Advanced POMs-Catalyzed Synthesis of 3-Methylene Isobenzofuran-1(3H)-one Derivatives for Commercial Scale-Up
Advanced POMs-Catalyzed Synthesis of 3-Methylene Isobenzofuran-1(3H)-one Derivatives for Commercial Scale-Up
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable, high-efficiency synthetic routes that minimize environmental impact while maximizing yield. A pivotal development in this domain is detailed in patent CN113004236A, which discloses a novel method for synthesizing 3-methylene isobenzofuran-1(3H)-one compounds. These phthalide derivatives are not merely academic curiosities; they serve as critical scaffolds in the development of bioactive molecules, exhibiting potent anti-inflammatory, anti-diabetic, and anticoagulant properties found in natural products like Chinese angelica. The patent introduces a dehydration coupling reaction between diphenylcarbinol compounds and 2-acylbenzoic acid compounds, catalyzed by polyoxometalate clusters (POMs). This approach represents a paradigm shift from traditional methodologies, offering a robust platform for the production of high-purity pharmaceutical intermediates with exceptional atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-methylene isobenzofuran-1(3H)-one skeleton has relied heavily on intermolecular cyclization strategies such as modified Perkin or Julia condensations, often requiring phthalic anhydride as a starting material. Alternatively, transition metal-catalyzed cyclization of benzoic acid derivatives with olefins or alkynes has been employed. However, these conventional pathways are fraught with significant industrial drawbacks that hinder large-scale adoption. They frequently necessitate harsh reaction conditions, including extreme temperatures and pressures, which escalate energy consumption and operational risks. Furthermore, the reliance on expensive transition metal catalysts and specialized ligands not only inflates the raw material costs but also introduces complex downstream processing challenges. The removal of trace heavy metals to meet stringent pharmaceutical purity standards often requires additional purification steps, generating substantial hazardous waste and extending production lead times, thereby compromising the overall efficiency of the supply chain.
The Novel Approach
In stark contrast, the methodology outlined in the patent leverages the unique acidic and redox properties of polyoxometalates to facilitate a direct dehydration coupling under remarkably mild conditions. By utilizing diphenylcarbinol and 2-acylbenzoic acid as readily available feedstocks, the process bypasses the need for pre-functionalized substrates or toxic reagents. The reaction proceeds efficiently at temperatures ranging from 70-100 °C, with chlorobenzene identified as the optimal solvent, ensuring high solubility and reaction kinetics without thermal degradation of sensitive functional groups. Crucially, the only byproduct generated is water, which simplifies the workup procedure and aligns perfectly with green chemistry principles. This streamlined approach not only enhances the safety profile of the manufacturing process but also significantly reduces the environmental footprint, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Polyoxometalate-Catalyzed Dehydration Cyclization
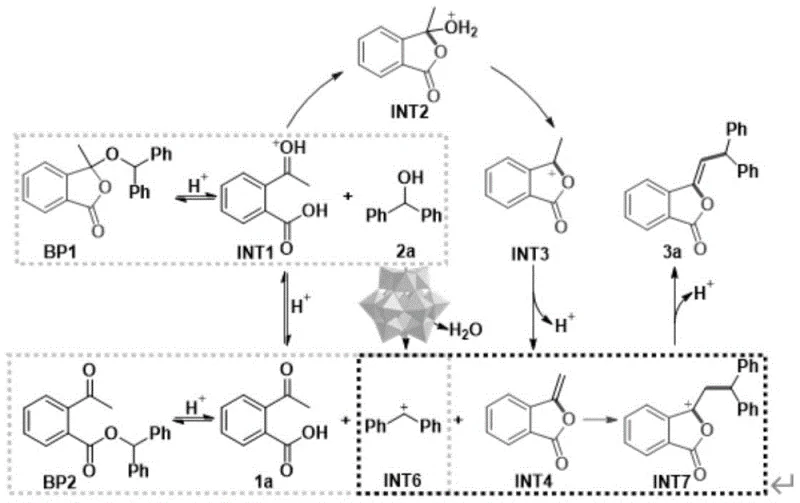
To fully appreciate the technical superiority of this route, one must delve into the intricate catalytic cycle facilitated by the phosphomolybdic acid catalyst. The mechanism initiates with the protonation of the carbonyl oxygen of the 2-acetylbenzoic acid by the strong Brønsted acid sites of the polyoxometalate cluster, generating a highly electrophilic intermediate (INT1). This activation is pivotal as it lowers the energy barrier for the subsequent nucleophilic attack. The carboxyl group then intramolecularly attacks the activated carbonyl, leading to the formation of a cyclic intermediate (INT2). Concurrently, the catalyst promotes the dehydration of the diphenylcarbinol substrate, generating a stabilized carbocation species (INT6). This dual activation strategy ensures that both coupling partners are primed for reaction, minimizing the formation of oligomeric byproducts that often plague acid-catalyzed condensations. The precise control over proton transfer events by the rigid polyoxometalate structure prevents over-reaction and ensures high regioselectivity.
Following the initial activation, the mechanism proceeds through a series of dehydration and deprotonation steps that drive the equilibrium towards the desired lactone product. Intermediate INT2 undergoes dehydration to form INT3, which subsequently loses a proton to yield the exocyclic methylene precursor INT4. The electrophilic carbocation INT6 then attacks INT4, forging the critical carbon-carbon bond that establishes the 3-methylene substitution pattern. The final deprotonation step releases the target 3-methylene isobenzofuran-1(3H)-one compound and regenerates the active catalytic species. This elegant cycle highlights the multifunctional nature of the POMs catalyst, which acts simultaneously as a dehydrating agent and a proton shuttle. Understanding this mechanistic pathway is essential for process chemists aiming to optimize reaction parameters, as it explains why electron-withdrawing substituents on the benzhydrol ring can enhance yields up to 97% by stabilizing the cationic intermediates involved in the rate-determining steps.
How to Synthesize 3-Methylene Isobenzofuran-1(3H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocols defined in the intellectual property to ensure reproducibility and maximum yield. The process is designed to be operationally simple, avoiding the need for inert atmospheres or anhydrous conditions that typically complicate scale-up. The key lies in the precise stoichiometric balance between the benzhydrol and the acylbenzoic acid, with a slight excess of the acid component often proving beneficial to drive the equilibrium forward. The choice of solvent is also critical; while several polar and non-polar solvents were screened, chlorobenzene emerged as the superior medium due to its ability to dissolve both organic substrates while withstanding the reaction temperature without participating in side reactions. The following guide outlines the standardized procedure derived from the patent examples.
- Charge a reaction vessel with benzhydrol compound (0.2-1 mmol), 2-acyl benzoic acid compound (0.2-1 mmol), and polyoxometalate catalyst (1-3 mol%).
- Add an organic solvent such as chlorobenzene (preferred), propylene carbonate, or hexafluoroisopropanol to the mixture.
- Heat the reaction mixture to 70-100 °C (optimally 80 °C) and stir for 0-3 hours until conversion is complete.
- Upon completion, isolate the target 3-methylene isobenzofuran-1(3H)-one product via column chromatography or standard purification techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this POMs-catalyzed technology offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of precious transition metals such as palladium, platinum, or rhodium removes a major source of cost volatility and supply risk from the bill of materials. Since polyoxometalates are composed of abundant earth metals like molybdenum and phosphorus, their pricing is stable and significantly lower than that of noble metals. Furthermore, the absence of heavy metals in the final reaction mixture drastically simplifies the purification workflow. In traditional processes, extensive scavenging steps are required to reduce metal content to parts-per-million levels, which consumes valuable time and resources. By circumventing this bottleneck, manufacturers can achieve faster batch turnover rates and reduce the overall cost of goods sold, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is compelling when analyzed through the lens of total process cost. By utilizing a catalyst loading as low as 1.5 mol%, the process minimizes the expenditure on catalytic materials while maintaining high turnover numbers. The mild reaction conditions (80 °C) translate to lower energy consumption compared to high-temperature reflux methods, reducing utility costs. Additionally, the generation of water as the sole byproduct means that waste disposal fees are negligible, and there is no need for expensive hazardous waste treatment protocols. The simplified downstream processing, potentially allowing for crystallization rather than complex chromatography on a large scale, further drives down operational expenditures, resulting in substantial cost savings for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. Both benzhydrol derivatives and 2-acylbenzoic acids are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction against varying substituent patterns means that the same process platform can be used to synthesize a diverse library of analogues without requalifying entirely new synthetic routes. This flexibility allows for rapid response to market demands or changes in drug development pipelines. Moreover, the stability of the polyoxometalate catalyst ensures consistent batch-to-batch quality, reducing the incidence of failed batches that can disrupt delivery schedules and strain client relationships.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges related to heat transfer and mixing, but this exothermic dehydration is well-suited for large-scale reactors. The low viscosity of the reaction mixture in chlorobenzene ensures efficient agitation and heat dissipation, mitigating the risk of thermal runaways. From a regulatory standpoint, the "green" nature of the process aligns with increasingly stringent environmental regulations globally. The absence of toxic solvents like DMF or DMSO in the preferred embodiment, coupled with the non-toxic nature of the catalyst, simplifies the permitting process for new manufacturing facilities. This environmental compliance not only safeguards the company's reputation but also future-proofs the supply chain against tightening ecological legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using Polyoxometalates (POMs) over transition metal catalysts for this synthesis?
A: The use of Polyoxometalates, specifically phosphomolybdic acid, eliminates the need for expensive and toxic transition metals like palladium or rhodium. This results in a significantly cleaner impurity profile, easier regulatory approval for pharmaceutical intermediates, and reduced costs associated with heavy metal removal processes.
Q: What is the substrate scope regarding substituents on the benzhydrol and acylbenzoic acid?
A: The method demonstrates excellent tolerance for various functional groups. Electron-withdrawing groups such as fluorine, chlorine, and bromine on the benzhydrol ring can yield up to 97%, while electron-donating groups like methyl and isopropyl also provide yields exceeding 85%. The acyl group substituents can range from hydrogen to n-butyl.
Q: How does this method address environmental concerns in fine chemical manufacturing?
A: This protocol is designed as a green synthesis method where water is the only byproduct generated during the dehydration coupling. Combined with the use of non-corrosive, recyclable solid acid catalysts and mild reaction temperatures, it drastically reduces the E-factor and waste treatment burden compared to traditional Perkin or Julia condensation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylene Isobenzofuran-1(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team of expert chemists has extensively evaluated the POMs-catalyzed route described in CN113004236A and is fully prepared to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-methylene isobenzofuran-1(3H)-one meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable, high-quality chemical solutions.
