Advanced Icariine Manufacturing: Overcoming Scale-Up Barriers With Lanthanide Catalysis
The pharmaceutical and nutraceutical industries are constantly seeking robust supply chains for high-value bioactive compounds, and Icariine stands out as a critical active monomer derived from Herba Epimedii. Recent advancements in chemical synthesis, specifically detailed in patent CN107163014B, have introduced a transformative approach to manufacturing this complex flavonoid. Unlike traditional extraction methods that suffer from seasonal variability and low concentrations, this synthetic pathway offers a consistent, high-purity alternative that is vital for modern drug development. The core innovation lies in the strategic application of lanthanide series metal Lewis acid catalysts during the rearrangement reaction, which fundamentally alters the efficiency profile of the production line. By shifting away from harsh thermal conditions, manufacturers can now access a route that delivers purity levels exceeding 98% while maintaining yields above 30% relative to the starting material. 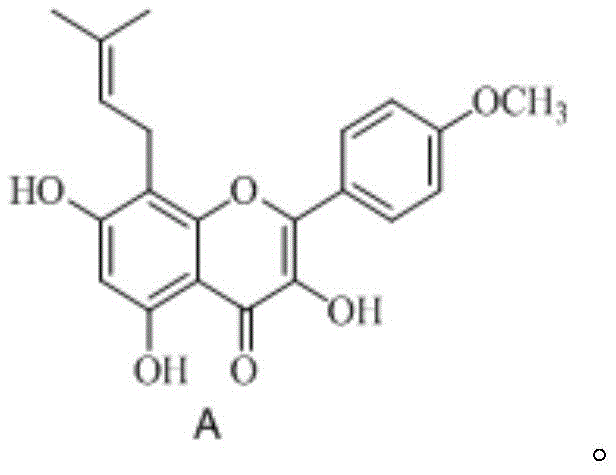
For R&D directors and procurement specialists, understanding the structural integrity of the target molecule is paramount. The synthesis begins with Kaempferol-4'-O-methyl ether, a readily accessible precursor that serves as the foundational scaffold. The ability to construct the specific prenyl side chain with precise regioselectivity is what distinguishes this patent from prior art. In the context of global supply chains, having a synthetic route that does not rely on biological extraction means decoupling production from agricultural constraints. This shift ensures that the reliable pharmaceutical intermediates supplier can maintain continuity of supply regardless of crop yields or geopolitical factors affecting raw botanical materials. The technical depth of this method provides a secure foundation for long-term commercial partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Icariine has been plagued by significant technical bottlenecks that hinder large-scale industrial adoption. Traditional methods often relied on enzymatic hydrolysis of Icariin, a process that is not only costly due to the price of enzymes but also suffers from slow reaction kinetics and difficult downstream processing. Furthermore, earlier synthetic attempts disclosed in patents such as CN101302548B required multiple steps with low overall efficiency, making them economically unviable for mass production. Another prevalent issue was the reliance on microwave-assisted Claisen rearrangement, as seen in prior literature, which poses severe challenges for scale-up due to the difficulty of penetrating microwave energy uniformly in large industrial reactors. 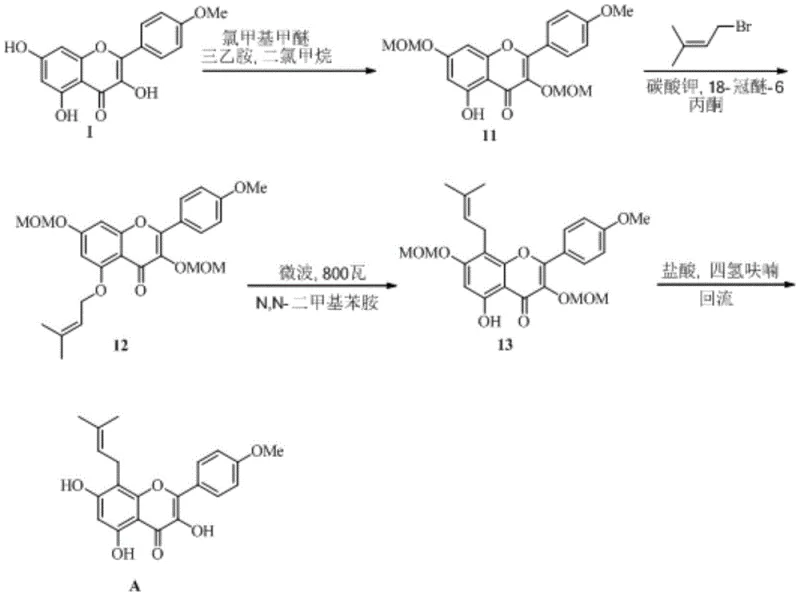
These conventional approaches also tended to generate complex impurity profiles that required extensive chromatographic purification, drastically increasing the cost of goods sold. The use of harsh conditions often led to decomposition of the sensitive flavonoid backbone, resulting in yields that were insufficient for commercial viability. For a procurement manager, these factors translate into unpredictable lead times and volatile pricing structures. The inability to consistently produce high-purity material without expensive purification steps creates a barrier to entry for many potential suppliers. Consequently, the industry has faced a shortage of cost reduction in pharmaceutical intermediates manufacturing, as the legacy technologies simply could not support the economies of scale required by multinational corporations.
The Novel Approach
The methodology outlined in CN107163014B represents a paradigm shift by replacing thermal or microwave energy with chemical catalysis using lanthanide metals. This novel approach utilizes specific salts, such as Europium triflate or Ytterbium triflate, to facilitate the Claisen rearrangement at mild temperatures ranging from -20°C to 40°C. This drastic reduction in thermal stress preserves the integrity of the molecule and significantly suppresses the formation of side products. By eliminating the need for specialized microwave equipment, the process becomes immediately compatible with standard stainless steel reactors found in most fine chemical facilities. This compatibility is a crucial factor for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Furthermore, the new route streamlines the synthesis into a concise four-step sequence from the key intermediate, reducing the operational complexity and labor costs associated with multi-step syntheses. The introduction of the isopentenyl group is achieved through efficient nucleophilic substitution, followed by the catalytic rearrangement and a final deprotection step. This logical flow minimizes the number of isolation points, thereby reducing material loss and solvent consumption. For supply chain heads, this translates to a more robust process with fewer failure points. The ability to achieve high yields without resorting to column chromatography for every step indicates a process designed with industrial practicality in mind, ensuring that reducing lead time for high-purity pharmaceutical intermediates becomes a tangible reality rather than just a promise.
Mechanistic Insights into Lanthanide-Catalyzed Claisen Rearrangement
The heart of this technological breakthrough lies in the mechanistic role of the lanthanide Lewis acid catalyst during the critical rearrangement step. In traditional thermal rearrangements, high energy is required to overcome the activation barrier, often leading to non-selective bond breaking and polymerization. However, the lanthanide ion, with its high oxophilicity and specific coordination geometry, activates the allyl vinyl ether intermediate by coordinating with the oxygen atoms. This coordination lowers the energy of the transition state, allowing the [3,3]-sigmatropic rearrangement to proceed smoothly at near-ambient temperatures. 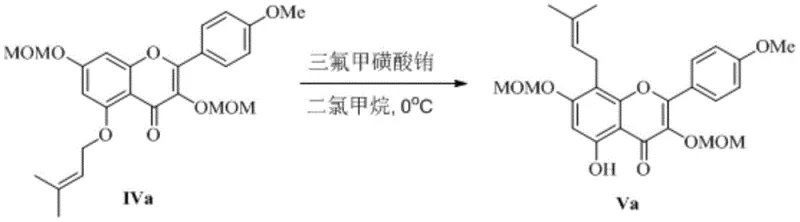
This catalytic cycle is highly specific, ensuring that the prenyl group migrates to the correct position on the flavonoid ring system with high regioselectivity. The use of triflate salts is particularly advantageous because the triflate anion is non-coordinating, leaving the metal center fully available for substrate activation. This results in a cleaner reaction profile where the desired product dominates the mixture. From an R&D perspective, this level of control over the reaction pathway is essential for managing the impurity spectrum. By minimizing the formation of structural isomers and degradation products, the burden on the quality control laboratory is significantly reduced. The mechanism demonstrates how advanced organometallic chemistry can be leveraged to solve practical manufacturing problems, providing a clear path to high-purity OLED material or pharmaceutical grade standards.
Moreover, the catalyst loading is kept relatively low, and the reaction conditions are mild enough to prevent the decomposition of sensitive functional groups present on the flavonoid scaffold. The subsequent deprotection step is also optimized to proceed under controlled acidic conditions, ensuring that the final Icariine molecule retains its biological activity. This attention to mechanistic detail ensures that the process is not just a laboratory curiosity but a viable industrial protocol. The ability to tune the catalyst by selecting between Europium or Ytterbium salts offers additional flexibility to optimize the process for specific production environments. This depth of chemical understanding is what separates a commodity chemical producer from a technology-driven partner capable of handling complex synthetic challenges.
How to Synthesize Icariine Efficiently
The implementation of this synthetic route requires a systematic approach to ensure reproducibility and safety at scale. The process begins with the protection of the phenolic hydroxyl groups on the Kaempferol derivative, followed by the alkylation with an isopentenyl halide. The critical third step involves the addition of the lanthanide catalyst under inert atmosphere to drive the rearrangement. Finally, the protecting groups are removed to reveal the active Icariine. Each step has been optimized in the patent examples to maximize yield and minimize waste, providing a clear roadmap for process engineers. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Protect the 3,7-phenolic hydroxyl groups of Kaempferol-4'-O-methyl ether using protecting groups like MOM or THP.
- Introduce the isopentenyl group via nucleophilic substitution under basic conditions to form the ether intermediate.
- Perform the key Claisen rearrangement using a lanthanide Lewis acid catalyst such as Europium triflate at mild temperatures.
- Execute deprotection under acidic conditions to yield high-purity Icariine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers substantial strategic advantages beyond mere technical feasibility. The elimination of microwave assistance and enzymatic steps removes significant capital expenditure barriers, allowing for production in existing multipurpose facilities. This flexibility enhances supply chain reliability by enabling manufacturing across different geographic locations without the need for specialized infrastructure. The simplified purification process, driven by the high selectivity of the lanthanide catalyst, reduces the consumption of expensive chromatography resins and solvents. This directly contributes to cost reduction in manufacturing, making the final product more competitive in the global market.
- Cost Reduction in Manufacturing: The shift from microwave-assisted reactions to standard thermal catalysis eliminates the need for expensive, hard-to-scale equipment. Additionally, the high selectivity of the lanthanide catalyst reduces the formation of by-products, which in turn lowers the cost associated with waste disposal and purification materials. By avoiding complex enzymatic processes, the reliance on costly biocatalysts is removed, leading to substantial cost savings in raw material procurement. The overall efficiency of the four-step route ensures that the cost per kilogram of the active ingredient is optimized for commercial viability.
- Enhanced Supply Chain Reliability: Synthetic routes are inherently more stable than extraction-based methods, which are subject to agricultural fluctuations. By using commercially available starting materials like Kaempferol-4'-O-methyl ether, the supply chain is insulated from crop failures or seasonal variations. The robustness of the chemical process ensures consistent batch-to-batch quality, which is critical for regulatory compliance in the pharmaceutical sector. This reliability allows partners to plan their inventory with greater confidence, reducing the lead time for high-purity pharmaceutical intermediates and preventing production stoppages.
- Scalability and Environmental Compliance: The process operates at mild temperatures and uses standard solvents, making it easier to scale from pilot plants to multi-ton reactors without significant re-engineering. The reduction in hazardous waste generation, due to higher yields and fewer purification steps, aligns with increasingly strict environmental regulations. This environmental compliance reduces the risk of regulatory fines and enhances the sustainability profile of the supply chain. The ability to scale up complex pharmaceutical intermediates efficiently ensures that market demand can be met without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the technical implementation and commercial viability of this synthesis method. They are derived from the specific pain points identified in the background technology and the beneficial effects claimed in the patent. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their supply chain. The answers reflect the objective data provided in the patent documentation.
Q: What is the primary advantage of using lanthanide catalysts in Icariine synthesis?
A: The use of lanthanide Lewis acid catalysts, such as Europium or Ytterbium triflates, allows the Claisen rearrangement to proceed at significantly lower temperatures compared to traditional microwave-assisted methods. This reduces energy consumption and minimizes the formation of thermal by-products, leading to higher purity profiles.
Q: How does this patent address the scalability issues of previous Icariine production methods?
A: Previous methods relied on microwave irradiation or enzymatic hydrolysis, which are difficult to control and scale in large industrial reactors. The patented chemical synthesis route utilizes standard reactor conditions and commercially available reagents, facilitating a smoother transition from laboratory bench to multi-ton commercial production.
Q: What purity levels can be achieved with this optimized synthetic route?
A: According to the patent data, the final Icariine product can achieve a purity of 98% or higher. The specific catalytic system reduces impurity generation during the critical rearrangement step, simplifying downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Icariine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercial reality. Our team of expert chemists has extensively evaluated the lanthanide-catalyzed route for Icariine and confirmed its potential for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions.
We invite you to discuss how this optimized synthesis can benefit your specific product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process. Let us help you secure a stable, high-quality supply of Icariine that drives your business forward. Contact us today to initiate a conversation about optimizing your supply chain with our advanced manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
