Scalable Manufacturing of High-Purity Taxane Derivatives via Convergent Esterification
Introduction to Advanced Taxane Synthesis Technologies
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for more efficient and scalable processes for critical active pharmaceutical ingredients. Patent CN101379046A introduces a groundbreaking methodology for the preparation of taxane derivatives, specifically addressing the longstanding challenges associated with the semi-synthesis of compounds like Paclitaxel, Docetaxel, and TPI287. This intellectual property outlines a novel convergent synthesis strategy that leverages cyclically protected side chain acids to esterify the sterically hindered 13-hydroxyl group of the taxane nucleus. By shifting away from traditional beta-lactam acylation methods, this technology offers a pathway to higher chemical purity and improved overall yields, which are paramount metrics for any reliable pharmaceutical intermediates supplier aiming to support global API production. The strategic implementation of mixed anhydride activation allows for milder reaction conditions, preserving the delicate stereochemistry of the taxane core while facilitating robust commercial scale-up.
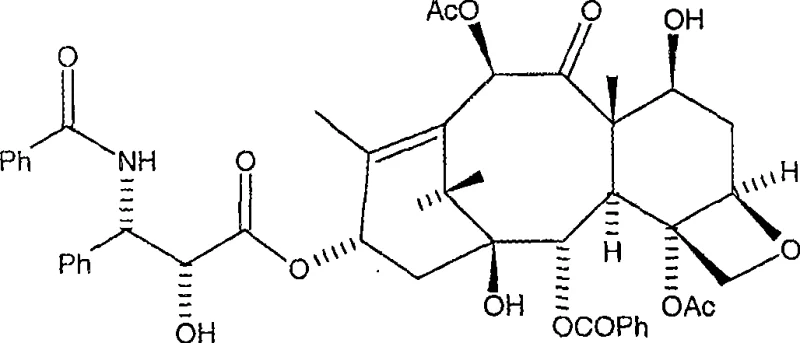
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of major taxane anticancer agents has relied heavily on the acylation of 10-deacetylbaccatin III or Baccatin III using beta-lactam intermediates. While effective, these conventional routes suffer from significant inherent drawbacks that impact manufacturing efficiency and cost structures. The C-13 hydroxyl group, which serves as the attachment point for the essential side chain, is located within a deeply concave and sterically hindered region of the hemispherical taxane backbone. This spatial constraint makes direct esterification or coupling exceptionally difficult, often requiring harsh reagents or forcing conditions that can lead to unwanted epimerization at the C-13 position. Furthermore, traditional methods frequently necessitate complex protection and deprotection sequences that add multiple unit operations, increasing solvent consumption and waste generation. The reliance on chromatographic purification to separate diastereomers and impurities further exacerbates production costs and limits the throughput capacity, creating bottlenecks for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative process disclosed in the patent data circumvents these steric and chemical hurdles by employing specially designed, cyclically protected side chain acids. Instead of struggling with the direct coupling of unstable beta-lactams, this method activates the side chain carboxylic acid in situ, typically forming a mixed anhydride with pivaloyl chloride. This activated species is significantly more reactive towards the hindered 13-hydroxyl group, allowing the esterification to proceed under much milder conditions, often at ambient or slightly elevated temperatures. A critical advantage of this approach is the stability of the cyclic protecting groups on the side chain, which prevents self-polymerization and ensures that the acylation occurs selectively at the taxane nucleus without over-acylation of other hydroxyl groups. As illustrated in the comprehensive reaction scheme below, this convergent strategy streamlines the synthesis into fewer, higher-yielding steps, directly supporting the commercial scale-up of complex pharmaceutical intermediates.
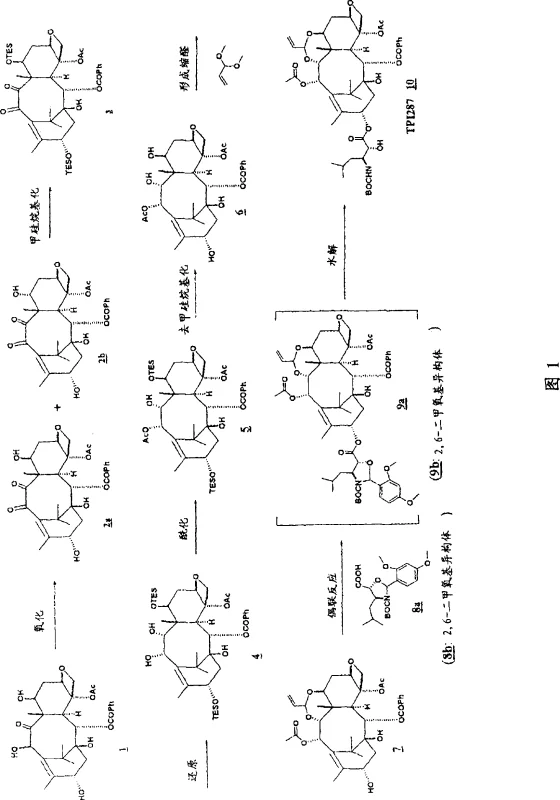
Mechanistic Insights into Mixed Anhydride Activation and Coupling
To fully appreciate the technical superiority of this synthesis route, one must examine the mechanistic details of the coupling reaction and the precursor modifications. The process begins with the selective oxidation of the taxane precursor, such as 10-deacetylbaccatin III, to form a 9,10-diketone intermediate. This oxidation is carefully controlled using reagents like copper chloride and triethylamine at low temperatures to prevent degradation of the sensitive oxetane ring. Following oxidation, the diketone undergoes a stereoselective reduction, often utilizing lithium borohydride, to establish the correct 9,10-diol stereochemistry required for biological activity. The subsequent protection of these hydroxyl groups, for instance as triethylsilyl ethers, is crucial for directing the regioselectivity of the final acylation. The core innovation lies in the activation of the side chain acid; by converting the acid into a mixed anhydride using pivaloyl chloride and a tertiary amine base like N-methylmorpholine, the electrophilicity of the carbonyl carbon is enhanced. This allows the nucleophilic attack by the C-13 oxygen to occur rapidly, even within the sterically crowded environment of the taxane core, minimizing the residence time of reactive intermediates and reducing the potential for side reactions.
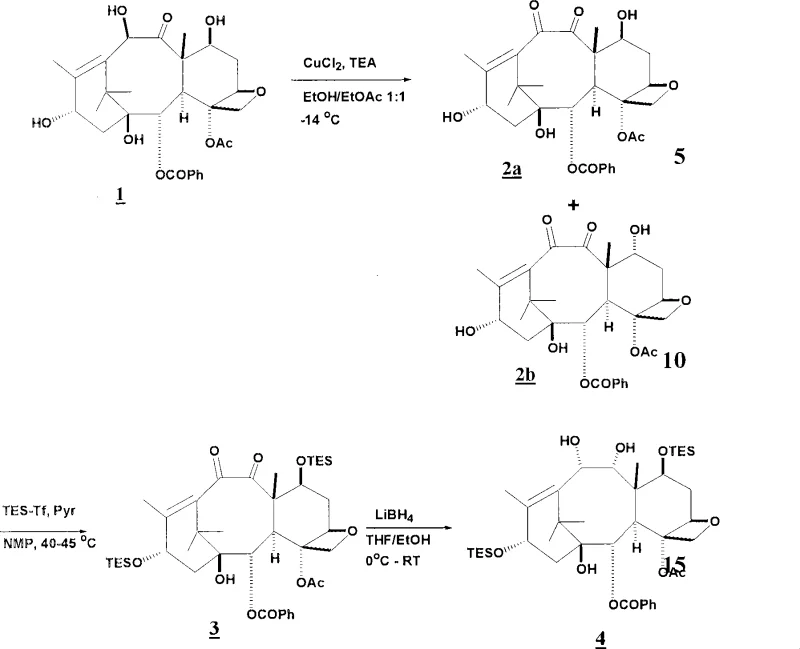
Impurity control is another pillar of this mechanistic design. In traditional syntheses, the risk of epimerization at the C-13 chiral center is a constant threat, especially during basic workups or prolonged reaction times. The use of cyclically protected side chains, such as N,O-acetals, provides a rigid structural framework that locks the stereochemistry of the side chain alpha-carbon. Furthermore, the deprotection steps described in the patent utilize mild acidic hydrolysis rather than harsh hydrogenolysis or strong bases. For example, the removal of the acetal protecting group and the final liberation of the free amine can be achieved using dilute hydrochloric acid in methanol at low temperatures. This gentle deprotection protocol ensures that the newly formed ester bond remains intact and that the stereochemical integrity of the molecule is preserved throughout the final stages of synthesis. By avoiding transition metal catalysts for deprotection where possible, the process also eliminates the risk of heavy metal contamination, simplifying the downstream purification requirements and ensuring compliance with stringent regulatory limits for residual metals in API production.
How to Synthesize TPI287 Efficiently
The practical execution of this synthesis involves a sequence of precise chemical transformations that convert readily available natural product precursors into high-value therapeutic intermediates. The process is characterized by its operational simplicity and the ability to isolate intermediates via crystallization rather than chromatography, which is a key factor for industrial viability. The following guide outlines the critical phases of the synthesis, emphasizing the conditions required to maximize yield and purity at each stage. Operators must pay close attention to temperature control during the oxidation and reduction steps, as well as the stoichiometry of the coupling reagents, to ensure consistent batch-to-batch performance. Detailed standardized operating procedures for these steps are essential for maintaining quality standards.
- Selective oxidation of 10-deacetylbaccatin III to form a 9,10-diketone intermediate using copper chloride and triethylamine.
- Protection of hydroxyl groups followed by stereoselective reduction to generate the 9,10-diol scaffold.
- Formation of an acetal protecting group and subsequent coupling with a cyclically protected side chain acid using mixed anhydride activation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers compelling benefits that extend beyond mere technical feasibility. For procurement managers and supply chain directors, the primary value proposition lies in the substantial simplification of the manufacturing workflow. By reducing the total number of synthetic steps and eliminating the need for resource-intensive chromatographic purifications, the process inherently lowers the cost of goods sold. The reliance on crystallization for purification not only reduces solvent usage but also accelerates the production cycle time, allowing for faster turnover of inventory and improved responsiveness to market demand fluctuations. Additionally, the use of stable, isolable intermediates enhances supply chain reliability, as key building blocks can be stockpiled without significant degradation, mitigating the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent consumption through crystallization-based purification drive significant operational cost savings. The avoidance of complex chromatographic separations reduces both capital expenditure on equipment and ongoing operational expenses related to silica gel and solvent disposal. Furthermore, the high yields reported in the patent examples suggest a more efficient utilization of the expensive taxane nucleus, maximizing the output per kilogram of starting material and improving the overall economic viability of the production campaign.
- Enhanced Supply Chain Reliability: The robustness of the mixed anhydride coupling method ensures consistent reaction performance, reducing the likelihood of batch failures that can disrupt supply schedules. The ability to use readily available reagents like pivaloyl chloride and common organic solvents means that the supply chain is less vulnerable to geopolitical constraints or shortages of exotic chemicals. This stability allows for more accurate forecasting and long-term planning, ensuring a continuous flow of high-purity intermediates to downstream API manufacturers without unexpected interruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The reduced generation of hazardous waste, particularly heavy metal residues and spent chromatography media, aligns with increasingly strict environmental regulations and corporate sustainability goals. This eco-friendly profile not only minimizes disposal costs but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain, facilitating easier regulatory approvals in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this taxane synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does this process improve upon traditional beta-lactam coupling methods?
A: This method utilizes cyclically protected side chain acids and mixed anhydrides, which significantly reduces steric hindrance issues at the C-13 position and minimizes epimerization risks compared to traditional beta-lactam acylation.
Q: What are the key advantages regarding purification in this synthesis route?
A: The process is designed to avoid extensive reverse-phase and normal-phase purification steps by utilizing crystallization and selective precipitation, thereby streamlining the isolation of high-purity intermediates.
Q: Can this method be applied to both Paclitaxel and Docetaxel synthesis?
A: Yes, the core methodology involving the esterification of the taxane nucleus with protected side chains is adaptable for producing Paclitaxel, Docetaxel, and analogs like TPI287 by varying the specific side chain precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TPI287 Supplier
As the demand for next-generation taxane therapies continues to grow, partnering with a technically proficient manufacturing partner is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of TPI287 or related taxane intermediate meets the highest international standards for safety and efficacy. Our facility is equipped to handle the specific challenges of taxane chemistry, including the precise temperature control and inert atmosphere conditions required for the mixed anhydride coupling steps described in the patent.
We invite you to collaborate with us to optimize your supply chain and reduce your time to market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, identifying opportunities to streamline your sourcing strategy. We encourage you to contact us to request specific COA data for our current inventory and to discuss route feasibility assessments for your custom synthesis projects. By choosing NINGBO INNO PHARMCHEM, you are selecting a partner dedicated to quality, reliability, and innovation in the field of pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
