Advanced Esterification Technology for High-Purity Dehydrolinalyl Acetate Production and Commercial Scale-Up
The global demand for high-quality fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly within the realm of fine chemical intermediates. Patent CN101209965A introduces a transformative methodology for the preparation of dehydrolinalyl acetate, a critical precursor in the synthesis of premium fragrances such as phantol and phantolid. This technical disclosure outlines a robust esterification process utilizing dehydrolinalool and acetic anhydride in the presence of a sulfosalicylic acid catalyst. Unlike traditional methods that suffer from sluggish kinetics and difficult downstream processing, this novel approach achieves exceptional conversion rates and selectivity under remarkably mild conditions. For R&D directors and process engineers, this patent represents a significant leap forward in optimizing the synthesis of terpene-derived esters, offering a pathway to reduce operational complexity while maintaining stringent purity standards required by the international flavor and fragrance market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of dehydrolinalyl acetate and related terpene esters has been plagued by inefficiencies inherent to traditional acid catalysis. Prior art methods, such as those utilizing phosphoric acid, often necessitate prolonged reaction times extending into tens of hours to achieve acceptable conversion levels. This extended duration not only ties up reactor capacity but also increases the risk of thermal degradation and unwanted side reactions. Furthermore, the removal of phosphoric acid from the organic product matrix is notoriously difficult, often requiring neutralization steps that generate substantial salt waste or complex extraction procedures that compromise overall yield. The reversible nature of the esterification reaction further complicates matters, as achieving high conversion typically drives down selectivity, leading to the formation of isomers and cyclic by-products that degrade the olfactory profile of the final fragrance application.
The Novel Approach
The methodology described in CN101209965A fundamentally alters the kinetic and thermodynamic landscape of this esterification. By employing sulfosalicylic acid as a homogeneous catalyst, the reaction proceeds with remarkable velocity, reaching completion within a window of merely 20 to 150 minutes at temperatures ranging from 25°C to 70°C. This dramatic reduction in reaction time is coupled with near-quantitative selectivity, effectively suppressing the formation of cyclic pyrans and other structural isomers that typically contaminate the product stream. The strategic advantage lies not only in the speed but also in the physicochemical properties of the catalyst itself; sulfosalicylic acid is highly soluble in the aqueous phase, allowing it to be effortlessly washed away along with excess acetic anhydride and acetic acid by-products. This simplifies the isolation of the target ester to a basic phase separation, significantly streamlining the workflow for a reliable flavor & fragrance intermediates supplier.
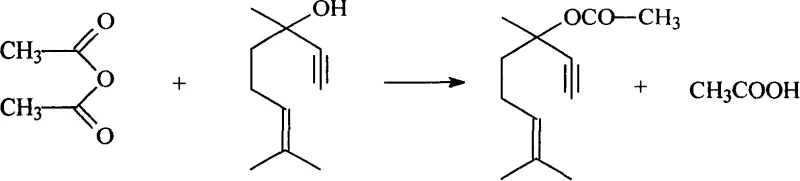
Mechanistic Insights into Sulfosalicylic Acid-Catalyzed Esterification
The efficacy of sulfosalicylic acid in this transformation can be attributed to its dual functionality as a strong organic acid and a chelating agent, which facilitates the activation of the acetic anhydride electrophile. In the catalytic cycle, the sulfonic acid group protonates the carbonyl oxygen of the acetic anhydride, increasing its susceptibility to nucleophilic attack by the hydroxyl group of dehydrolinalool. This activation lowers the energy barrier for the formation of the tetrahedral intermediate, thereby accelerating the rate-determining step of the acylation. Unlike mineral acids that may promote carbocation rearrangements leading to skeletal isomerization of the sensitive terpene backbone, sulfosalicylic acid provides a controlled acidic environment that preserves the integrity of the allylic alcohol structure. This mechanistic precision is crucial for maintaining the specific stereochemical and structural attributes required for high-value fragrance applications, ensuring that the resulting dehydrolinalyl acetate possesses the correct olfactory characteristics.
Impurity control is another critical aspect where this catalytic system excels, particularly regarding the suppression of cyclization side reactions. Under harsher acidic conditions or elevated temperatures, dehydrolinalool is prone to intramolecular cyclization, forming pyran derivatives that are difficult to separate and detrimental to product quality. The mild operating temperature of 25°C to 70°C permitted by the high activity of sulfosalicylic acid inherently mitigates these thermal side pathways. Additionally, the rapid reaction kinetics ensure that the starting material is consumed quickly, minimizing the residence time of the reactive alcohol in the acidic medium. This combination of kinetic acceleration and thermal moderation results in a product profile with exceptionally low levels of impurities, often achieving selectivity figures approaching 100%, which drastically reduces the burden on downstream purification units and enhances the overall economic viability of cost reduction in fine chemical manufacturing.
How to Synthesize Dehydrolinalyl Acetate Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and mixing to maximize the benefits of the sulfosalicylic acid catalyst. The process begins with the charging of dehydrolinalool and acetic anhydride into a reactor equipped with agitation and temperature control, typically under a nitrogen blanket to prevent oxidation. The molar ratio of reactants is flexible, ranging from 1:1 to 1:5, though a slight excess of acetic anhydride is often preferred to drive the equilibrium towards the ester product. Once the reagents are mixed, the catalyst is introduced, and the exothermic nature of the reaction is managed to maintain the temperature within the optimal 35°C to 60°C range. Detailed standardized synthesis steps for laboratory and pilot scale operations are provided in the guide below.
- Charge a reactor with dehydrolinalool and acetic anhydride at a molar ratio ranging from 1: 1 to 1:5, ensuring an inert nitrogen atmosphere.
- Add sulfosalicylic acid catalyst (0.05-5 wt% of total raw materials) and maintain reaction temperature between 25°C and 70°C for 20 to 150 minutes.
- Upon completion, wash the reaction mixture with water to remove the water-soluble catalyst and excess acetic acid, separating the oil phase to obtain the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this sulfosalicylic acid-catalyzed process offers tangible strategic benefits that extend beyond mere chemical yield. The primary value proposition lies in the drastic simplification of the production workflow, which translates directly into reduced operational expenditures and enhanced throughput. By eliminating the need for complex catalyst neutralization and filtration steps associated with solid or insoluble acid catalysts, manufacturers can significantly shorten the batch cycle time. This increased asset utilization allows for greater production volume without the need for capital-intensive expansion of reactor farms, providing a buffer against market volatility and ensuring consistent supply continuity for downstream fragrance blenders and perfume houses.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive and time-consuming purification stages. Since the catalyst is water-soluble, it is removed during the standard aqueous wash, negating the need for specialized adsorbents or distillation columns dedicated to catalyst recovery. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes the loss of valuable raw materials to by-products. These factors combine to lower the variable cost per kilogram of production, creating a more competitive pricing structure for high-purity dehydrolinalyl acetate in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable and reliable supply chain. The tolerance for a wide range of reaction temperatures and catalyst loadings means that the process is less susceptible to minor fluctuations in utility availability or raw material quality. This operational stability reduces the risk of batch failures or off-spec production runs, which are common causes of supply disruptions. Consequently, suppliers utilizing this method can offer more dependable lead times and maintain safety stock levels with greater confidence, assuring customers of uninterrupted access to this critical fragrance intermediate.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles. The reduction in reaction time and temperature lowers the carbon footprint of the manufacturing process, while the ease of catalyst removal minimizes the generation of hazardous solid waste. The aqueous waste stream containing the sulfosalicylic acid can be treated using standard effluent protocols, avoiding the complications associated with heavy metal or persistent organic pollutant disposal. This environmental compatibility facilitates easier permitting for commercial scale-up of complex fragrance intermediates and ensures long-term compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of dehydrolinalyl acetate using this patented methodology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and product quality assurance. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their supply chains.
Q: What are the primary advantages of using sulfosalicylic acid over phosphoric acid for this esterification?
A: Sulfosalicylic acid offers significantly higher catalytic activity, reducing reaction times from tens of hours to under 150 minutes. Furthermore, its high water solubility allows for facile removal during the washing step, eliminating complex purification processes required for phosphoric acid residues.
Q: How does this process control impurities like cyclic pyrans?
A: The mild reaction conditions (25-70°C) and the specific selectivity of the sulfosalicylic acid catalyst minimize side reactions such as isomerization and cyclization. This results in product selectivity approaching 100%, drastically reducing the formation of unwanted by-products like ring-type pyrans.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process operates at normal pressure and moderate temperatures, which reduces energy consumption and equipment stress. The simplified work-up procedure involving simple phase separation makes it highly scalable for commercial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydrolinalyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the competitiveness of the fine chemicals sector. Our technical team has extensively analyzed the potential of the sulfosalicylic acid-catalyzed esterification pathway and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and risk-mitigated. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of dehydrolinalyl acetate meets the exacting standards required for high-end fragrance applications.
We invite procurement professionals and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of this advanced manufacturing process. Let us help you optimize your supply chain and secure a competitive advantage in the dynamic flavor and fragrance market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
