Advanced Oxidation Process for High-Purity 4-Methyl-5-Formylthiazole Commercial Production
The pharmaceutical industry continuously demands more efficient pathways for critical synthons, and the production of 4-methyl-5-formylthiazole stands as a prime example of where process innovation drives commercial viability. Patent CN1307165C details a breakthrough methodology that transforms the manufacturing landscape for this essential heterocyclic compound, which serves as a pivotal building block in the synthesis of modern cephalosporin antibiotics and neuroprotective agents. Historically, the synthesis of this thiazole derivative has been plagued by low yields and complex purification challenges, creating bottlenecks for reliable pharmaceutical intermediates supplier networks globally. This new technical insight reveals a robust oxidation protocol that leverages a biphasic solvent system to achieve unprecedented purity levels, reportedly reaching 99.7% in specific embodiments. By shifting the paradigm from traditional high-temperature gas-phase reactions to a controlled liquid-phase oxidation, the technology offers a tangible pathway for cost reduction in API intermediate manufacturing. The strategic implementation of this patent allows production teams to bypass the formation of stubborn tar-like byproducts that have historically compromised batch consistency. As we delve into the technical specifics, it becomes clear that this process is not merely a laboratory curiosity but a scalable solution designed for the rigorous demands of commercial scale-up of complex pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements detailed in the patent data, the industrial synthesis of 4-methyl-5-formylthiazole relied heavily on methodologies that were inherently inefficient and environmentally burdensome. One of the earliest established routes involved the conversion of 4-methyl-5-methoxycarbonylthiazole, a process that suffered from a dismal overall yield of merely 23%, rendering it economically unfeasible for large-scale operations. Another historical approach attempted to utilize gas-phase hydrogenation at extreme temperatures reaching 315°C, which not only consumed excessive energy but also resulted in a conversion rate of only 74% with limited selectivity. Furthermore, alternative oxidation methods using pyridinium dichromate in homogeneous systems generated substantial amounts of solid waste, requiring six moles of oxidant per mole of substrate. These conventional techniques often resulted in the formation of difficult-to-separate tar-like mixtures, complicating the isolation of the target aldehyde and necessitating expensive purification steps. The reliance on such harsh conditions and inefficient stoichiometry created significant supply chain vulnerabilities, making it difficult to ensure reducing lead time for high-purity thiazole derivatives. Consequently, manufacturers faced persistent challenges in maintaining consistent quality while managing the high operational costs associated with waste disposal and energy consumption.
The Novel Approach
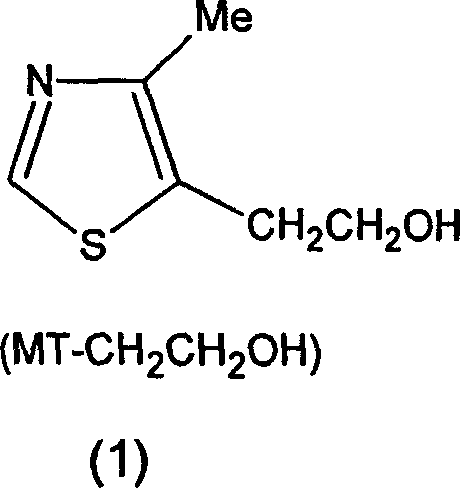
The innovative process described in the patent data introduces a fundamentally different strategy by utilizing 4-methyl-5-(2-hydroxyethyl)thiazole as the primary starting material, which is readily accessible through the degradation of Vitamin B1. This novel approach employs a two-phase solvent system comprising water and an immiscible organic solvent, such as benzene or dichloromethane, to facilitate the oxidation reaction under mild acidic conditions. By carefully selecting oxidants like chromium oxide or inorganic dichromates, the reaction can proceed at temperatures between 20°C and 50°C, drastically reducing the energy footprint compared to previous high-temperature methods. The key breakthrough lies in the phase transfer mechanism, where the generated 4-methyl-5-formylthiazole immediately partitions into the organic phase, effectively protecting it from over-oxidation to the corresponding carboxylic acid. This dynamic separation ensures that the reaction equilibrium favors the desired aldehyde product, leading to significantly improved yields and purity profiles without the formation of intractable tars. The ability to use stoichiometric amounts of oxidant, specifically 1.2 to 2.5 moles of CrO3 or 0.6 to 1.2 moles of dichromate, minimizes chemical waste and simplifies the downstream workup. This method represents a substantial leap forward in process chemistry, offering a reliable pharmaceutical intermediates supplier with a route that is both economically and environmentally superior.
Mechanistic Insights into Two-Phase Oxidation Catalysis
The core of this technological advancement rests on the sophisticated interplay between the aqueous and organic phases during the oxidation of the hydroxyethyl side chain. In this mechanism, the starting material 4-methyl-5-(2-hydroxyethyl)thiazole is initially dissolved or suspended within the acidic aqueous phase where the oxidant is also present. As the oxidation proceeds, the resulting 4-methyl-5-formylthiazole, being more lipophilic than its precursor, preferentially migrates into the organic solvent layer. This continuous extraction acts as a driving force for the reaction, pulling the equilibrium towards product formation while simultaneously removing the aldehyde from the oxidative environment of the aqueous phase. This spatial separation is critical because it prevents the aldehyde functionality from undergoing further oxidation to form 4-methylthiazole-5-carboxylic acid, a common side reaction that plagues homogeneous oxidation systems. The choice of organic solvent is paramount, with halogenated hydrocarbons like dichloromethane showing superior extraction efficiency for the target molecule. Furthermore, the acid catalyst, typically sulfuric acid, plays a dual role in activating the oxidant and maintaining the protonation state necessary for the reaction kinetics. Understanding this mechanistic nuance is essential for R&D teams aiming to replicate the high-purity 4-methyl-5-formylthiazole specifications required for sensitive pharmaceutical applications. The precise control of molar ratios and temperature ensures that the reaction proceeds cleanly, avoiding the generation of complex impurity profiles that could compromise downstream drug synthesis.
Impurity control is another critical aspect where this two-phase system excels, providing a robust framework for maintaining stringent purity specifications. In traditional homogeneous oxidations, the accumulation of byproducts often leads to the formation of polymeric tars that are notoriously difficult to remove, requiring extensive chromatographic purification. However, the biphasic nature of this novel process inherently limits the contact time between the product and the oxidizing agents, thereby suppressing the formation of these high-molecular-weight impurities. The patent data indicates that by maintaining the reaction temperature within the narrow window of 25°C to 30°C, the formation of the carboxylic acid byproduct is virtually eliminated. Additionally, the use of specific oxidants like sodium dichromate or potassium dichromate allows for fine-tuning of the oxidation potential, ensuring that only the primary alcohol is converted to the aldehyde without affecting the sensitive thiazole ring. The resulting crude product often requires minimal purification, with some examples showing direct isolation of high-purity material after simple solvent removal and drying. This level of control over the impurity spectrum is vital for regulatory compliance in the pharmaceutical sector, where trace impurities can have significant implications for drug safety. By minimizing the generation of hazardous waste and simplifying the purification train, this process aligns with modern green chemistry principles while delivering commercial-grade quality.
How to Synthesize 4-Methyl-5-Formylthiazole Efficiently
Implementing this synthesis route requires careful attention to the preparation of the biphasic reaction mixture and the controlled addition of reagents to ensure optimal performance. The process begins with the preparation of the oxidant solution in water, acidified with sulfuric acid to the specified molar excess, which is then added gradually to the organic solution containing the hydroxyethyl thiazole precursor. Maintaining the temperature within the recommended range is crucial to prevent thermal runaway or the formation of side products, necessitating the use of efficient cooling systems during the exothermic oxidation phase. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Prepare a two-phase solvent system consisting of water and an immiscible organic solvent such as benzene or dichloromethane.
- Dissolve 4-methyl-5-(2-hydroxyethyl)thiazole in the organic phase and add an acid catalyst such as sulfuric acid to the aqueous phase.
- Add chromium oxide or inorganic dichromate oxidant gradually while maintaining temperature between 20°C and 50°C to prevent over-oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this oxidation technology offers profound benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies for critical intermediates. The shift to a milder, liquid-phase process eliminates the need for specialized high-temperature reactors and the associated energy infrastructure, leading to substantial cost savings in manufacturing overhead. By utilizing readily available starting materials derived from Vitamin B1 degradation, the supply chain becomes more resilient against raw material shortages that often plague specialty chemical markets. The reduction in solid waste generation and the simplification of the purification process translate directly into lower disposal costs and reduced environmental compliance burdens. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials, improving the overall material efficiency of the production line. These factors combine to create a more predictable and cost-effective supply model, enabling partners to secure high-purity 4-methyl-5-formylthiazole with greater confidence. The ability to scale this process from laboratory to commercial production without significant re-engineering ensures that supply continuity can be maintained even as demand fluctuates. Ultimately, this technology empowers organizations to achieve significant cost reduction in API intermediate manufacturing while adhering to strict quality and sustainability standards.
- Cost Reduction in Manufacturing: The elimination of high-temperature gas-phase reactors and the reduction in oxidant stoichiometry drastically lower energy and raw material expenses. By avoiding the formation of tar-like byproducts, the need for expensive chromatographic purification is minimized, further reducing operational costs. The use of common inorganic oxidants and acids ensures that reagent costs remain stable and predictable, avoiding the volatility associated with specialized catalysts. This streamlined process flow allows for higher throughput in existing facilities, maximizing asset utilization and driving down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: Sourcing 4-methyl-5-(2-hydroxyethyl)thiazole is more stable compared to specialized esters, as it can be derived from abundant Vitamin B1 sources. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supply or environmental conditions, ensuring consistent output. The simplified workup procedure reduces the turnaround time between batches, allowing for more frequent production runs and better inventory management. This reliability is crucial for maintaining the continuity of supply for downstream pharmaceutical manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The two-phase system is inherently scalable, as the mass transfer dynamics remain consistent when moving from pilot to commercial scale reactors. The significant reduction in solid waste and the use of recyclable organic solvents align with increasingly stringent environmental regulations globally. By minimizing the generation of hazardous byproducts, the facility reduces its environmental footprint and the associated regulatory reporting burdens. This sustainable approach not only mitigates risk but also enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation process, based on the detailed patent specifications. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of the two-phase oxidation system for MT-CHO synthesis?
A: The two-phase system allows the product to extract into the organic layer immediately, preventing further oxidation to the carboxylic acid byproduct and ensuring high purity.
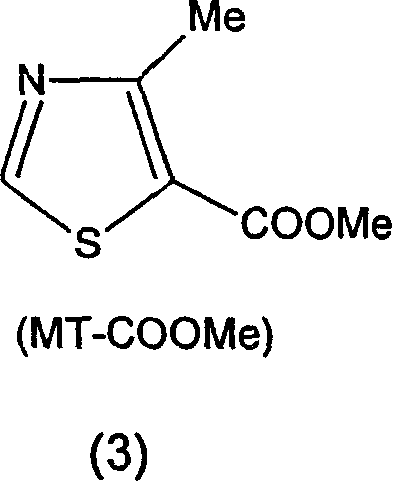
Q: Why is 4-methyl-5-(2-hydroxyethyl)thiazole preferred over MT-COOMe as a starting material?
A: MT-CH2CH2OH is readily available from Vitamin B1 degradation and avoids the harsh high-temperature gas-phase hydrogenation required for MT-COOMe, reducing energy costs.
Q: How does this process impact environmental compliance in pharmaceutical manufacturing?
A: By optimizing oxidant stoichiometry and avoiding tar-like mixtures, the process significantly reduces solid waste generation and simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-5-Formylthiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving pharmaceuticals, and we are committed to delivering excellence in every batch. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of 4-methyl-5-formylthiazole meets the exacting standards required for API synthesis. Our commitment to quality is matched by our dedication to process safety and environmental stewardship, making us a partner of choice for global pharmaceutical companies. By leveraging advanced oxidation technologies like the one described in CN1307165C, we continue to push the boundaries of what is possible in fine chemical manufacturing.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements and supply chain goals. Request a Customized Cost-Saving Analysis to understand how our optimized processes can improve your bottom line without compromising on quality. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to transparency and technical excellence. Let us help you secure a reliable supply of this critical intermediate, ensuring your drug development timelines remain on track.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
