Scaling High-Purity Sucralose-6-Acetate: A Technical Breakthrough in Cascade Chlorination
Scaling High-Purity Sucralose-6-Acetate: A Technical Breakthrough in Cascade Chlorination
The global demand for high-intensity sweeteners continues to drive innovation in the synthesis of key intermediates like sucralose-6-acetate, a critical precursor in the manufacturing of sucralose. Patent CN102690299A introduces a transformative approach utilizing a tandem reaction mechanism that fundamentally alters the production landscape for this valuable compound. By integrating esterification and chlorination into a seamless cascade process, this technology addresses long-standing inefficiencies associated with traditional multi-step syntheses. For R&D directors and process engineers, the shift towards such consolidated reaction pathways represents a significant opportunity to enhance throughput while maintaining rigorous quality standards. This report analyzes the technical merits of this quaternary ammonium salt-mediated chlorination method, providing a strategic overview for stakeholders evaluating reliable sucralose intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chlorination of sucrose derivatives has been plagued by harsh reaction conditions and significant environmental burdens. Traditional methodologies often rely on Vilsmeier-Haack reagents or thionyl chloride in solvents like DMF, which necessitate stringent temperature controls and generate copious amounts of hazardous byproducts. The evolution of hydrogen chloride and sulfur dioxide gases during these processes not only poses severe safety risks to plant personnel but also mandates the installation of expensive gas scrubbing and neutralization infrastructure. Furthermore, the use of polar aprotic solvents complicates downstream processing, often requiring energy-intensive distillation steps to recover solvents and isolate the product. These factors collectively inflate the operational expenditure (OpEx) and extend the production cycle time, creating bottlenecks for manufacturers aiming to meet the escalating market demand for cost reduction in sweetener manufacturing.
The Novel Approach
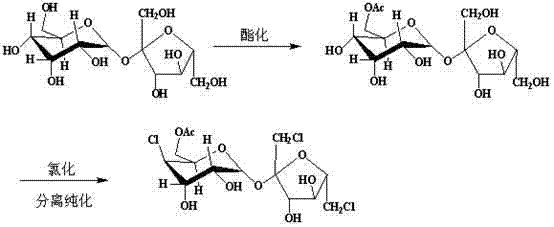
In stark contrast, the methodology outlined in CN102690299A employs a sophisticated cascade reaction strategy that mitigates these historical pain points through elegant chemical design. The process initiates with the selective esterification of the sucrose C-6 hydroxyl group using trimethyl orthoacetate under mild acidic catalysis, followed immediately by an in situ chlorination step mediated by a quaternary ammonium salt generated from tri-tert-butylamine and benzyl chloride. This tandem approach eliminates the need for isolating unstable intermediates, thereby reducing material handling losses and exposure to atmospheric moisture. Crucially, the reaction proceeds without the evolution of toxic corrosive gases, marking a substantial improvement in workplace safety and environmental compliance. The ability to conduct these transformations in a unified reaction vessel significantly streamlines the workflow, offering a compelling value proposition for supply chain heads focused on reducing lead time for high-purity sucralose intermediates.
Mechanistic Insights into Quaternary Ammonium Salt-Mediated Chlorination
The core innovation of this synthesis lies in the generation and utilization of a reactive quaternary ammonium species as the chlorinating agent. In the second stage of the cascade, tri-tert-butylamine reacts with benzyl chloride to form a quaternary ammonium salt within the reaction matrix. This species acts as a potent nucleophilic catalyst and chlorinating source, facilitating the substitution of the hydroxyl group at the C-6 position with high regioselectivity. Unlike free radical chlorination methods which can lead to indiscriminate substitution patterns and complex impurity profiles, this ionic mechanism ensures that the chlorination is directed specifically to the activated primary alcohol site. The reaction temperature is maintained between 90°C and 120°C, preferably around 100°C to 110°C, which provides sufficient thermal energy to overcome the activation barrier for the substitution without inducing thermal degradation of the sensitive carbohydrate backbone. This precise control over reaction kinetics is essential for achieving the reported purity levels exceeding 98.0%.
Furthermore, the mechanistic pathway inherently suppresses the formation of common side products associated with acid-catalyzed degradation of sucrose esters. The absence of strong mineral acids or Lewis acids during the chlorination phase prevents the hydrolysis of the acetate protecting groups or the glycosidic bond cleavage that often plagues alternative routes. The use of ethyl acetate as the extraction solvent in the workup phase further capitalizes on the polarity differences between the desired product and the organic byproducts, such as the amine salts. This selectivity simplifies the purification protocol, allowing for effective recrystallization to remove trace impurities. For technical teams, understanding this mechanism underscores the robustness of the process, validating its potential for commercial scale-up of complex food additive intermediates where consistency is paramount.
How to Synthesize Sucralose-6-Acetate Efficiently
The operational protocol derived from the patent data offers a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the protection of the sucrose molecule, where sucrose and trimethyl orthoacetate are stirred in the presence of a tosic acid catalyst at a controlled temperature of 40°C for approximately 5 hours. Following this esterification, deionized water is introduced to hydrolyze the orthoester, selectively revealing the C-6 hydroxyl group while maintaining protection at other positions. The subsequent addition of tri-tert-butylamine and benzyl chloride triggers the chlorination cascade, which proceeds for 3 to 6 hours at elevated temperatures. The detailed standardized synthesis steps, including precise molar ratios and specific workup parameters, are provided in the technical guide below to ensure reproducibility and safety during implementation.
- Perform esterification of sucrose C-6 hydroxyl using trimethyl orthoacetate and tosic acid catalyst at 40°C.
- Execute selective chlorination by adding tri-tert-butylamine and benzyl chloride to form an active quaternary ammonium salt species in situ.
- Complete the process with ethyl acetate extraction and recrystallization to isolate the final product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cascade reaction technology presents a multitude of strategic benefits that extend beyond mere chemical efficiency. For procurement managers, the elimination of hazardous gas generation translates directly into reduced capital expenditure on safety infrastructure and lower ongoing costs for waste treatment and regulatory compliance. The simplified workflow, which combines multiple synthetic steps into a single pot operation, drastically reduces the consumption of solvents and utilities, leading to substantial cost savings in sweetener manufacturing. Additionally, the use of relatively benign reagents and the avoidance of heavy metal catalysts minimize the risk of supply chain disruptions caused by the scarcity or price volatility of specialized chemical inputs. This resilience is critical for maintaining continuous production schedules in a competitive global market.
- Cost Reduction in Manufacturing: The consolidation of reaction steps significantly lowers the operational overhead by reducing the number of unit operations required. By avoiding the generation of hydrogen chloride and sulfur dioxide, the facility saves on the costs associated with gas scrubbing systems, neutralization agents, and hazardous waste disposal. Furthermore, the ability to recycle the reaction system and the use of standard solvents like ethyl acetate contribute to a leaner cost structure, allowing for more competitive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and stoichiometric ratios, ensures high batch-to-batch consistency. This reliability minimizes the risk of failed batches and production delays, securing a steady flow of materials for downstream customers. The use of commercially available starting materials such as sucrose, benzyl chloride, and tri-tert-butylamine reduces dependency on exotic reagents, thereby mitigating supply chain risks associated with geopolitical instability or logistics bottlenecks.
- Scalability and Environmental Compliance: The green chemistry attributes of this process align perfectly with modern sustainability goals, facilitating easier permitting and community acceptance for manufacturing sites. The reduction in three wastes (wastewater, waste gas, and solid residues) simplifies environmental management and lowers the carbon footprint of the production process. This environmental stewardship not only future-proofs the supply chain against tightening regulations but also enhances the brand value for end-users seeking sustainably sourced ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cascade synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for stakeholders assessing the viability of integrating this method into their existing production portfolios or sourcing strategies.
Q: How does this cascade method improve environmental compliance compared to traditional Vilsmeier chlorination?
A: Unlike traditional methods that generate significant amounts of hydrogen chloride and sulfur dioxide gases requiring complex scrubbing systems, this patented cascade reaction operates without producing these toxic byproducts, significantly simplifying waste gas treatment and reducing environmental liability.
Q: What is the expected purity and yield profile for this synthesis route?
A: Experimental data from the patent indicates a consistent yield of approximately 48% with a final product purity exceeding 98.0% as determined by HPLC, achieved through a streamlined recrystallization process following ethyl acetate extraction.
Q: Can this process be adapted for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability by combining multiple reaction steps into a single pot (tandem reaction), which reduces equipment footprint and operational complexity, making it highly suitable for industrial scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sucralose-6-Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern fine chemicals industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN102690299A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of sucralose-6-acetate adheres to the highest international standards. Our capability to manage complex cascade reactions allows us to offer a stable supply of high-quality intermediates that support the growing demand for non-caloric sweeteners globally.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with confidence and precision. Let us be your partner in driving efficiency and excellence in the production of next-generation food additives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
