Advanced Carbonyl Azetidine Naphthalimide Dyes for High-Resolution Bio-Imaging and Commercial Scale-Up
Advanced Carbonyl Azetidine Naphthalimide Dyes for High-Resolution Bio-Imaging and Commercial Scale-Up
Introduction to Patent CN112939936A Technology
The landscape of organic fluorescent dyes is undergoing a significant transformation driven by the escalating demands of super-resolution fluorescence imaging and single-molecule detection technologies. Patent CN112939936A introduces a groundbreaking class of carbonyl azetidine substituted naphthalimide fluorescent dyes that address critical limitations in photostability and brightness found in conventional fluorophores. This innovation leverages a 1,8-naphthalimide fluorophore parent structure, covalently linking a carbonyl azetidine group at the 4-position to create a robust electron donor-acceptor system. The technical breakthrough lies in the specific molecular architecture which compensates for the deficiencies of standard azetidine rings, thereby drastically improving the fluorescence quantum yield and resistance to photobleaching under intense illumination. For R&D directors and procurement specialists in the fine chemical and pharmaceutical sectors, this patent represents a viable pathway to acquiring high-performance imaging reagents that maintain signal integrity over extended observation periods. The synthesis method described is notably straightforward, utilizing accessible raw materials and avoiding complex catalytic systems, which inherently lowers the barrier for commercial scale-up and ensures a reliable supply chain for these specialized electronic chemical materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic fluorescent dyes, particularly those based on standard 1,8-naphthalimide scaffolds, often suffer from insufficient molar absorptivity and limited photostability when subjected to the rigorous conditions of modern confocal microscopy. Conventional modifications involving simple azetidine rings have shown some improvement in brightness but fail to deliver the necessary quantum efficiency required for emerging applications like super-resolution imaging. The electron transfer mechanisms in these older dye generations are often inefficient, leading to rapid signal degradation and high background noise during long-term biomolecule tracking. Furthermore, many existing synthetic routes rely on expensive transition metal catalysts or harsh reaction conditions that generate significant hazardous waste, complicating the purification process and inflating the overall cost of goods sold. These technical bottlenecks result in a supply of fluorescent dyes that are not only costly but also inconsistent in quality, posing a significant risk to research continuity and data reliability for pharmaceutical and biotechnology companies relying on precise imaging data for drug discovery and development pipelines.
The Novel Approach
The novel approach detailed in the patent data overcomes these historical challenges by integrating a carbonyl azetidine moiety directly into the electron donor end of the naphthalimide structure. This specific structural modification creates a more favorable charge transfer state, resulting in a fluorescence quantum yield in water that can reach up to 0.49, a substantial improvement over prior art. The synthetic strategy employs a nucleophilic substitution reaction followed by an amidation step, both of which operate under relatively mild conditions using common organic solvents like dimethyl sulfoxide and N,N-dimethylformamide. 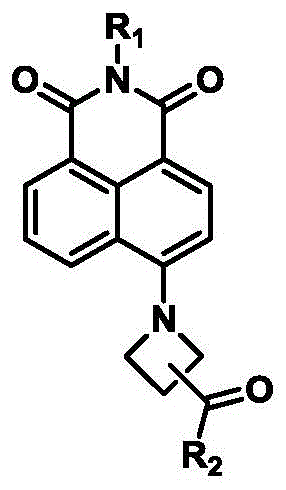 This design not only enhances the photophysical properties, such as wider absorption and emission peaks with a large Stokes shift, but also simplifies the downstream processing. By eliminating the need for precious metal catalysts, the new method reduces the complexity of impurity profiles, making purification via silica gel column chromatography highly effective. This streamlined process translates directly into commercial advantages, offering a more sustainable and cost-efficient manufacturing route that aligns with the increasing regulatory pressures on environmental compliance in the fine chemical industry.
This design not only enhances the photophysical properties, such as wider absorption and emission peaks with a large Stokes shift, but also simplifies the downstream processing. By eliminating the need for precious metal catalysts, the new method reduces the complexity of impurity profiles, making purification via silica gel column chromatography highly effective. This streamlined process translates directly into commercial advantages, offering a more sustainable and cost-efficient manufacturing route that aligns with the increasing regulatory pressures on environmental compliance in the fine chemical industry.
Mechanistic Insights into Carbonyl Azetidine Substitution
The enhanced performance of these dyes is rooted in the sophisticated electronic interplay between the naphthalimide core and the carbonyl azetidine substituent. Mechanistically, the 1,8-naphthalimide acts as a strong electron acceptor, while the nitrogen atom in the azetidine ring serves as an electron donor, facilitating an intramolecular charge transfer (ICT) upon excitation. The introduction of the carbonyl group adjacent to the azetidine ring modulates the electron-donating capability of the nitrogen, preventing excessive electron density that can lead to non-radiative decay pathways. This fine-tuning ensures that a higher proportion of the absorbed energy is emitted as fluorescence, thereby boosting the quantum efficiency. Additionally, the rigid structure of the azetidine ring restricts intramolecular rotation, a common cause of fluorescence quenching in flexible dye molecules, thus contributing to the observed superior photostability. For technical teams evaluating this technology, understanding this mechanism is crucial as it validates the dye's suitability for demanding applications where signal consistency is paramount, such as tracking dynamic biological processes in living cells without the interference of rapid photobleaching.
Impurity control is another critical aspect of the mechanistic design, as the synthetic route minimizes side reactions that typically plague naphthalimide derivatization. The use of potassium carbonate as a base in the initial substitution step ensures selective reaction at the 4-bromo position without affecting the imide functionality, which is sensitive to harsh nucleophiles. Subsequent amidation using EDC and HOBt activates the carboxylic acid intermediate efficiently, promoting high conversion rates while minimizing the formation of urea byproducts often associated with carbodiimide coupling. 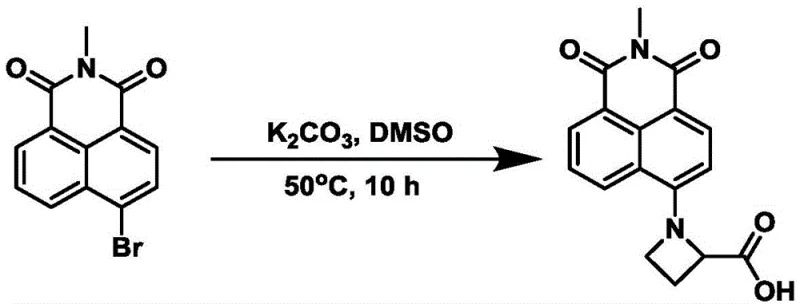 This high selectivity results in a cleaner crude product, reducing the burden on purification steps and ensuring that the final dye meets the stringent purity specifications required for biological applications. The ability to control the impurity profile through precise stoichiometry and temperature management, as evidenced by the reaction conditions of 50-140°C, demonstrates a robust process that can be reliably transferred from laboratory scale to commercial production environments without compromising product quality or batch-to-batch consistency.
This high selectivity results in a cleaner crude product, reducing the burden on purification steps and ensuring that the final dye meets the stringent purity specifications required for biological applications. The ability to control the impurity profile through precise stoichiometry and temperature management, as evidenced by the reaction conditions of 50-140°C, demonstrates a robust process that can be reliably transferred from laboratory scale to commercial production environments without compromising product quality or batch-to-batch consistency.
How to Synthesize Carbonyl Azetidine Naphthalimide Efficiently
The synthesis of these high-value fluorescent dyes follows a logical two-step sequence that prioritizes yield and purity while utilizing standard chemical engineering unit operations. The process begins with the nucleophilic displacement of the bromine atom on the naphthalimide core, followed by the coupling of the resulting carboxylic acid intermediate with various amine functionalities to tune the solubility and targeting properties of the final dye. This modular approach allows for the rapid generation of a library of derivatives, such as the NI-Aze series, by simply varying the amine component in the second step. The operational simplicity, involving standard heating and stirring under nitrogen protection, makes this route highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of optoelectronic materials. 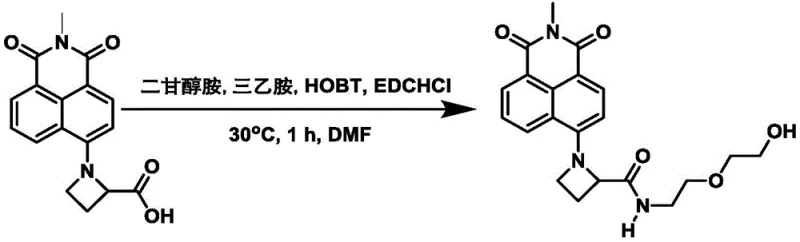 The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and purification protocols that ensure optimal results.
The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and purification protocols that ensure optimal results.
- Perform nucleophilic substitution of N-methyl-4-bromo-1,8-naphthalimide with azetidine carboxylic acid in DMSO using potassium carbonate at elevated temperatures.
- Activate the intermediate carboxylic acid using EDC/HOBt coupling reagents in dry DMF.
- React the activated intermediate with diglycolamine or methoxy ethyl amine derivatives to finalize the dye structure with high quantum yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical performance. The elimination of expensive transition metal catalysts, which are often subject to volatile market pricing and supply constraints, significantly stabilizes the raw material cost structure. This shift to organocatalytic or base-mediated reactions reduces the dependency on critical minerals, thereby mitigating supply chain risks associated with geopolitical instability or mining disruptions. Furthermore, the use of common, bulk-available solvents like DMSO and DMF simplifies logistics and inventory management, allowing for just-in-time manufacturing strategies that reduce warehousing costs and improve cash flow. For supply chain heads, this translates into a more resilient sourcing strategy where the continuity of supply is less likely to be interrupted by external factors, ensuring that R&D and production timelines are met without delay.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent inherently lowers manufacturing costs by removing the need for costly metal scavenging steps that are mandatory when using transition metal catalysts in pharmaceutical intermediate production. By avoiding these complex purification stages, the overall processing time is shortened, and the consumption of auxiliary materials such as specialized resins is drastically reduced. This streamlined process flow leads to substantial cost savings in terms of both labor and material inputs, making the final fluorescent dye more price-competitive in the global market. Additionally, the high yields reported in the examples suggest a material-efficient process that minimizes waste generation, further contributing to a lower cost of goods sold and improved profit margins for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as N-methyl-4-bromo-1,8-naphthalimide and azetidine carboxylic acids, ensures that the supply chain is not bottlenecked by exotic or scarce reagents. These precursors are produced by multiple suppliers globally, fostering a competitive sourcing environment that protects against price gouging and availability issues. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry or anaerobic environments beyond standard nitrogen protection, means that production can be maintained across different manufacturing sites with varying levels of infrastructure. This flexibility enhances supply chain reliability, allowing for diversified production networks that can quickly adapt to regional demands or unexpected disruptions in specific facilities.
- Scalability and Environmental Compliance: Scaling this synthesis from laboratory to commercial production is facilitated by the absence of hazardous reagents and the use of standard exothermic control measures. The waste stream is primarily composed of organic salts and solvents that can be managed through established recovery and treatment protocols, aligning with increasingly strict environmental regulations in the fine chemical sector. The process does not generate heavy metal waste, which is a significant liability in terms of disposal costs and regulatory compliance. This environmental friendliness not only reduces the operational risk associated with waste management but also enhances the corporate social responsibility profile of the manufacturer, making the product more attractive to end-users who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of these advanced fluorescent dyes. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing workflows or product lines. The information covers aspects from synthesis scalability to application performance, ensuring a comprehensive overview of the value proposition.
Q: What are the photophysical advantages of carbonyl azetidine substitution?
A: The introduction of a carbonyl azetidine ring at the 4-position of the naphthalimide core significantly enhances fluorescence quantum efficiency and photostability compared to traditional azetidine substitutions, making it ideal for long-term imaging.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the process utilizes common solvents like DMSO and DMF and avoids expensive transition metal catalysts, facilitating straightforward purification and scalable manufacturing from kilograms to metric tons.
Q: What is the expected purity profile for imaging applications?
A: The method allows for silica gel column chromatography purification, ensuring high purity levels necessary for single-molecule detection and super-resolution fluorescence imaging without background interference.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl Azetidine Naphthalimide Dye Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies like CN112939936A into commercial reality, offering unparalleled expertise in the scale-up of fine chemical intermediates and specialty dyes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. For R&D directors and procurement managers, this means partnering with a supplier who not only understands the chemistry but also the critical quality attributes required for high-stakes applications like super-resolution imaging and biomolecule labeling.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized processes can reduce your overall material costs while maintaining the highest quality standards. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal requirements. Whether you need a reliable fluorescent dye supplier for immediate research needs or a long-term strategic partner for commercial production, NINGBO INNO PHARMCHEM is equipped to support your growth and innovation in the competitive landscape of electronic materials and pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
