Advanced Manufacturing of 1,3,4-Thiadiazole Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for heterocyclic compounds, particularly 1,3,4-thiadiazole derivatives, which serve as critical scaffolds for antibacterial agents like acetazolamide and various other bioactive molecules. Patent CN102060804B introduces a transformative methodology for preparing these derivatives, specifically targeting the synthesis of 2-propionamido-5-N-methylpiperazinesulfonyl-1,3,4-thiadiazole. This innovation addresses long-standing challenges in organic synthesis by replacing hazardous operational protocols with a streamlined, low-toxicity approach. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener chemistry without compromising yield or structural integrity. The disclosed route not only enhances the safety profile of the manufacturing process but also aligns perfectly with modern regulatory demands for cleaner production environments. By leveraging mild reaction conditions and readily available reagents, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex thiadiazole frameworks has been plagued by severe operational constraints that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional methods often necessitate the use of highly toxic reagents and extreme reaction conditions, such as elevated pressures or temperatures that exceed the safety limits of standard processing equipment. These harsh environments frequently lead to the formation of difficult-to-remove impurities, thereby complicating downstream purification and drastically increasing production costs. Furthermore, the instability of certain intermediates under conventional conditions often results in inconsistent batch-to-batch quality, posing significant risks to supply chain continuity. The reliance on aggressive chemical transformations also raises substantial environmental concerns, requiring expensive waste treatment protocols that erode profit margins. Consequently, manufacturers have struggled to find a balance between high throughput and the stringent safety standards required for producing high-purity thiadiazole derivatives.
The Novel Approach
In stark contrast to legacy techniques, the novel approach detailed in the patent utilizes a温和 (mild) three-step sequence that prioritizes safety and scalability. The process initiates with a controlled acylation, followed by a precise bromination, and concludes with a nucleophilic substitution, all conducted within a temperature window of 0°C to 80°C. This moderate thermal profile allows for the use of standard glass-lined steel reactors, eliminating the need for specialized high-pressure vessels. The strategic selection of solvents, such as glacial acetic acid and absolute ethanol, ensures excellent solubility of reactants while facilitating easy product isolation through simple filtration and recrystallization. Moreover, the stoichiometry is carefully optimized, with molar ratios like 1:1.2 to 1.5 for the acylation step, minimizing excess reagent waste. This methodological refinement translates directly into cost reduction in pharma manufacturing by reducing energy consumption and simplifying waste management logistics.
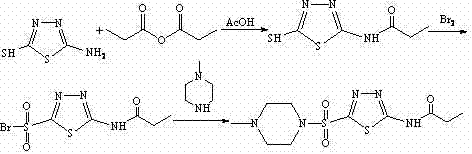
Mechanistic Insights into Propionylation and Sulfonyl Bromide Formation
The core of this synthetic success lies in the meticulous control of the electrophilic substitution and oxidation mechanisms. In the first step, the nucleophilic amino group of 2-amino-5-mercapto-1,3,4-thiadiazole attacks the carbonyl carbon of propionic anhydride. This acylation is catalyzed by the acidic environment of glacial acetic acid, which protonates the anhydride to enhance its electrophilicity. Maintaining the temperature at 70-80°C is critical; it provides sufficient activation energy for the reaction to proceed to completion within 20-40 minutes without degrading the sensitive thiadiazole ring. The subsequent cooling and filtration at pH 5.0-6.0 ensure that the 2-propionamido-5-mercapto intermediate precipitates in high purity, effectively removing unreacted anhydride and acetic acid byproducts. This precise pH control is a key factor in achieving the reported yields of over 90%, demonstrating the robustness of the mechanism.
The second mechanistic phase involves the oxidation of the thiol group to a sulfonyl bromide using liquid bromine. This transformation is highly exothermic and requires strict temperature control at 0-5°C to prevent over-oxidation to sulfonic acids or bromination of the heterocyclic ring. The use of 30-35wt% aqueous acetic acid as the solvent medium provides a polar environment that stabilizes the ionic intermediates formed during the bromination. The molar ratio of liquid bromine to the thiol substrate is maintained between 1:2.5 and 1:4, ensuring complete conversion while accounting for potential side reactions. TLC monitoring confirms the disappearance of the starting material, indicating a clean conversion to the sulfonyl bromide species. This intermediate is crucial as it serves as a highly reactive electrophile for the final amination step, enabling the efficient introduction of the N-methylpiperazine moiety under mild conditions.
How to Synthesize 2-Propionamido-5-N-methylpiperazinesulfonyl-1,3,4-thiadiazole Efficiently
Executing this synthesis requires adherence to strict procedural parameters to maximize yield and purity. The process begins with the preparation of the mercapto precursor, followed by its conversion to the sulfonyl bromide, and finally the coupling with N-methylpiperazine. Each stage demands precise temperature regulation and stoichiometric balance to avoid the accumulation of impurities that could compromise the final API quality. The detailed standardized synthesis steps below outline the exact conditions, reagent grades, and workup procedures necessary to replicate the high success rates observed in the patent embodiments. Operators should pay particular attention to the cooling phases during bromination and the recrystallization steps to ensure the product meets stringent pharmaceutical specifications.
- Prepare 2-propionamido-5-mercapto-1,3,4-thiadiazole by reacting 2-amino-5-mercapto-1,3,4-thiadiazole with propionic anhydride in glacial acetic acid at 70-80°C.
- Synthesize 2-propionamido-5-sulfonyl bromide-1,3,4-thiadiazole by treating the mercapto intermediate with liquid bromine in aqueous acetic acid at 0-5°C.
- Complete the synthesis by reacting the sulfonyl bromide intermediate with N-methylpiperazine in absolute ethanol to form the final sulfonamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of highly toxic reagents and the shift to mild operating conditions significantly reduce the regulatory burden associated with hazardous material handling and storage. This translates into lower insurance premiums and reduced compliance costs, contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the high yields observed in each step minimize raw material consumption, allowing companies to procure smaller quantities of starting materials for the same output volume. The simplicity of the workup procedures, primarily involving filtration and recrystallization, reduces the demand for complex separation equipment and skilled labor, streamlining the production workflow. These factors collectively enhance the resilience of the supply chain against market volatility and raw material shortages.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing inexpensive and widely available starting materials such as propionic anhydride and N-methylpiperazine. The high conversion rates in each step mean that less raw material is wasted, directly lowering the cost of goods sold. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further driving down operational expenditures. The simplified purification process eliminates the need for expensive chromatography or complex distillation setups, resulting in a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals rather than exotic or custom-synthesized reagents, manufacturers can secure a more stable supply of inputs. The robustness of the reaction conditions ensures consistent output quality, reducing the risk of batch failures that could disrupt delivery schedules. This reliability is crucial for maintaining long-term contracts with downstream pharmaceutical clients who demand uninterrupted supply of high-purity intermediates. The ability to scale the process from laboratory to industrial volumes without significant re-engineering further strengthens supply chain continuity.
- Scalability and Environmental Compliance: The process is inherently scalable due to its use of standard unit operations and benign solvents. The reduced toxicity of the reagents simplifies waste treatment, making it easier to meet increasingly strict environmental regulations. This eco-friendly profile not only mitigates legal risks but also enhances the corporate social responsibility standing of the manufacturer. The ability to handle large volumes safely in standard reactors ensures that production can be ramped up quickly to meet surging market demand without compromising safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiadiazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity provided here aims to facilitate informed decision-making regarding process adoption and partnership opportunities.
Q: What are the primary advantages of this thiadiazole synthesis method over conventional routes?
A: This method significantly reduces raw material toxicity and utilizes milder reaction conditions compared to traditional processes. It avoids harsh operating environments, making it highly suitable for industrial-scale production while maintaining high product purity.
Q: Is this process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process is explicitly designed for industrial suitability. The reaction temperatures range from 0°C to 80°C, which are easily manageable in standard glass-lined reactors, ensuring consistent quality and safety during large-scale operations.
Q: What purity levels can be achieved with this synthetic route?
A: Experimental embodiments demonstrate that the final product can achieve purity levels exceeding 97% after recrystallization. The high yields in each step (over 85%) contribute to a robust impurity profile suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4-Thiadiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the potential of this patented methodology and is fully prepared to assist clients in translating this laboratory success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,3,4-thiadiazole derivatives meets the highest industry standards. We are committed to delivering excellence in both product quality and service.
We invite you to collaborate with us to unlock the full potential of this advanced synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. By partnering with us, you gain access to our deep expertise in process optimization and supply chain management. Please contact us today to request specific COA data and route feasibility assessments, and let us help you streamline your manufacturing operations for a more profitable and sustainable future.
