Advanced Aqueous Synthesis of 4-Hydroxy-5-Ethoxyaniline for Commercial Veterinary Drug Production
The pharmaceutical and veterinary industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical intermediates. Patent CN101948398A introduces a groundbreaking methodology for the preparation of 4-hydroxy-5-ethoxyaniline, a pivotal building block in the production of Decoquinate, a widely used anticoccidial agent. This novel approach fundamentally reengineers the traditional synthetic landscape by replacing hazardous organic solvents and expensive noble metal catalysts with a benign aqueous system and economical reducing agents. For R&D directors and procurement specialists alike, this shift represents not merely a chemical optimization but a strategic supply chain enhancement. By leveraging diazotization, azo coupling, and sodium hydrosulfite reduction, the process achieves a remarkable total yield of 86% while maintaining an HPLC purity exceeding 95%. This report analyzes the technical merits and commercial implications of adopting this superior manufacturing route for high-volume veterinary drug intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-hydroxy-5-ethoxyaniline has been plagued by inefficient multi-step sequences and reliance on environmentally taxing reagents. One prevalent conventional method initiates with pyrocatechol, undergoing diethylation, nitration, de-ethylation, and finally, palladium-carbon catalytic hydrogenation. As illustrated in the reaction scheme below, this pathway is fraught with operational complexities, including the use of glacial acetic acid and concentrated nitric acid, which generate substantial acidic wastewater and impose severe corrosion requirements on reactor equipment.
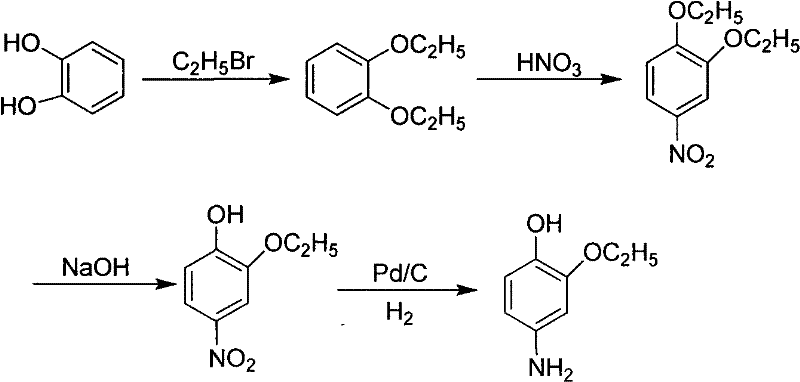
Furthermore, the cumulative yield of this traditional pyrocatechol route hovers around a mere 60%, indicating significant loss of raw materials and increased cost per kilogram of final product. Another existing method utilizes aniline via diazotization and coupling but relies on Pd/C hydrogenation for the final reduction step. This dependence on precious metals introduces volatility in production costs due to fluctuating palladium prices and necessitates rigorous purification steps to ensure residual metal levels meet strict pharmaceutical standards. The separation difficulties associated with these older methods often result in lower overall recovery rates, sometimes dropping as low as 48.3%, rendering them economically unviable for large-scale commercial operations seeking margin optimization.
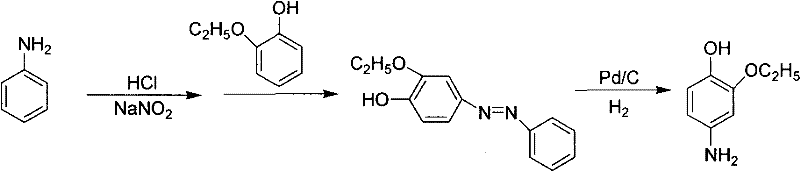
The Novel Approach
In stark contrast, the novel methodology detailed in CN101948398A circumvents these bottlenecks by employing a streamlined three-step sequence that prioritizes atom economy and operational safety. The process begins with the diazotization of aniline, followed by coupling with 2-ethoxyphenol, and concludes with a reduction using sodium hydrosulfite (insurance powder) in an aqueous medium. This strategic substitution of catalytic hydrogenation with chemical reduction using sodium hydrosulfite eliminates the capital expenditure associated with high-pressure hydrogenation reactors and the recurring cost of noble metal catalysts. Moreover, the reaction proceeds in water, avoiding the use of volatile organic solvents during the critical reduction phase, which simplifies solvent recovery and significantly lowers the environmental footprint. The intermediates are processed without isolation and purification between steps, a telescoping strategy that reduces labor hours, minimizes material handling losses, and accelerates the overall production cycle time.
Mechanistic Insights into Azo Coupling and Hydrosulfite Reduction
The core of this technological advancement lies in the precise control of the azo coupling and subsequent reduction mechanisms. The process generates a phenyl diazonium salt from aniline under strictly controlled low-temperature conditions (-5 to 0°C) to prevent decomposition. This electrophilic species then couples with 2-ethoxyphenol under alkaline conditions (pH 8-9) to form the stable azo compound intermediate. The structural integrity of this intermediate is crucial, as shown in the diagram below, where the azo linkage serves as the precursor to the final amine functionality. Maintaining the pH within the optimal range during coupling ensures regioselectivity and prevents the formation of undesired diazo-amino byproducts, thereby safeguarding the purity profile of the downstream product.

Following the coupling, the reduction step utilizes sodium hydrosulfite as the electron donor in an aqueous alkaline environment. The mechanism involves the cleavage of the nitrogen-nitrogen double bond of the azo group to yield the primary amine. By conducting this reaction in water at temperatures between 50°C and 90°C, the process ensures high solubility of the inorganic reagents and efficient heat transfer, which is critical for exothermic reduction reactions. The addition of alkali, such as sodium hydroxide or potassium carbonate, maintains the system pH between 8 and 14, which is essential for stabilizing the reducing agent and preventing the formation of sulfurous acid byproducts that could degrade the product. This aqueous-based mechanism not only enhances safety by removing flammable organic solvents but also facilitates the recycling of unreacted aniline from the filtrate, further boosting the overall mass balance and economic efficiency of the synthesis.
How to Synthesize 4-Hydroxy-5-Ethoxyaniline Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize the reported 86% yield. The process is designed to be robust, allowing for the direct use of crude intermediates without intermediate purification, which significantly simplifies the operational workflow for plant managers. The standardized protocol involves preparing the diazonium salt, coupling it with the phenol derivative in a methanol-water mix, and finally reducing the resulting azo dye with insurance powder in a heated aqueous alkaline solution. Detailed standard operating procedures regarding specific molar equivalents, addition rates, and workup protocols are essential for reproducibility.
- Perform diazotization of aniline using sodium nitrite and hydrochloric acid at low temperatures (-5 to 0°C) to form the diazonium salt intermediate.
- Couple the diazonium salt with 2-ethoxyphenol in a methanol-water mixture under alkaline conditions (pH 8-9) to form the azo compound.
- Reduce the azo compound using sodium hydrosulfite (insurance powder) in an aqueous alkaline solution at 50-90°C to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers profound strategic advantages beyond mere technical feasibility. The elimination of palladium catalysts removes a significant variable from the raw material cost structure, insulating the production budget from the volatility of precious metal markets. Furthermore, the shift to an aqueous reduction system drastically simplifies waste management protocols, reducing the costs associated with hazardous waste disposal and environmental compliance auditing. The ability to recycle solvents and raw materials like aniline directly within the process loop enhances the overall material utilization rate, leading to substantial cost savings in raw material procurement over the lifecycle of the product.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with commodity chemicals like sodium hydrosulfite results in a direct and significant reduction in variable production costs. By avoiding the need for specialized high-pressure hydrogenation equipment and the associated maintenance and safety certifications, capital expenditure requirements are lowered. Additionally, the telescoped nature of the synthesis, where intermediates are not isolated, reduces energy consumption related to drying and solvent exchange, further driving down the cost of goods sold (COGS) for this veterinary drug intermediate.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically aniline and 2-ethoxyphenol, are commodity chemicals with robust global supply chains, minimizing the risk of supply disruptions compared to specialized catalysts. The simplified processing steps reduce the overall lead time for manufacturing batches, allowing for more responsive inventory management and faster fulfillment of customer orders. The high yield and purity consistency ensure that fewer batches are rejected due to quality failures, stabilizing the available supply volume for downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: Conducting the reduction in an aqueous medium inherently improves the safety profile of the process, making it easier to scale from pilot plants to multi-ton commercial reactors without significant engineering hurdles. The reduction in organic solvent usage and the minimization of three-waste discharge align with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory tightening. This green chemistry approach enhances the corporate sustainability profile, which is becoming a critical factor in vendor selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on yield expectations, impurity profiles, and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: What is the primary advantage of using sodium hydrosulfite over palladium catalysts?
A: Using sodium hydrosulfite eliminates the need for expensive noble metal catalysts like Pd/C, significantly reducing raw material costs and removing the complex step of heavy metal residue removal required for pharmaceutical compliance.
Q: How does this method improve environmental compliance?
A: The process utilizes water as the primary reaction medium for the reduction step and allows for the recycling of aniline and solvents, drastically reducing the discharge of organic waste and hazardous three-waste emissions.
Q: What purity levels can be achieved with this synthetic route?
A: The optimized process consistently achieves an HPLC content of over 95% with a total cumulative yield reaching approximately 86%, ensuring high quality suitable for downstream veterinary drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-5-Ethoxyaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel aqueous reduction method are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-hydroxy-5-ethoxyaniline meets the exacting standards required for veterinary drug synthesis. We are committed to delivering high-quality intermediates that support your downstream manufacturing efficiency.
We invite you to collaborate with us to optimize your supply chain for Decoquinate and related veterinary pharmaceuticals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
