Advanced Synthesis of Cefaloglycin Intermediates: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry constantly seeks robust synthetic routes for beta-lactam antibiotics, particularly third-generation cephalosporins like cefaloglycin. A pivotal advancement in this domain is detailed in patent CN101838278B, which discloses a highly efficient method for synthesizing a critical cefaloglycin intermediate. This technical breakthrough addresses long-standing challenges in the production of pharmaceutically acceptable intermediates by leveraging common organic solvents and mild reaction conditions to achieve superior stability. Unlike traditional methods that often suffer from complex operational procedures and harsh environments, this novel approach utilizes a series of well-controlled synthesis routes starting from Penicillin G potassium. The resulting intermediate demonstrates exceptional stability, a crucial factor for maintaining quality during the extended supply chains typical of global API manufacturing. For R&D directors and procurement specialists, understanding the nuances of this patent provides a strategic advantage in sourcing high-purity pharmaceutical intermediates that ensure consistent downstream processing and final drug efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of cefaloglycin and its precursors has been plagued by significant technical hurdles that impact both cost and reliability. Traditional intermediate products, often derived from cynnematin manufacturing procedures, are characterized by complicated synthetic pathways that are not easy to operate on a commercial scale. These legacy methods frequently require stringent reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management and safety protocols. Furthermore, the intermediates produced via these conventional routes often exhibit poor stability, leading to degradation during storage or transport, which directly translates to yield loss and increased costs for the supply chain. The complexity of purification in these older methods also introduces variability in the impurity profile, posing risks for regulatory compliance in strict markets like the US and EU. Consequently, manufacturers relying on these outdated technologies face continuous pressure to optimize their processes to remain competitive in the global fine chemical market.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a streamlined synthesis that fundamentally alters the production landscape for this key antibiotic intermediate. By employing a sequence of reactions under low-cost and mild conditions, the new route eliminates many of the operational bottlenecks associated with traditional cephalosporin synthesis. The process begins with the protection of Penicillin G potassium, followed by a controlled oxidation and a phosphite-mediated rearrangement, all of which are designed to maximize yield while minimizing side reactions. This approach not only simplifies the operational workflow but also ensures that the final intermediate possesses good stability, making it ideal for long-term storage and international shipping. For a reliable pharmaceutical intermediates supplier, adopting such a method means offering clients a product with a superior quality profile and a more predictable supply timeline. The use of common solvents like toluene and dichloromethane further enhances the feasibility of this route, as it aligns with standard industrial capabilities without requiring specialized, expensive equipment.
Mechanistic Insights into Phosphite-Mediated Ring Expansion
The core of this synthetic innovation lies in the sophisticated manipulation of the beta-lactam and thiazolidine rings, specifically through a phosphite-mediated rearrangement. The process initiates with the protection of the carboxylic acid group of Penicillin G using p-nitrobenzyl chloride, facilitated by phase transfer catalysts like Tetrabutyl ammonium bromide. This protection step is critical for preventing unwanted side reactions during the subsequent oxidation phase. Following protection, the sulfur atom in the thiazolidine ring is selectively oxidized to a sulfoxide using acetic anhydride and hydrogen peroxide. This sulfoxide formation is a prerequisite for the subsequent ring expansion, as it activates the sulfur for nucleophilic attack. The pivotal step involves the reaction of the sulfoxide with trimethyl phosphite in benzene under reflux conditions. This reaction triggers a rearrangement that expands the five-membered thiazolidine ring into a six-membered dihydrothiazine ring, effectively constructing the cephalosporin nucleus. The mechanism likely involves the formation of a phosphorane intermediate which facilitates the migration of the double bond and the expulsion of the sulfinyl group, resulting in the formation of the 6-benzyl-2-oxygen-3-nitrogen-(isopropyl alkene)-5-thia-dicyclo structure.
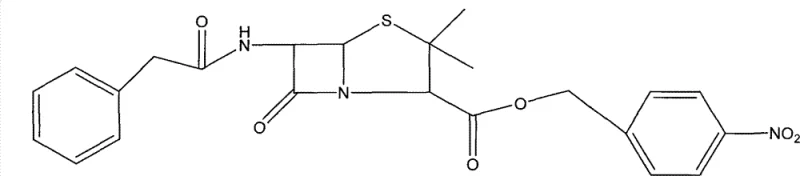
Following the ring expansion, the process includes an isomerization step using triethylamine in dichloromethane to shift the double bond to the thermodynamically more stable position, yielding the final 6-benzyl-2-oxygen-3-nitrogen-(2-isopropyl alkene)-5-thia-dicyclo (3,2,0) to p-Nitrobenzyl. This precise control over the double bond position is vital for the biological activity of the final cephalosporin antibiotic. The entire sequence is designed to minimize the formation of by-products, thereby simplifying the downstream purification process. For R&D teams, understanding this mechanism highlights the importance of reagent stoichiometry and temperature control, particularly during the reflux stage which lasts for 28-32 hours. The careful selection of solvents and the stepwise addition of reagents ensure that the reaction proceeds with high selectivity, preserving the integrity of the sensitive beta-lactam ring throughout the transformation. This mechanistic clarity allows for better process optimization and troubleshooting in a commercial manufacturing setting.
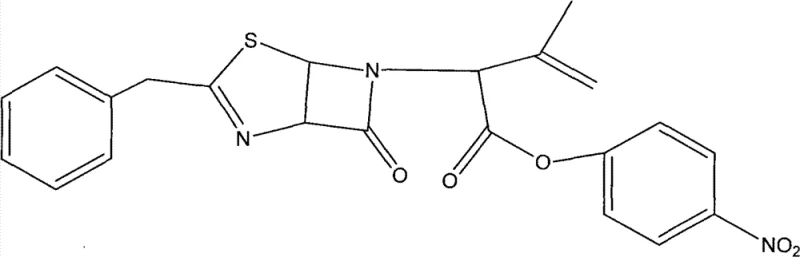
How to Synthesize Cefaloglycin Intermediate Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process is divided into four distinct stages: protection, oxidation, rearrangement, and isomerization. Each stage utilizes specific molar ratios and temperature ranges that have been empirically determined to maximize efficiency. For instance, the initial protection step requires heating to 55-65°C to ensure complete conversion of the Penicillin G potassium salt. The subsequent oxidation must be conducted at low temperatures, around 0°C, to prevent over-oxidation or degradation of the beta-lactam ring. The rearrangement step is the most time-consuming, requiring prolonged reflux in benzene, which demands robust reactor capabilities. Finally, the isomerization is a rapid process that must be quenched appropriately to isolate the desired isomer. Detailed standardized synthesis steps see the guide below.
- Protect Penicillin G potassium with p-Nitrobenzyl chloride using Tetrabutyl ammonium bromide and 2,5-dimethyl furan at 55-65°C.
- Oxidize the protected intermediate using acetic anhydride and hydrogen peroxide at low temperatures to form the sulfoxide derivative.
- Perform ring expansion and rearrangement using trimethyl phosphite in benzene under reflux, followed by isomerization with triethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant cost reduction in API intermediate manufacturing driven by the use of readily available and inexpensive raw materials. By utilizing common organic solvents and avoiding exotic catalysts, the overall material cost is drastically simplified, allowing for more competitive pricing structures in the global market. Furthermore, the mild reaction conditions reduce the energy consumption associated with heating and cooling, contributing to lower operational expenditures. The enhanced stability of the intermediate also translates to reduced waste and lower inventory losses, as the product can withstand longer storage periods without degradation. This reliability is crucial for maintaining a continuous supply chain, especially in the face of global logistical disruptions. Additionally, the simplified purification process reduces the time and resources required for quality control, accelerating the time-to-market for the final pharmaceutical product.
- Cost Reduction in Manufacturing: The elimination of complex and hazardous reagents in favor of common chemicals like toluene and acetic anhydride leads to substantial cost savings. By avoiding the need for specialized transition metal catalysts, manufacturers can bypass expensive procurement channels and reduce the dependency on volatile raw material markets. The mild conditions also imply lower energy costs, as the process does not require extreme heating or cryogenic cooling beyond standard industrial capabilities. This economic efficiency allows suppliers to offer more attractive pricing models to their pharmaceutical clients, fostering long-term partnerships based on value and reliability. Moreover, the high stability of the intermediate reduces the financial risk associated with product spoilage, ensuring that capital is not tied up in degrading inventory.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials such as Penicillin G potassium ensures a robust and resilient supply chain. Unlike routes that depend on niche precursors with limited suppliers, this method leverages commodities that are produced in vast quantities globally. This abundance minimizes the risk of supply shortages and price spikes, providing procurement teams with greater predictability in their planning. The simplified operational steps also mean that the manufacturing process is less prone to delays caused by technical failures or complex troubleshooting. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly reduced, enabling faster response to market demands. This reliability is a key differentiator for suppliers aiming to serve large-scale pharmaceutical companies that require consistent and timely deliveries.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that are easily transferable from pilot to production scale. The use of standard solvents and equipment means that existing manufacturing facilities can be adapted with minimal capital investment. From an environmental perspective, the avoidance of heavy metals and hazardous reagents simplifies waste treatment and disposal, aligning with increasingly stringent global environmental regulations. The reduced generation of toxic by-products lowers the burden on waste management systems and reduces the overall environmental footprint of the manufacturing process. This compliance not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the manufacturer, appealing to eco-conscious stakeholders and clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this cefaloglycin intermediate. These answers are derived directly from the technical specifications and beneficial effects described in the underlying patent data. They are intended to provide clarity on the process capabilities and the quality standards associated with this manufacturing route. Understanding these details is essential for making informed procurement decisions and ensuring that the intermediate meets the specific requirements of your downstream synthesis.
Q: What are the key stability advantages of this cefaloglycin intermediate synthesis method?
A: The method described in patent CN101838278B utilizes mild reaction conditions and specific protection groups that significantly enhance the stability of the intermediate compared to traditional cynnematin manufacturing procedures, reducing degradation during storage and transport.
Q: Does this process require expensive or hazardous catalysts?
A: No, the process relies on common organic solvents such as toluene, dichloromethane, and benzene, along with reagents like trimethyl phosphite and triethylamine, avoiding the need for rare transition metal catalysts often found in complex beta-lactam syntheses.
Q: How does this route impact the scalability of cephalosporin production?
A: By simplifying the operational steps and utilizing readily available raw materials like Penicillin G potassium, the route offers a more straightforward path for commercial scale-up, minimizing complex purification steps that often bottleneck large-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefaloglycin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our technical team has extensively analyzed the route described in CN101838278B and is fully equipped to execute this synthesis with precision and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are designed to handle the specific solvent systems and reaction conditions required for this process, maintaining stringent purity specifications throughout the manufacturing cycle. With our rigorous QC labs, we guarantee that every batch of cefaloglycin intermediate meets the highest industry standards, providing you with a solid foundation for your final API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a reliable source of complex intermediates that can accelerate your drug development timelines and enhance your market competitiveness. Let us handle the complexities of synthesis so you can focus on innovation and patient care.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
