Scalable Synthesis of 4'-Substituted-9-Deoxo-9a-Aza-9a-Homoerythromycin A Derivatives for Commercial API Production
Scalable Synthesis of 4'-Substituted-9-Deoxo-9a-Aza-9a-Homoerythromycin A Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of next-generation macrolide antibiotics, particularly those derived from the erythromycin scaffold. Patent CN1503804A discloses a highly efficient process for the preparation of 4'-substituted-9-deoxo-9a-aza-9a-homoerythromycin A derivatives, which serve as critical intermediates in the synthesis of potent antibacterial agents. This technology addresses longstanding challenges in macrolide synthesis, specifically focusing on improving yield, purity, and operational safety during the key ring-expansion and functionalization steps. By leveraging a novel epoxide ring-opening strategy in isopropanol, the method circumvents the need for harsh pressure conditions often associated with traditional azalide synthesis. For a reliable API intermediate supplier, understanding the nuances of this chemistry is essential for ensuring consistent supply chains and meeting the stringent quality requirements of global regulatory bodies.
The significance of this patent lies in its ability to produce compounds with exceptional purity profiles, exceeding 98% as determined by HPLC analysis, which is a prerequisite for parenteral formulations. The process involves a sophisticated sequence of protection, oxidation, epoxidation, and nucleophilic substitution, each optimized to minimize impurity formation. Unlike earlier methods that suffered from low yields and unstable intermediates, this approach utilizes stable salt forms, such as the trifluoroacetate salt, to facilitate isolation and storage. This technical advancement not only enhances the chemical efficiency but also provides a solid foundation for cost reduction in pharmaceutical manufacturing by reducing waste and processing time. As we delve deeper into the mechanistic details, it becomes clear why this route represents a superior choice for commercial scale-up of complex antibiotic derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-deoxo-9a-aza-9a-homoerythromycin derivatives has been plagued by significant technical hurdles that impede efficient large-scale production. Traditional methods often rely on the direct amination of epoxide intermediates under conditions that require elevated pressures or the use of volatile amines without solvent, leading to safety concerns and operational complexity. For instance, previous protocols described in prior art documents necessitated reactions at the boiling point of pure alkylamines, which forced the use of pressurized reactors to maintain liquid phase at higher temperatures, thereby increasing capital expenditure and risk. Furthermore, these conventional routes frequently resulted in low conversion rates, sometimes yielding less than 11% of the desired product, due to competing hydrolysis reactions and the instability of the epoxide ring under acidic or basic conditions. The presence of unwanted impurities, such as hydrolyzed byproducts or incomplete reaction species, complicated the downstream purification processes, requiring extensive chromatography or recrystallization steps that drove up costs and reduced overall throughput.
The Novel Approach
In stark contrast, the methodology outlined in CN1503804A introduces a streamlined and safer approach that operates under atmospheric pressure using isopropanol as a co-solvent. This innovation allows the reaction between the epoxide intermediate and tri-n-propylamine to proceed efficiently at moderate temperatures ranging from 50°C to 55°C, eliminating the need for specialized pressure vessels. The use of isopropanol not only solubilizes the reactants effectively but also stabilizes the transition state, leading to significantly improved yields exceeding 85%. This shift in process parameters translates directly to enhanced supply chain reliability, as the manufacturing setup becomes less dependent on complex engineering controls and more focused on standard chemical processing equipment. The ability to conduct the reaction without additional catalysts further simplifies the workup procedure, reducing the load on waste treatment systems and minimizing the risk of metal contamination in the final active pharmaceutical ingredient.
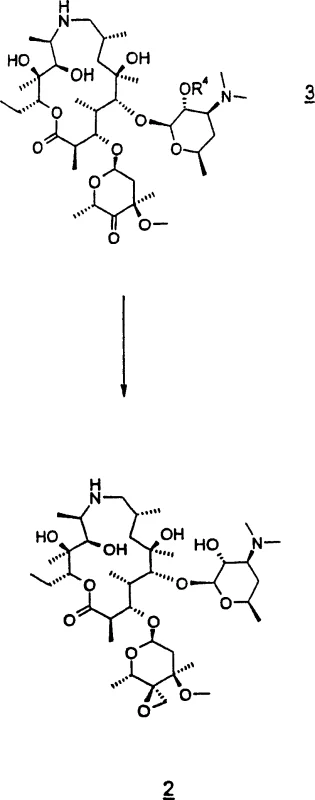
Mechanistic Insights into Epoxide Ring-Opening and Oxidation
The core of this synthetic strategy relies on a precise sequence of functional group transformations that ensure high regioselectivity and stereochemical integrity. The process begins with the selective protection of the 2'-hydroxyl group using benzyl chloroformate, which prevents unwanted side reactions during the subsequent oxidation step. Following protection, the 4'-hydroxyl group is oxidized to a ketone using an activated DMSO protocol, commonly known as Swern oxidation conditions, which are conducted at low temperatures to prevent degradation of the sensitive macrolide backbone. This oxidation step is critical, as the resulting ketone serves as the precursor for the epoxidation reaction, where a sulfonium ylide is employed to introduce the epoxide ring at the 4'-position. The careful control of stoichiometry and temperature during this phase ensures that the epoxide is formed with minimal formation of di-CBZ impurities, which are difficult to remove in later stages.
Once the epoxide intermediate is secured, the pivotal ring-opening reaction takes place, driven by the nucleophilic attack of tri-n-propylamine. The mechanism involves the opening of the strained epoxide ring, which is facilitated by the polar protic environment provided by the isopropanol solvent. This solvent choice is crucial, as it helps to stabilize the developing charge in the transition state without promoting hydrolysis, a common pitfall in aqueous or overly acidic environments. The reaction proceeds through a concerted mechanism that retains the stereochemical configuration required for biological activity, ultimately yielding the 4'-substituted amine derivative. The final product is isolated as a stable phosphate salt or free base, depending on the specific formulation requirements, with the entire pathway designed to maximize atom economy and minimize the generation of hazardous waste streams.
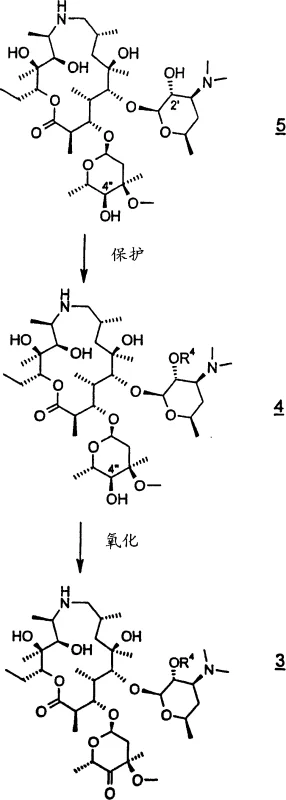
How to Synthesize 4'-Substituted-9-Deoxo-9a-Aza-9a-Homoerythromycin A Efficiently
Executing this synthesis requires strict adherence to the optimized conditions regarding temperature, solvent ratios, and reagent addition rates to ensure reproducibility and safety. The initial protection step must be maintained below 5°C to control exothermicity, followed by a controlled warm-up to facilitate complete conversion before proceeding to the oxidation phase. Operators must ensure that the DMSO activation is performed at cryogenic temperatures, typically around -70°C, to avoid side reactions that could compromise the purity of the ketone intermediate. The subsequent epoxidation and ring-opening steps demand precise monitoring of reaction progress via HPLC to determine the optimal quenching point, ensuring that the maximum amount of starting material is converted while minimizing degradation. Detailed standardized synthesis steps are provided below to guide technical teams in implementing this robust manufacturing protocol.
- Protect the 2'-hydroxyl group of the starting macrolide using benzyl chloroformate in methylene dichloride to form the Cbz-protected intermediate.
- Oxidize the 4'-hydroxyl group to a ketone using activated DMSO (Swern conditions) followed by isolation as a trifluoroacetate salt.
- Convert the ketone to an epoxide using sulfonium ylide, then perform ring-opening with tri-n-propylamine in isopropanol at 50-55°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented process offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of high-pressure reactors and exotic catalysts significantly lowers the barrier to entry for manufacturing partners, allowing for a broader base of qualified suppliers who can meet demand without massive capital investment. This flexibility is crucial for maintaining supply continuity, especially in a market where demand for macrolide antibiotics can fluctuate rapidly due to seasonal infections or emerging resistance patterns. By simplifying the equipment requirements, companies can reduce lead time for high-purity API intermediates, as the setup and teardown times for batch processes are drastically shortened compared to complex high-pressure operations. Furthermore, the use of common solvents like isopropanol and methylene dichloride ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from volatility associated with specialized reagents.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and high-pressure containment systems, which are major cost drivers in traditional macrolide synthesis. The high yield of the ring-opening step means that less raw material is wasted, directly lowering the cost of goods sold per kilogram of finished intermediate. Additionally, the ability to isolate stable salt intermediates reduces the loss of material during storage and transport, ensuring that the value created during synthesis is preserved until the final formulation stage. These efficiencies compound over large production volumes, resulting in substantial cost savings that can be passed down to the end customer or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The robustness of the chemical route ensures that production schedules are less susceptible to delays caused by equipment failure or complex troubleshooting. Since the reaction conditions are milder and operate at atmospheric pressure, the risk of unplanned shutdowns due to safety interlocks or pressure deviations is minimized. This reliability allows supply chain managers to forecast delivery dates with greater confidence, facilitating better inventory planning and reducing the need for excessive safety stock. The stability of the intermediates also means that production can be decoupled, with key precursors manufactured in advance and stored safely, providing a buffer against sudden spikes in demand or disruptions in the upstream supply of starting materials.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of complex engineering constraints like high-pressure mixing or cryogenic continuous flow requirements. The solvent systems used are well-understood in the industry, making waste treatment and recycling processes easier to implement and validate according to environmental regulations. By reducing the generation of heavy metal waste and minimizing solvent consumption through high-concentration reactions, the process aligns with modern green chemistry principles. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing product portfolios. The clarity provided here aims to resolve uncertainties regarding purity, safety, and operational requirements, fostering confidence in the technology's commercial viability.
Q: What are the purity specifications for the final API intermediate?
A: The patented process achieves a purity of at least 98% as measured by HPLC, which is suitable for parenteral formulation preparation without extensive further purification.
Q: Does the synthesis require high-pressure reactors?
A: No, the novel ring-opening step operates effectively at atmospheric pressure or slight vacuum, eliminating the need for expensive high-pressure equipment required by previous methods.
Q: How is the stability of the intermediates managed during storage?
A: The process utilizes stable trifluoroacetate salt forms for key intermediates, which prevents degradation and allows for safe long-term storage and transport before the final deprotection step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Substituted-9-Deoxo-9a-Aza-9a-Homoerythromycin A Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. Our technical team is adept at navigating the complexities of macrolide chemistry, utilizing stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to handle sensitive intermediates with the utmost care and precision. By partnering with us, you gain access to a wealth of process knowledge that can help optimize your specific application, whether for oral, parenteral, or veterinary formulations.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific product pipeline. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this more efficient manufacturing method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume requirements and timeline. Let us help you secure a stable, high-quality supply of critical antibiotic intermediates that will support your long-term business goals and enhance your competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
