Advanced Palladium-Catalyzed Synthesis of Amide-Substituted Branched Conjugated Dienes for Pharma
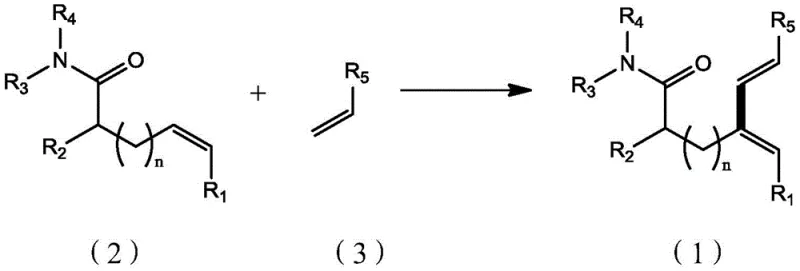
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex carbon skeletons, particularly conjugated diene systems which serve as vital precursors for bioactive molecules. Patent CN110642737B introduces a groundbreaking preparation method for amide-substituted (E,E)-configuration branched conjugated diene derivatives, addressing a significant gap in current synthetic capabilities. Unlike traditional approaches that often struggle with regioselectivity or require harsh conditions, this invention leverages a sophisticated palladium-catalyzed oxidative coupling strategy. By utilizing non-conjugated (Z)-alkenyl amides and electron-deficient olefins, the process achieves high stereoselectivity and yield under remarkably mild conditions. This technological advancement represents a pivotal shift towards more efficient and sustainable manufacturing of high-value chemical intermediates, offering substantial potential for reducing production costs and enhancing supply chain reliability for global pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conjugated diene compounds has relied heavily on methods that are often restricted to conjugated alkenyl amides as substrates, limiting the structural diversity of accessible products. Prior art, such as iridium-catalyzed reactions, frequently necessitates expensive metal catalysts and specific directing groups like NH-Ts amides, which add complexity to the synthesis and purification stages. Furthermore, many existing protocols predominantly yield linear coupling products due to the activation of ortho-alkenyl carbon-hydrogen bonds, failing to provide access to the coveted branched architectures required for certain drug candidates. These conventional pathways often suffer from poor atom economy, require stoichiometric amounts of toxic reagents, or demand extreme reaction temperatures that compromise the integrity of sensitive functional groups, thereby creating significant bottlenecks in the development of new therapeutic agents.
The Novel Approach
The innovative methodology disclosed in the patent overcomes these historical barriers by employing a palladium-catalyzed system that specifically targets non-conjugated (Z)-alkenyl amides. A distinguishing feature of this approach is the use of inexpensive amino acids, such as N-acetyl-L-phenylalanine, as ligands, which drastically reduces the cost of goods compared to proprietary phosphine ligands or noble metals like iridium. The reaction proceeds through a unique cyclometallic transition state where the double bond is outside the ring, enabling selective activation of the homocarbon alkenyl C-H bond. This specificity ensures the formation of branched (E,E)-configured dienes with exceptional stereocontrol. Additionally, the compatibility with a wide range of electron-deficient olefins, including various acrylates, allows for the rapid generation of diverse chemical libraries, facilitating faster lead optimization in drug discovery programs without the need for extensive protecting group chemistry.
Mechanistic Insights into Pd-Catalyzed Oxidative Coupling
The core of this synthetic breakthrough lies in the precise mechanistic pathway involving palladium-catalyzed C-H activation. The reaction initiates with the coordination of the amide directing group to the palladium center, followed by the formation of a cyclometallic intermediate. Crucially, mechanistic studies utilizing deuterium-labeled substrates have confirmed that the reaction proceeds via a specific transition state that activates the alkenyl C-H bond at the same carbon position as the double bond, rather than through an allylic palladium intermediate. This distinction is vital for understanding the regioselectivity observed in the final products. The presence of silver carbonate as an oxidant facilitates the regeneration of the active palladium species, while the amino acid ligand stabilizes the catalytic cycle, ensuring high turnover numbers. The inclusion of trifluoroethanol as an additive further enhances the reaction efficiency by modulating the polarity of the medium and stabilizing key intermediates, leading to the exclusive formation of the thermodynamically stable (E,E)-isomer.
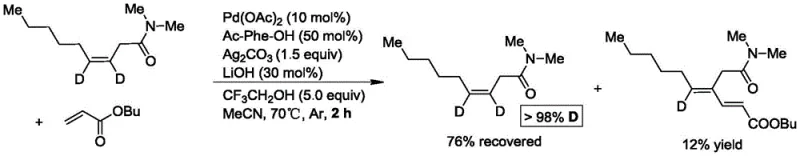
Impurity control is inherently built into the mechanism of this reaction due to its high chemoselectivity and stereospecificity. The rigorous exclusion of allylic palladium intermediates prevents the formation of unwanted linear byproducts or isomeric mixtures that typically plague diene synthesis. The use of a well-defined ligand sphere around the palladium center minimizes side reactions such as polymerization of the acrylate or over-oxidation of the substrate. Furthermore, the mild reaction temperature of 70°C prevents thermal degradation of sensitive functional groups, ensuring that the crude reaction mixture contains a high proportion of the desired product. This inherent purity simplifies downstream processing, as fewer chromatographic steps are required to remove structurally similar impurities. For quality control teams, this means that the impurity profile is predictable and manageable, adhering to stringent regulatory standards required for pharmaceutical intermediate manufacturing without the need for complex recrystallization protocols.
How to Synthesize Amide-Substituted Branched Conjugated Dienes Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these valuable intermediates. The process begins by charging a clean reaction vessel with the palladium catalyst, amino acid ligand, oxidant, base, and additive in an organic solvent such as acetonitrile. The specific molar ratios are optimized to balance reaction rate and cost, typically using 10 mol% of Pd(OAc)2 and 50 mol% of the amino acid ligand. After establishing an inert atmosphere, the non-conjugated alkenyl amide and the electron-deficient olefin are introduced, and the mixture is heated to promote the coupling. The detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different batches and scales.
- Prepare the reaction vessel with Pd(OAc)2 catalyst, Ac-Phe-OH ligand, Ag2CO3 oxidant, LiOH base, and TFE additive in acetonitrile.
- Add non-conjugated (Z)-alkenyl amide and electron-deficient olefin (e.g., acrylate) under an inert argon atmosphere.
- Heat the mixture to 70°C for 16 hours, then purify the resulting (E,E)-branched conjugated diene via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers compelling advantages by significantly lowering the barrier to entry for producing complex diene structures. The substitution of expensive iridium catalysts with abundant palladium salts and cheap amino acid ligands translates directly into reduced raw material costs. Moreover, the reaction conditions are mild enough to be performed in standard glass-lined reactors without the need for specialized high-pressure or cryogenic equipment, which lowers capital expenditure requirements for manufacturing facilities. The high yield reported, reaching up to 74% in optimized examples, ensures efficient utilization of starting materials, minimizing waste disposal costs and maximizing output per batch. This efficiency is critical for maintaining healthy margins in the competitive fine chemical market.
- Cost Reduction in Manufacturing: The elimination of costly noble metal catalysts like iridium and the use of commercially available amino acids as ligands drastically reduce the bill of materials. Additionally, the mild reaction temperature of 70°C lowers energy consumption compared to processes requiring reflux or high heat. The high selectivity reduces the need for extensive purification, saving on solvent usage and labor costs associated with chromatography. These factors combine to create a leaner, more cost-effective manufacturing process that enhances overall profitability.
- Enhanced Supply Chain Reliability: The starting materials, including various acrylates and alkenyl amides, are commodity chemicals with robust global supply chains, reducing the risk of shortages. The simplicity of the reaction setup means that production can be easily transferred between different manufacturing sites without significant re-validation efforts. The stability of the reagents and the tolerance of the reaction to various functional groups allow for flexible sourcing strategies, ensuring continuous supply even if one vendor faces disruptions. This resilience is paramount for long-term project planning and meeting delivery commitments.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, moving seamlessly from gram-scale laboratory experiments to multi-kilogram production runs. The use of silver carbonate as an oxidant generates manageable byproducts, and the overall atom economy is favorable compared to traditional cross-coupling methods. The ability to operate in common solvents like acetonitrile simplifies waste stream management and solvent recovery. These environmental benefits align with modern green chemistry principles, helping companies meet increasingly strict regulatory compliance standards regarding hazardous waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing workflows. The answers reflect the proven capabilities of the method as demonstrated in the provided examples.
Q: What is the key advantage of this Pd-catalyzed method over traditional Ir-catalyzed routes?
A: This method utilizes cheap amino acid ligands instead of expensive iridium catalysts, operates under milder conditions, and specifically targets non-conjugated substrates to form branched rather than linear products.
Q: What is the stereoselectivity of the resulting diene products?
A: The reaction exclusively produces (E,E)-configuration branched conjugated dienes, as confirmed by NMR coupling constants and NOE effects, ensuring high stereochemical purity for downstream applications.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process uses readily available raw materials like acrylates and alkenyl amides, operates at moderate temperatures (70°C), and achieves yields up to 74%, making it highly suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide-Substituted Branched Conjugated Diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis for advancing pharmaceutical research and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for complex intermediates like these branched conjugated dienes. We are committed to delivering high-quality materials that meet the exacting standards of the global pharmaceutical industry, leveraging our technical expertise to optimize every step of the manufacturing process for maximum efficiency and consistency.
We invite you to collaborate with us to explore how this innovative synthesis route can accelerate your drug development timeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to supporting your success with cost-effective, high-purity chemical solutions.
