Advanced Manufacturing of N-Substituted Pyrazole Compounds for Global Agrochemical Supply Chains
Advanced Manufacturing of N-Substituted Pyrazole Compounds for Global Agrochemical Supply Chains
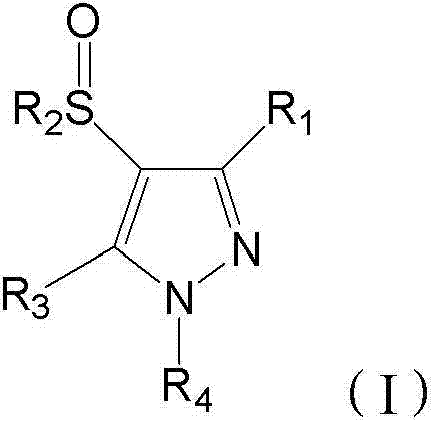
The global demand for high-performance insecticides continues to drive innovation in the synthesis of heterocyclic intermediates, particularly within the pyrazole class. Patent CN103044332A introduces a transformative methodology for preparing N-substituted sulfinyl-pyrazole derivatives, specifically targeting compounds with potent insecticidal activity such as 5-amino-3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)-4-trifluoromethyl sulphinyl pyrazole. This technical breakthrough addresses long-standing challenges in industrial organic synthesis by eliminating the reliance on highly corrosive and expensive reagents like trifluoroacetic acid (TFA). For R&D directors and procurement specialists, this patent represents a pivotal shift towards safer, more economically viable manufacturing protocols that do not compromise on yield or purity. The core innovation lies in a catalytic oxidation system that utilizes hydrogen peroxide as a sole oxidant in the absence of trifluoroperacetic acid, fundamentally altering the risk profile and cost structure of producing these valuable agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of sulfinyl-pyrazole derivatives has been plagued by significant safety and equipment integrity issues associated with traditional oxidation protocols. Prior art, such as EP 0295117 A and WO 01/30760, typically relies on the use of 3-chloroperoxybenzoic acid (mCPBA) or the in situ generation of trifluoroperacetic acid via the reaction of hydrogen peroxide with trifluoroacetic acid (TFA). While these methods can achieve the desired chemical transformation, they introduce severe operational hazards. The formation of trifluoroperacetic acid often leads to the generation of hydrogen fluoride (HF) as a decomposition byproduct, which is notoriously corrosive to glass-lined steel reactors commonly used in fine chemical manufacturing. This corrosion necessitates frequent equipment replacement and rigorous safety monitoring, driving up capital expenditure and operational downtime. Furthermore, the use of mCPBA involves handling shock-sensitive peroxides on a large scale, posing inherent explosion risks that complicate logistics and storage for supply chain managers.
The Novel Approach
The methodology disclosed in the patent data offers a robust alternative by decoupling the oxidation step from the need for corrosive carboxylic acids. The novel approach employs a catalytic system comprising sodium tungstate and hydrochloric acid to activate hydrogen peroxide, enabling the selective oxidation of the sulfide precursor to the sulfoxide without generating HF. This process operates effectively at mild temperatures, typically between 0°C and 10°C, which enhances thermal safety and reduces energy consumption compared to high-temperature alternatives. By utilizing hydrogen peroxide as the sole oxidant, the process eliminates the introduction of fluorinated organic waste streams, simplifying effluent treatment and reducing environmental compliance costs. This strategic shift not only preserves reactor integrity but also streamlines the purification workflow, as the absence of TFA removes the need for complex neutralization and removal steps associated with strong fluorinated acids.
Mechanistic Insights into Tungsten-Catalyzed Oxidation
The heart of this technological advancement lies in the specific interaction between the tungsten catalyst and the peroxide oxidant. In the absence of trifluoroperacetic acid, the oxidation of the thioether group (R2-S-) to the sulfinyl group (R2-S(=O)-) requires a highly active oxygen transfer agent. The patent specifies that a mixture of sodium tungstate and hydrochloric acid generates a peroxotungstate species in situ, which acts as the active oxygen donor. This mechanistic pathway allows for precise control over the oxidation state, preventing the common issue of over-oxidation to the sulfone (R2-SO2-), which is a difficult-to-remove impurity in pyrazole chemistry. The reaction kinetics are carefully managed by maintaining the temperature between 5°C and 8°C, ensuring that the rate of oxygen transfer matches the stability of the intermediate species. This level of control is critical for maintaining high assay values in the final product, directly impacting the efficacy of the downstream agrochemical formulation.
Furthermore, the impurity profile of this catalytic method is significantly cleaner than acid-mediated oxidations. Traditional methods often result in chlorination side reactions or ring degradation due to the harsh acidic environment. In contrast, the buffered acidic conditions provided by the tungsten/hydrochloric acid system are mild enough to preserve the sensitive cyano and amino functionalities on the pyrazole ring. The process described involves reacting the sulfenyl intermediate with 1.1 to 1.7 moles of hydrogen peroxide, a stoichiometric ratio optimized to drive conversion to completion while minimizing excess oxidant that could lead to byproduct formation. Post-reaction workup involves simple neutralization with sodium bicarbonate, avoiding the complex extraction procedures required to remove residual TFA, thereby enhancing the overall mass balance and yield of the manufacturing process.
How to Synthesize 5-Amino-3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)-4-trifluoromethyl sulphinyl pyrazole Efficiently
The synthesis of this specific high-value intermediate follows a logical two-stage sequence designed for maximum throughput and safety. The first stage involves the sulfenylation of the pyrazole precursor using trifluoromethanesulfenyl chloride, followed by the critical oxidation step detailed in the patent examples. The process is scalable from kilogram to multi-ton quantities, utilizing standard stainless steel or glass-lined equipment without the fear of HF corrosion. Operators must strictly adhere to the temperature constraints during the exothermic addition of hydrogen peroxide to ensure safety and selectivity. The detailed standardized synthesis steps, including specific solvent volumes, addition rates, and workup procedures, are outlined in the technical guide below for immediate implementation by process chemists.
- React the N-substituted pyrazole precursor with trifluoromethanesulfenyl chloride (CF3SCl) in an organic solvent like methylene dichloride at controlled temperatures (10°C to 30°C) to form the sulfenyl intermediate.
- Oxidize the sulfenyl intermediate using hydrogen peroxide (30% aqueous solution) in the presence of a sodium tungstate and hydrochloric acid catalyst system at low temperatures (5°C to 8°C).
- Purify the crude sulfinyl-pyrazole product via crystallization or washing processes to achieve high purity specifications suitable for agrochemical applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this non-corrosive oxidation technology translates into tangible operational efficiencies and risk mitigation. The elimination of trifluoroacetic acid and m-chloroperoxybenzoic acid removes two of the most costly and logistically challenging reagents from the bill of materials. This shift not only lowers the direct cost of goods sold but also reduces the regulatory burden associated with transporting and storing hazardous peroxides and corrosive acids. The simplified waste stream, devoid of fluorinated organic acids, allows for more straightforward effluent treatment, aligning with increasingly stringent environmental regulations in major manufacturing hubs. Consequently, this process offers a sustainable pathway for the commercial scale-up of complex agrochemical intermediates, ensuring long-term supply continuity without the threat of equipment failure due to corrosion.
- Cost Reduction in Manufacturing: The substitution of expensive oxidants like mCPBA with commodity-grade hydrogen peroxide results in substantial raw material savings. Additionally, the removal of TFA eliminates the need for specialized corrosion-resistant alloys or frequent reactor re-lining, significantly lowering capital depreciation costs. The simplified workup procedure reduces solvent consumption and labor hours required for purification, further driving down the overall production cost per kilogram. These cumulative efficiencies allow for a more competitive pricing structure in the global agrochemical market.
- Enhanced Supply Chain Reliability: By relying on widely available reagents such as hydrogen peroxide and sodium tungstate, the manufacturing process becomes less susceptible to supply disruptions often seen with specialty peracids. The robustness of the reaction conditions, operating at atmospheric pressure and mild temperatures, ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability is crucial for maintaining just-in-time delivery schedules for downstream formulators who depend on a steady flow of high-purity intermediates.
- Scalability and Environmental Compliance: The process is inherently safer for large-scale operations due to the absence of shock-sensitive peroxides and corrosive HF gas generation. This safety profile facilitates easier regulatory approval for plant expansions and new facility construction. Moreover, the reduced generation of hazardous waste simplifies compliance with environmental protection standards, minimizing the risk of fines and shutdowns. The ability to scale this chemistry from pilot plants to 100 MT annual production capacities without fundamental process changes makes it an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of switching to this catalytic oxidation method. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams negotiating supply agreements based on improved manufacturing economics.
Q: Why is the avoidance of trifluoroacetic acid (TFA) critical in this synthesis?
A: Conventional methods utilizing TFA and hydrogen peroxide generate trifluoroperacetic acid in situ, which often leads to the formation of highly corrosive hydrogen fluoride (HF). This causes severe etching of glass-lined industrial reactors, increasing maintenance costs and safety risks. The novel process eliminates TFA entirely.
Q: What represents the primary cost-saving advantage of this catalytic oxidation method?
A: By replacing expensive and hazardous oxidants like m-chloroperoxybenzoic acid (mCPBA) or corrosive TFA systems with commercially available hydrogen peroxide and a reusable tungsten catalyst system, the process significantly reduces raw material costs and waste disposal expenses associated with hazardous byproducts.
Q: How does the temperature control impact the selectivity of the oxidation step?
A: Maintaining the reaction temperature between 5°C and 8°C is crucial to prevent over-oxidation to the sulfone derivative. This precise thermal control ensures high selectivity for the desired sulfinyl group while minimizing impurity formation, thereby simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Amino-3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)-4-trifluoromethyl sulphinyl pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the agrochemical sector. Our CDMO capabilities are perfectly aligned with the requirements of this patent, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We have invested heavily in state-of-the-art reactor systems that can safely handle the specific temperature and mixing requirements of the tungsten-catalyzed oxidation process. Our rigorous QC labs ensure that every batch meets stringent purity specifications, guaranteeing that the sulfinyl-pyrazole intermediates supplied to our partners are free from corrosive residues and over-oxidized impurities.
We invite global agrochemical manufacturers to collaborate with us to optimize their supply chains using this superior technology. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us help you transition to a safer, more efficient, and cost-effective manufacturing strategy that secures your position in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
