Advanced Synthesis of Chiral Imidazolone NHC Precursors for High-Performance Asymmetric Catalysis
Advanced Synthesis of Chiral Imidazolone NHC Precursors for High-Performance Asymmetric Catalysis
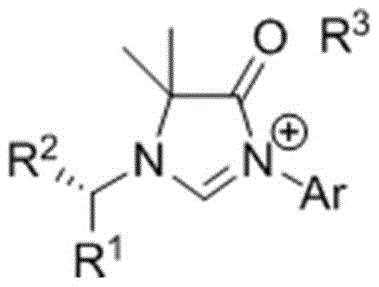
The landscape of homogeneous catalysis has been profoundly transformed by the advent of N-heterocyclic carbenes (NHCs), which have emerged as superior alternatives to traditional phosphine ligands due to their robust thermal stability and tunable electronic properties. A groundbreaking development in this field is detailed in patent CN108947909B, which discloses a novel class of chiral nitrogen heterocyclic carbene precursor compounds featuring an imidazolone skeleton. This innovation addresses a critical gap in ligand design by introducing a carbonyl functional group directly into the heterocyclic backbone, thereby creating a unique electronic environment that enhances catalytic performance in asymmetric synthesis. The patent outlines a highly efficient, three-step synthetic strategy that converts inexpensive bromoacetyl bromide into sophisticated chiral precursors, offering a streamlined pathway for producing high-value pharmaceutical intermediates. By enabling the precise introduction of chiral centers on the nitrogen side chains, this technology empowers chemists to fine-tune steric and electronic parameters, ultimately facilitating the construction of complex chiral molecules with exceptional enantioselectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-heterocyclic carbene ligands has relied heavily on five-membered imidazole or dihydroimidazole scaffolds, which, while effective, often lack the electronic versatility required for next-generation catalytic challenges. Traditional methods for introducing chirality into these systems frequently involve multi-step resolutions or the use of expensive chiral pool starting materials, leading to prohibitive costs and low atom economy. Furthermore, conventional imidazolium salts often exhibit limited stability under harsh reaction conditions or fail to provide the necessary electronic modulation for difficult transformations, such as the asymmetric addition of organozinc reagents to aldehydes. The absence of electron-withdrawing groups like carbonyls in the standard backbone restricts the ligand's ability to stabilize specific metal oxidation states, limiting the scope of applicable reactions. Consequently, the industry has faced a persistent bottleneck in accessing ligands that combine high stereocontrol with economic viability, necessitating a paradigm shift in synthetic design.
The Novel Approach
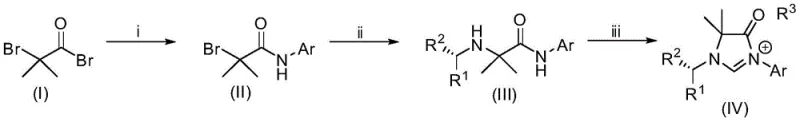
The methodology presented in patent CN108947909B revolutionizes this landscape by leveraging a concise three-step sequence that begins with the commodity chemical bromoacetyl bromide. This approach bypasses the need for complex heterocycle construction from scratch, instead building the imidazolone core through a logical progression of amidation, nucleophilic substitution, and cyclization. The key innovation lies in the strategic placement of the carbonyl group within the ring, which not only stabilizes the carbene center through resonance but also imparts distinct electrophilic character to the scaffold. As illustrated in the comprehensive reaction scheme below, the process allows for the modular incorporation of diverse aromatic amines and chiral primary amines, generating a library of structurally novel precursors with high efficiency. This modularity ensures that researchers can rapidly iterate on ligand structures to optimize performance for specific substrates, significantly accelerating the development of new catalytic processes for drug discovery.
Mechanistic Insights into Imidazolone Skeleton Formation
The electronic superiority of the imidazolone skeleton stems from the interplay between the carbene carbon's sp2 hybridized lone pair and the adjacent carbonyl group's pi-system. Unlike traditional imidazol-2-ylidenes, where stabilization is primarily achieved through the lone pairs of two nitrogen atoms, the imidazolone variant introduces a competitive electron-withdrawing effect that fine-tunes the sigma-donating strength of the carbene. This results in a ligand that maintains strong metal-carbon bonding while offering enhanced resistance to oxidative degradation. The cyclization step, which converts the acyclic chiral diamine intermediate into the final imidazolone salt, proceeds via a Lewis acid-catalyzed condensation with orthoformates. This mechanism ensures the formation of a rigid, planar heterocyclic core that locks the chiral information from the N-substituents into a defined spatial arrangement. Such structural rigidity is paramount for inducing high levels of enantioselectivity in catalytic cycles, as it minimizes conformational flexibility that could otherwise lead to racemic background reactions.
Furthermore, the presence of the carbonyl functionality allows for potential secondary interactions with substrates or counterions, adding another layer of control over the reaction trajectory. In the context of metal complexation, these ligands form stable organometallic complexes with metals such as rhodium, iridium, and copper, which are pivotal in cross-coupling and addition reactions. The electron-deficient nature of the imidazolone ring can facilitate the oxidative addition step in catalytic cycles, often the rate-determining step in many transformations. By understanding these mechanistic nuances, process chemists can better predict the behavior of these ligands in scale-up scenarios, ensuring that the high selectivity observed in the laboratory is maintained during commercial production. The ability to systematically vary the R-groups on the nitrogen atoms further allows for the optimization of steric bulk, preventing unwanted dimerization of the metal center and extending the catalyst's lifetime.
How to Synthesize Chiral Imidazolone NHC Precursors Efficiently
The synthesis of these advanced ligand precursors is designed for operational simplicity, utilizing standard laboratory equipment and readily available reagents to ensure reproducibility across different manufacturing sites. The process begins with the controlled amidation of bromoacetyl bromide, followed by the stereoselective introduction of chirality, and concludes with a thermal cyclization to close the heterocyclic ring. Each step has been optimized to maximize yield and minimize byproduct formation, making it suitable for both small-scale discovery and larger pilot plant operations. For detailed procedural specifics regarding stoichiometry, solvent choices, and purification techniques, please refer to the standardized guide below which encapsulates the core technical workflow.
- React bromoacetyl bromide with an aromatic amine in dichloromethane with triethylamine to form the alpha-bromo amide intermediate.
- Perform nucleophilic substitution by reacting the alpha-bromo amide with a chiral primary amine in THF using sodium hydride as a base.
- Cyclize the chiral diamine intermediate with triethyl orthoformate and ammonium tetrafluoroborate at elevated temperature to yield the final imidazolone salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits, primarily driven by the utilization of low-cost feedstock and the elimination of complex purification bottlenecks. The reliance on bromoacetyl bromide as a starting material decouples the supply chain from volatile markets associated with exotic chiral building blocks, ensuring a stable and predictable flow of raw materials. Moreover, the high atom economy of the three-step sequence means that less waste is generated per unit of product, aligning with modern sustainability goals and reducing disposal costs. For supply chain managers, the robustness of the reaction conditions—operating effectively at ambient or moderately elevated temperatures—translates to lower energy consumption and reduced risk of thermal runaways, enhancing overall plant safety and operational continuity.
- Cost Reduction in Manufacturing: The economic viability of this process is anchored in the replacement of expensive, multi-step ligand syntheses with a direct route from commodity chemicals. By avoiding the use of precious metal catalysts during the ligand synthesis phase itself and relying on simple organic transformations, the direct material costs are drastically lowered. Additionally, the high yields reported in the patent examples indicate that minimal raw material is lost to side reactions, further improving the cost-per-gram metric. This efficiency allows pharmaceutical manufacturers to allocate budget towards other critical areas of drug development while maintaining access to high-performance catalytic tools.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the sourcing of diverse aromatic amines and chiral amines from multiple global suppliers, mitigating the risk of single-source dependency. Since the core scaffold is constructed from a stable, non-perishable bromo-acetyl derivative, inventory management becomes more straightforward, with longer shelf-life expectations for intermediates. This flexibility ensures that production schedules can be maintained even when specific chiral auxiliaries face temporary shortages, as the core process remains unchanged and only the side-chain inputs need adjustment.
- Scalability and Environmental Compliance: The synthetic pathway employs common organic solvents such as dichloromethane and tetrahydrofuran, which are well-understood in terms of recovery and recycling protocols, facilitating compliance with environmental regulations. The absence of cryogenic conditions or ultra-high pressure requirements simplifies the engineering controls needed for scale-up, allowing existing multipurpose reactors to be utilized without major capital investment. This ease of translation from bench to plant ensures that the lead time for bringing new chiral intermediates to market is significantly compressed, providing a competitive edge in fast-paced therapeutic areas.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral imidazolone technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, aiming to clarify the practical implications for process development teams. Understanding these aspects is crucial for integrating this new ligand class into existing catalytic platforms and maximizing its potential value.
Q: What is the primary advantage of the imidazolone skeleton over traditional imidazole NHC ligands?
A: The introduction of a carbonyl functional group into the imidazole ring skeleton significantly alters the electron cloud density distribution. This unique electronic profile enhances sigma-donating properties while modifying pi-accepting characteristics, leading to superior reactivity and selectivity in metal-catalyzed asymmetric reactions compared to traditional phosphine or standard NHC ligands.
Q: How does this synthesis method improve cost efficiency for pharmaceutical intermediate production?
A: The process utilizes bromoacetyl bromide, a low-cost and readily available industrial raw material, as the starting point. By streamlining the synthesis into only three robust steps with high yields and avoiding expensive transition metal catalysts in the ligand preparation itself, the overall manufacturing cost is substantially reduced, making high-performance chiral ligands more accessible for large-scale API synthesis.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the methodology relies on standard organic transformations such as amidation and nucleophilic substitution using common solvents like dichloromethane and THF. The reaction conditions are mild to moderate (0°C to 120°C), and the purification involves standard column chromatography or crystallization, ensuring that the process can be effectively scaled up from laboratory grams to multi-kilogram commercial batches without significant engineering hurdles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Imidazolone NHC Precursor Supplier
As the demand for enantiomerically pure pharmaceutical intermediates continues to surge, the ability to deploy advanced catalytic technologies like those described in CN108947909B becomes a key differentiator for success. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services that bridge the gap between innovative academic research and commercial reality. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral precursor meets the exacting standards required for GMP-grade API synthesis.
We invite forward-thinking R&D and procurement leaders to collaborate with us to unlock the full potential of these imidazolone-based ligands for your specific synthesis challenges. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current process bottlenecks. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in chiral intermediate manufacturing can drive efficiency and innovation in your supply chain.
