. Let us help you secure a competitive advantage in the market with our premium quality thiazole intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
