Scalable Electrochemical Synthesis of Tetraarylhydrazines for Advanced Pharmaceutical Applications
Scalable Electrochemical Synthesis of Tetraarylhydrazines for Advanced Pharmaceutical Applications
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN111235599A, which discloses a novel electrochemical approach for the synthesis of tetraarylhydrazine compounds. These unique molecular scaffolds are not merely academic curiosities; they serve as the critical structural backbone for complex natural products such as dixiamycin A and dixiamycin B, and function as potent electrocatalysts in energy conversion systems. 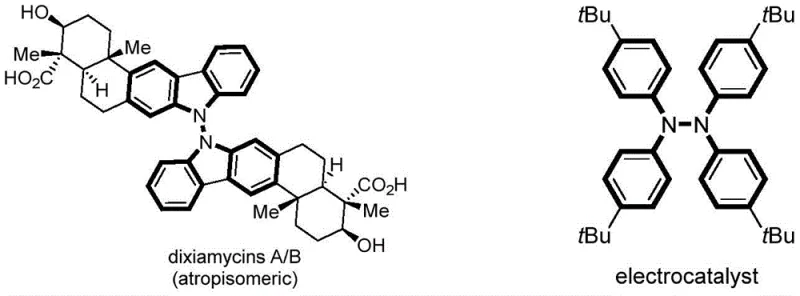 The traditional reliance on stoichiometric oxidants and transition metals poses substantial challenges regarding toxicity and waste management. This patent introduces a metal-free, oxidant-free electrochemical dehydrogenation strategy that operates under mild conditions, offering a compelling solution for the production of high-purity pharmaceutical intermediates and specialty chemicals.
The traditional reliance on stoichiometric oxidants and transition metals poses substantial challenges regarding toxicity and waste management. This patent introduces a metal-free, oxidant-free electrochemical dehydrogenation strategy that operates under mild conditions, offering a compelling solution for the production of high-purity pharmaceutical intermediates and specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the N-N bond in tetraarylhydrazines has relied heavily on classical chemical oxidation protocols that are increasingly viewed as unsustainable in modern industrial settings. Prominent literature precedents, such as those by Yu Wenquan, utilize elemental iodine as a stoichiometric oxidant to drive the coupling of secondary amines, a process that generates significant amounts of iodine-containing waste streams requiring complex disposal procedures. Furthermore, other established methods employ transition metal catalysts, including various iron complexes or copper systems like CuBr·Me2S combined with amine ligands. While effective on a laboratory scale, these metal-catalyzed pathways introduce severe complications for large-scale manufacturing, primarily due to the stringent regulatory limits on residual heavy metals in active pharmaceutical ingredients. The removal of trace metal contaminants often necessitates additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which drastically erode overall process efficiency and increase the cost of goods sold.
The Novel Approach
In stark contrast to these legacy techniques, the electrochemical method described in CN111235599A leverages electricity as a traceless reagent to drive the oxidative dimerization of diarylamines. By utilizing an undivided electrolytic cell equipped with inert platinum electrodes, the process achieves direct anodic oxidation of the substrate without the need for external chemical oxidants. The system employs tetrabutylammonium iodide not merely as a supporting electrolyte to ensure conductivity, but critically as a redox mediator that facilitates the electron transfer process. This dual functionality allows the reaction to proceed under neutral conditions, avoiding the use of harsh acids or bases that could degrade sensitive functional groups. The result is a streamlined synthetic route that minimizes auxiliary reagents, reduces the E-factor (environmental factor), and aligns perfectly with the principles of green chemistry, making it an ideal candidate for the commercial scale-up of complex organic intermediates.
Mechanistic Insights into Electrochemical Dehydrogenative Coupling
The core of this transformative technology lies in the precise control of electrochemical potential to generate reactive radical cation intermediates from the diarylamine starting materials. Upon application of a constant current, the diarylamine undergoes a single-electron oxidation at the anode surface to form a radical cation species. 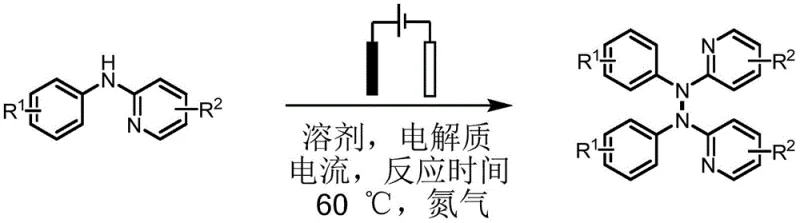 These highly reactive intermediates subsequently couple to form the hydrazine N-N bond, followed by a second oxidation and deprotonation sequence to yield the final tetraarylhydrazine product. The presence of iodide ions from the tetrabutylammonium iodide electrolyte plays a crucial mediating role, potentially forming transient iodine species that lower the overpotential required for substrate oxidation, thereby enhancing energy efficiency. This mechanism avoids the high-energy barriers associated with direct thermal oxidation, allowing the reaction to proceed at a moderate temperature of 60°C, which preserves the integrity of thermally labile substituents.
These highly reactive intermediates subsequently couple to form the hydrazine N-N bond, followed by a second oxidation and deprotonation sequence to yield the final tetraarylhydrazine product. The presence of iodide ions from the tetrabutylammonium iodide electrolyte plays a crucial mediating role, potentially forming transient iodine species that lower the overpotential required for substrate oxidation, thereby enhancing energy efficiency. This mechanism avoids the high-energy barriers associated with direct thermal oxidation, allowing the reaction to proceed at a moderate temperature of 60°C, which preserves the integrity of thermally labile substituents.
From an impurity control perspective, the electrochemical method offers superior selectivity compared to chemical oxidants which often suffer from over-oxidation issues. The constant current mode ensures that the oxidation potential is maintained within a narrow window sufficient to drive the desired coupling but below the threshold that would degrade the product or the solvent. The specific solvent system, comprising a 14:1 volume ratio of acetonitrile to methanol, is engineered to optimize proton management; methanol acts as a proton acceptor at the cathode to prevent the accumulation of acidity that could lead to side reactions, while acetonitrile provides the necessary dielectric environment. This balanced reaction environment minimizes the formation of by-products such as C-C coupled dimers or over-oxidized quinone-imine species, resulting in a crude product profile that is significantly cleaner and easier to purify via standard column chromatography.
How to Synthesize Tetraarylhydrazine Compounds Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and parameter optimization to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture in an undivided cell, where the precise molar ratio of electrolyte to substrate is critical for maintaining consistent conductivity throughout the reaction duration. Operators must ensure that the nitrogen atmosphere is rigorously maintained to exclude oxygen, which could interfere with the radical intermediates or lead to unwanted oxidative side pathways. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring that R&D teams can replicate the high yields reported in the literature.
- Prepare the electrolytic cell by adding diarylamine compounds, acetonitrile/methanol solvent mixture, and tetrabutylammonium iodide electrolyte.
- Insert platinum plate electrodes, purge with nitrogen, and apply constant current (1.5-10 mA) at 60°C.
- After reaction completion, purify the crude tetraarylhydrazine product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis route presents a strategic opportunity to optimize the cost structure and reliability of the supply chain for nitrogen-containing heterocycles. By shifting away from dependency on volatile transition metal catalysts and stoichiometric oxidants, manufacturers can insulate their production costs from the price fluctuations of precious metals like palladium or copper. The elimination of these expensive reagents directly translates to a reduction in raw material expenditure, while the simplified workup procedure reduces the consumption of solvents and silica gel during purification. Furthermore, the absence of heavy metals removes the need for costly analytical testing and specialized metal-scavenging steps, accelerating the release of batches for downstream processing and significantly shortening the overall manufacturing cycle time.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the drastic simplification of the reagent profile. By replacing expensive metal catalysts and chemical oxidants with electricity and a reusable electrolyte, the variable cost per kilogram of product is substantially lowered. Additionally, the high atom economy of the dehydrogenative coupling means that less raw material is wasted in the form of stoichiometric by-products, maximizing the yield from every gram of starting diarylamine. This efficiency gain allows for more competitive pricing strategies in the global market for pharmaceutical intermediates without compromising margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity-grade starting materials and reagents. Diarylamines and tetrabutylammonium iodide are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks that often plague specialized catalyst supply chains. The robustness of the electrochemical method against variations in substrate electronics—tolerating both electron-rich and electron-poor groups—means that a single platform technology can be applied to synthesize a diverse range of derivatives, simplifying inventory management and reducing the need for multiple specialized production lines.
- Scalability and Environmental Compliance: Scaling electrochemical processes is inherently safer and more linear than scaling exothermic chemical oxidations, as the reaction rate is controlled by current rather than heat dissipation limits. This facilitates a smoother transition from pilot plant to full commercial production, reducing the time-to-market for new drug candidates. Moreover, the green nature of the process, characterized by the absence of toxic heavy metal waste and reduced solvent usage, ensures compliance with increasingly stringent environmental regulations, mitigating the risk of regulatory shutdowns and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and applicability.
Q: What are the advantages of electrochemical synthesis over traditional chemical oxidation for tetraarylhydrazines?
A: Electrochemical synthesis eliminates the need for stoichiometric chemical oxidants like iodine or expensive transition metal catalysts such as copper or iron. This results in a cleaner reaction profile, reduced toxic waste generation, and simplified downstream purification processes, significantly lowering the environmental footprint.
Q: Why is the mixed solvent system of acetonitrile and methanol critical for this reaction?
A: The specific ratio of acetonitrile to methanol is essential for balancing conductivity and proton scavenging. Methanol facilitates the partial reduction of electrons at the cathode, preventing side reactions, while acetonitrile ensures adequate solubility and conductivity. Using either solvent alone leads to drastically reduced yields or complete reaction failure.
Q: Can this electrochemical method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate scope, successfully accommodating both electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing groups such as fluorine, chlorine, and bromine. This versatility makes it highly suitable for synthesizing diverse libraries of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraarylhydrazine Compounds Supplier
As the demand for complex nitrogenous scaffolds continues to rise in the pharmaceutical and agrochemical sectors, partnering with a technically proficient manufacturer is essential for securing a stable supply of high-quality intermediates. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. Our facility is equipped with state-of-the-art electrochemical reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetraarylhydrazine compounds meets the exacting standards required for GMP manufacturing environments.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this green electrochemical route into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your target molecules. Please contact our technical procurement team today to discuss your project needs, request specific COA data for our catalog compounds, or initiate a route feasibility assessment for your proprietary intermediates.
