Scalable Synthesis of Epothilone C1-6 Fragments for Commercial Pharmaceutical Production
Scalable Synthesis of Epothilone C1-6 Fragments for Commercial Pharmaceutical Production
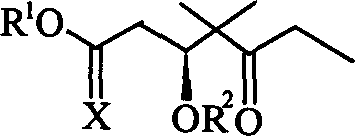
The pharmaceutical industry continuously seeks robust and scalable pathways for complex antineoplastic agents, and the epothilone class represents a pinnacle of modern oncology research. Patent CN1087729C introduces a specialized class of carbon 1-6 fragment compounds derived from macrocyclic epothilones, which serve as critical building blocks for the total synthesis of epothilone and isoepothilone. These fragments are not merely theoretical constructs but are validated intermediates designed to streamline the production of potent microtubule-stabilizing drugs that exhibit cytotoxic activity orders of magnitude higher than paclitaxel against multidrug-resistant tumor cells. The innovation lies in the specific substitution patterns involving triethylsilyl, tert-butyldimethylsilyl, and p-methoxybenzyl groups, which offer enhanced stability and reactivity control during the assembly of the macrocyclic core. By establishing a reliable epothilone intermediate supplier network based on this technology, manufacturers can secure a consistent supply of high-quality precursors essential for next-generation cancer therapeutics. The structural versatility of these fragments, where R1 and R2 groups can be modulated between hydrogen, silyl ethers, and benzyl ethers, allows for fine-tuning of the synthetic route to match specific downstream coupling requirements. This patent provides the foundational chemistry required to transition these complex molecules from laboratory curiosities to commercially viable pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of epothilone fragments has been plagued by inefficient routes that rely on harsh reaction conditions and expensive transition metal catalysts which are difficult to remove to trace levels. Conventional methods often involve multiple protection and deprotection steps that significantly lower the overall yield and increase the waste profile, making cost reduction in pharmaceutical intermediate manufacturing a challenging endeavor. Many existing pathways require cryogenic temperatures or ultra-high pressures that demand specialized equipment, thereby limiting the ability for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the impurity profiles generated by older synthetic routes often include difficult-to-separate byproducts that compromise the stringent purity specifications required for oncology drugs. The reliance on scarce natural products as starting materials in some traditional approaches also introduces supply chain volatility, reducing lead time for high-purity epothilone fragments and creating bottlenecks for production planning. These technical barriers have historically prevented the widespread adoption of epothilone-based therapies, as the cost of goods sold remained prohibitively high for broad market access. Additionally, the lack of modularity in older fragment syntheses meant that any change in the target analog required a complete redesign of the synthetic route, stifling innovation in drug development.
The Novel Approach
The methodology outlined in CN1087729C represents a paradigm shift by utilizing a carbonyl methanolylation strategy that operates under moderate and controllable conditions, significantly simplifying the process infrastructure. This novel approach leverages the reactivity of epoxide precursors under carbon monoxide pressure in the presence of iron or cobalt carbonyl catalysts to directly install the necessary ester functionality with high regioselectivity. By integrating robust protecting group strategies using TBS and PMB moieties early in the sequence, the route ensures that sensitive functional groups are preserved throughout the synthesis, minimizing side reactions and degradation. The flexibility of this method allows for the generation of multiple fragment variants (Compounds 7, 8, and 9) from a common intermediate, providing a versatile platform for synthesizing various epothilone analogs. This modularity is crucial for pharmaceutical R&D teams exploring structure-activity relationships, as it enables rapid access to diverse structural motifs without compromising process efficiency. The use of standard organic solvents and commercially available reagents further enhances the practicality of this route, making it an ideal candidate for technology transfer to large-scale manufacturing facilities. Ultimately, this approach addresses the core limitations of previous methods by balancing chemical elegance with industrial pragmatism.
Mechanistic Insights into Carbonyl Methanolylation and Protection Chemistry
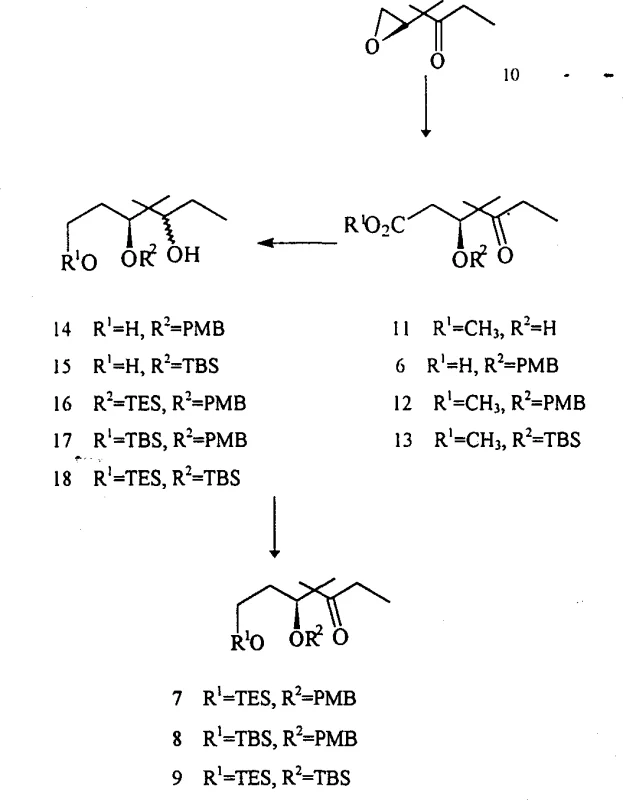
The core of this synthetic innovation relies on the transition metal-catalyzed carbonyl methanolylation of the epoxide starting material, a reaction that elegantly opens the three-membered ring to form a beta-hydroxy ester. In the presence of catalysts such as dicobalt octacarbonyl or iron carbonyl, carbon monoxide inserts into the carbon-oxygen bond, followed by methanolysis to yield the methyl ester intermediate (Compound 11). This transformation is highly sensitive to temperature and pressure, with the patent specifying a range of 50-90°C and 1-100 atm to optimize conversion while minimizing decomposition. The mechanism proceeds through a metal-carbonyl complex that coordinates with the epoxide oxygen, facilitating nucleophilic attack by methanol and subsequent rearrangement. Understanding this mechanistic pathway is vital for process chemists to control the stereochemistry at the newly formed chiral centers, ensuring that the resulting fragment possesses the correct configuration for downstream macrocyclization. The choice of catalyst and ligand environment can be tuned to favor specific regioisomers, providing a level of control that is often absent in non-catalytic ring-opening reactions. This step sets the foundation for the entire synthesis, establishing the carbon skeleton and oxygenation pattern required for the final epothilone structure.
Following the initial ring opening, the synthesis employs a sophisticated sequence of protection, reduction, and oxidation steps to install the final functionality with high fidelity. The hydroxyl groups are protected using silyl chlorides or benzyl halides under basic or acidic conditions, a step that is critical for preventing unwanted side reactions during subsequent reductions. The ester moiety is then selectively reduced to a primary alcohol using hydride sources like lithium aluminum hydride or sodium borohydride, a transformation that must be carefully monitored to avoid over-reduction or epimerization. The resulting diol is then differentially protected to yield compounds such as 16, 17, and 18, which serve as precursors for the final oxidation to the ketone fragments 7, 8, and 9. The oxidation step, utilizing reagents like Dess-Martin periodinane or chromium-based oxidants, converts the secondary alcohol to a ketone without affecting the sensitive silyl ethers. This precise control over oxidation states is essential for maintaining the integrity of the fragment and ensuring compatibility with the thiazole-containing side chain in the final coupling step. The entire sequence demonstrates a deep understanding of functional group tolerance and orthogonal protection strategies.
How to Synthesize Epothilone C1-6 Fragment Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters and purification techniques to ensure high yield and purity. The process begins with the loading of the autoclave with the epoxide starting material, catalyst, and solvent, followed by pressurization with carbon monoxide to drive the methanolylation reaction to completion. Detailed standard operating procedures for temperature ramping and pressure maintenance are essential to replicate the patent results on a larger scale. Following the reaction, the crude mixture is subjected to aqueous workup and silica gel chromatography to isolate the methyl ester intermediate, which serves as the branching point for the various fragment analogs. The subsequent protection and reduction steps are performed under inert atmosphere to prevent moisture sensitivity issues associated with silyl reagents and hydride reducing agents. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during manufacturing operations.
- Perform carbonyl methanolylation on the starting epoxide compound using iron or cobalt carbonyl catalysts under carbon monoxide pressure to generate the methyl ester intermediate.
- Protect the hydroxyl groups of the intermediate using silyl chlorides or p-methoxybenzyl reagents in the presence of acid or base catalysts to form protected esters.
- Reduce the ester functionality to the corresponding alcohol using hydride reducing agents, followed by secondary protection and oxidation to yield the final ketone fragments.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies for oncology intermediates. The elimination of exotic reagents and the use of moderate reaction conditions translate directly into lower operational expenditures and reduced capital investment requirements for manufacturing facilities. By simplifying the synthetic sequence, the process minimizes the number of unit operations, which in turn reduces the potential for material loss and improves the overall mass balance of the production campaign. This efficiency is critical for maintaining competitive pricing in the global pharmaceutical market, where cost pressures are constantly increasing. Furthermore, the robustness of the chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The ability to produce multiple fragment variants from a common intermediate also provides supply chain flexibility, allowing manufacturers to respond quickly to changes in demand for specific epothilone analogs.
- Cost Reduction in Manufacturing: The process achieves cost optimization by utilizing inexpensive and readily available catalysts such as iron carbonyl and cobalt carbonyl, which are significantly cheaper than precious metal alternatives like palladium or rhodium. The moderate temperature and pressure requirements reduce energy consumption and allow for the use of standard glass-lined or stainless steel reactors rather than specialized high-pressure equipment. Additionally, the high selectivity of the reactions minimizes the formation of byproducts, reducing the cost associated with waste disposal and solvent recovery. The overall yield improvement across the multi-step sequence contributes to a lower cost per kilogram of the final fragment, making the final drug product more economically viable. These factors combine to create a manufacturing process that is both economically efficient and environmentally sustainable.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents ensures that the supply chain is resilient to disruptions in the availability of specialized reagents. The synthetic route is designed to be robust, with wide operating windows for temperature and pressure that accommodate variations in raw material quality without compromising product integrity. This reliability is crucial for long-term supply agreements with pharmaceutical companies, where consistency and on-time delivery are paramount. The scalability of the process means that production volumes can be increased rapidly to meet surges in demand without the need for extensive process re-validation. By securing a source of these intermediates based on this technology, procurement teams can mitigate the risk of supply shortages that often plague complex pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions that translate smoothly from laboratory bench scale to multi-ton commercial production. The use of standard workup procedures such as extraction and crystallization facilitates the handling of large volumes of material with minimal safety risks. Furthermore, the reduced waste profile and the use of less hazardous reagents align with modern environmental regulations and green chemistry principles. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of environmental fines or shutdowns. The ability to operate within strict environmental guidelines while maintaining high productivity makes this route an attractive option for companies committed to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these epothilone fragments. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains. The information covers aspects of reaction safety, purity control, and regulatory compliance to provide a comprehensive overview of the process capabilities.
Q: What are the key advantages of this C1-6 fragment synthesis route?
A: The patent describes a route that utilizes mild reaction conditions, specifically carbonyl methanolylation at moderate temperatures (50-90°C) and pressures, avoiding extreme conditions that complicate scale-up. The use of common protecting groups like TBS and PMB ensures compatibility with downstream macrocyclization steps.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production. The reagents used, such as iron carbonyl and standard silyl protecting agents, are commercially available in bulk, and the purification relies on standard silica gel chromatography which is scalable.
Q: What is the purity profile of the resulting fragments?
A: The examples in the patent demonstrate high purity through rigorous column chromatography purification. The specific stereochemistry is maintained through the use of chiral starting materials and stereospecific reduction steps, ensuring the fragments meet the stringent requirements for antineoplastic drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epothilone Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent literature into commercial reality. Our team of experienced chemists is well-versed in the nuances of macrocyclic synthesis and fragment coupling, ensuring that the transition from lab scale to production is seamless and efficient. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, demonstrating our capacity to handle the rigorous demands of the pharmaceutical industry. Our facilities are equipped with state-of-the-art reaction vessels and purification systems capable of maintaining stringent purity specifications required for oncology intermediates. With rigorous QC labs and a commitment to quality assurance, we guarantee that every batch of epothilone fragment meets the highest standards of identity, strength, and purity. Partnering with us means gaining access to a reliable supply chain partner dedicated to your success.
We invite you to discuss your specific requirements with our technical procurement team to explore how this synthesis route can benefit your project. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic advantages of adopting this technology for your manufacturing needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to provide you with the data and support necessary to make informed decisions about your supply chain strategy. Let us help you accelerate your drug development timeline with our proven expertise in pharmaceutical intermediate synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
