Advanced Regioselective Selenization for Scalable Production of Naphthyl Intermediates
Advanced Regioselective Selenization for Scalable Production of Naphthyl Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign pathways to construct complex heteroatom-containing scaffolds. A significant advancement in this domain is detailed in patent CN113149879A, which discloses a novel preparation method for 4-seleno-naphthyl esters and 4-seleno-naphthyl amides. These selenium-functionalized naphthalene derivatives are critical building blocks, possessing immense potential as intermediates for bioactive compounds and functional materials due to the unique redox properties of selenium. The patented methodology leverages phenyliodine(III) bis(trifluoroacetate) (PIFA) as a mild yet powerful oxidant to achieve direct regioselective selenization at the 4-position of the naphthalene ring. This approach represents a paradigm shift from traditional harsh conditions, offering a streamlined route that operates at room temperature in common solvents like dichloromethane (DCM) or dimethyl sulfoxide (DMSO). For R&D directors and procurement specialists, this technology promises not only high yields but also a significant reduction in operational complexity and waste generation.
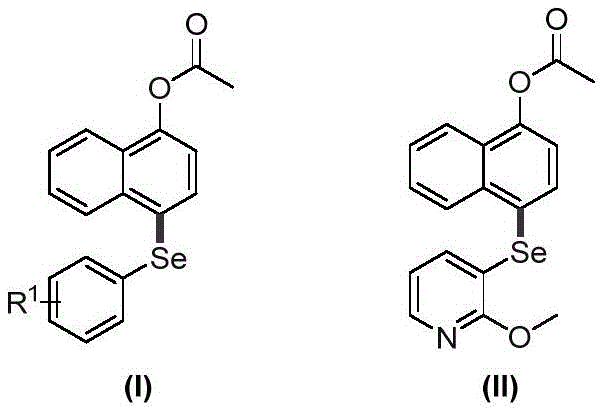
The structural versatility of the products is evident, allowing for the incorporation of various substituents such as alkyl, halogen, nitro, or cyano groups on the phenyl ring attached to the selenium atom. This flexibility is crucial for medicinal chemists aiming to optimize the pharmacokinetic profiles of lead compounds. Furthermore, the ability to synthesize both ester and amide variants expands the utility of this platform across different therapeutic classes. As a reliable pharmaceutical intermediate supplier, understanding these structural nuances is key to delivering custom synthesis solutions that meet the rigorous demands of drug discovery pipelines. The patent explicitly highlights the simplicity of the raw materials, utilizing readily available naphthyl esters, naphthylamides, and diselenides, which directly translates to better supply chain stability and cost predictability for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct selenization of naphthalene rings has been fraught with challenges that hinder industrial adoption. Prior art, such as the methods reported by the Kumar research group in 2013, relied on potassium persulfate promotion in trifluoroacetic acid at elevated temperatures around 80°C. Such conditions are not only energy-intensive but also pose safety risks due to the corrosive nature of the solvent and the strong oxidant at high heat. Subsequent developments by Yang's group utilized silver salts (AgSbF6) and copper acetate, introducing expensive transition metals into the process. The presence of these metals necessitates rigorous purification steps to meet the strict residual metal limits imposed by regulatory bodies for pharmaceutical ingredients. Additionally, earlier methods often suffered from poor regioselectivity or required specialized substrates like 1-methoxy naphthalenes, limiting their scope. The reliance on harsh acidic environments or precious metal catalysts creates a bottleneck in cost reduction in API manufacturing, as it increases both the bill of materials and the environmental footprint of the synthesis.
The Novel Approach
In stark contrast, the method described in CN113149879A utilizes a hypervalent iodine reagent, PIFA, to drive the reaction under remarkably mild conditions. The process occurs at room temperature, eliminating the need for external heating and significantly reducing energy consumption. By employing a metal-free oxidative system, the protocol avoids the introduction of heavy metal contaminants, thereby simplifying the downstream purification workflow. The reaction demonstrates excellent regioselectivity, specifically targeting the 4-position of the naphthyl ring, which is often difficult to access with high fidelity using electrophilic aromatic substitution alone. This precision minimizes the formation of isomeric byproducts, leading to higher crude purity and improved overall yields. The general reaction scheme illustrates the convergence of the naphthyl substrate and the diselenide reagent mediated by PIFA, showcasing a clean transformation that is highly amenable to scale-up.
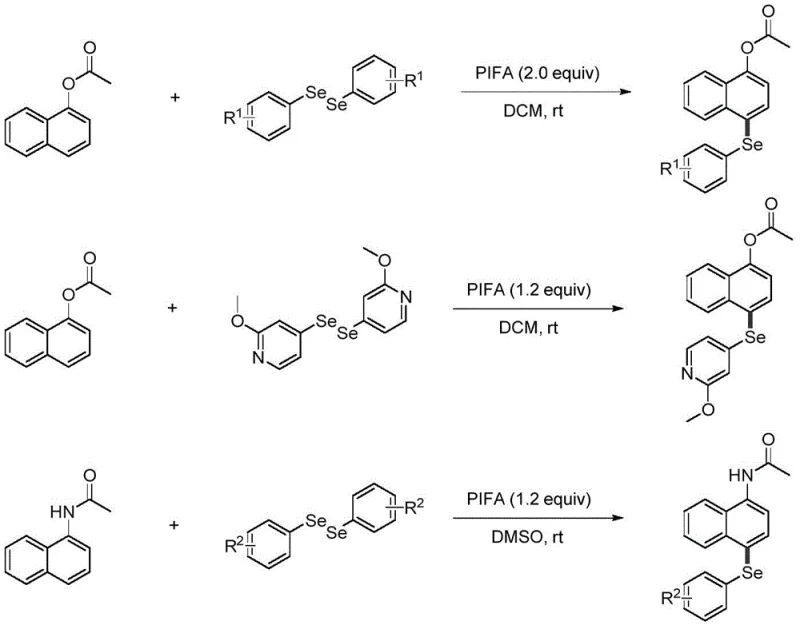
The adaptability of this novel approach is further underscored by its compatibility with a wide range of functional groups. Whether the substrate is an electron-rich or electron-deficient diselenide, the reaction proceeds efficiently, albeit with varying reaction times that can be optimized. For instance, electron-deficient diselenides might require longer stirring times, but they still afford the desired products in good yields without decomposing. This robustness makes the method suitable for the commercial scale-up of complex pharmaceutical intermediates, where batch-to-batch consistency is paramount. The use of common organic solvents like DCM and DMSO ensures that the process can be easily integrated into existing manufacturing infrastructure without requiring specialized equipment or hazardous solvent handling protocols.
Mechanistic Insights into PIFA-Mediated Oxidative Selenization
Understanding the mechanistic underpinnings of this transformation is vital for process chemists aiming to optimize reaction parameters. The proposed mechanism begins with the interaction between PIFA and the diselenide bond. PIFA acts as a single-electron oxidant, cleaving the Se-Se bond to generate a reactive selenyl radical species along with a trifluoroacetoxy iodobenzene radical. This radical generation step is critical as it initiates the catalytic cycle without the need for external initiators or light. The selenyl radical then undergoes an electrophilic attack on the electron-rich naphthalene ring of the ester or amide substrate. Due to the directing effects of the ester or amide group at the 1-position (or 8-position depending on numbering convention used in the patent context, effectively ortho/para directing relative to the fused system), the radical attack occurs selectively at the 4-position. This regioselectivity is driven by the stability of the resulting radical intermediate and steric factors inherent to the naphthalene scaffold.
Following the radical addition, the intermediate carbon-centered radical is further oxidized by the iodine(III) species present in the solution. This second oxidation step converts the radical into a cationic selenonium intermediate. The final step involves deprotonation of this cationic species to restore aromaticity, yielding the stable 4-seleno-naphthyl product. This radical-cationic pathway explains the high efficiency and mild conditions of the reaction. From an impurity control perspective, the absence of transition metals means that metal-chelated side products are non-existent. The primary impurities likely arise from over-oxidation or homocoupling of the diselenide, both of which can be minimized by controlling the stoichiometry of PIFA. The patent specifies optimal molar ratios, such as 1:2.0 for esters and 1:1.2 for amides, which are empirically determined to maximize yield while minimizing reagent waste. This mechanistic clarity allows for precise tuning of the process to ensure high-purity pharmaceutical intermediates are consistently produced.
How to Synthesize 4-Seleno-Naphthyl Derivatives Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable selenium-containing compounds. The procedure is straightforward, involving the mixing of the naphthyl substrate and diselenide in the chosen solvent, followed by the addition of PIFA. Reaction monitoring is typically performed via thin-layer chromatography (TLC), allowing for real-time assessment of conversion. Upon completion, a standard aqueous workup involving extraction with dichloromethane and washing with water effectively removes the iodine byproducts and excess reagents. The crude product is then purified using column chromatography, typically with a petroleum ether and ethyl acetate gradient. Detailed standardized synthetic steps for specific examples are provided below to guide laboratory implementation.
- Prepare the reaction mixture by combining naphthyl ester or naphthylamide with a diselenide compound in the appropriate solvent (DCM for esters, DMSO for amides).
- Add PIFA (phenyliodine(III) bis(trifluoroacetate)) as the oxidant, maintaining a molar ratio of 1: 2.0 for esters or 1:1.2 for amides relative to the substrate.
- Stir the reaction at room temperature under air atmosphere until completion, monitored by TLC, followed by standard aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PIFA-mediated selenization technology offers tangible strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts like silver and copper directly impacts the cost structure of the supply chain. Transition metals are subject to significant price volatility and geopolitical supply risks; replacing them with a stable organic oxidant like PIFA mitigates these risks. Furthermore, the elimination of heavy metals simplifies the quality control burden. Removing trace metals to ppm levels often requires specialized scavenging resins or multiple recrystallizations, which add time and cost to the manufacturing process. By avoiding these metals entirely, the process inherently reduces the number of unit operations required, leading to a leaner and more cost-effective production flow.
- Cost Reduction in Manufacturing: The economic advantages of this method are multifaceted. Firstly, the raw materials—naphthyl esters, amides, and diselenides—are commodity chemicals that are widely available from multiple global suppliers, ensuring competitive pricing and supply security. Secondly, the reaction operates at room temperature, which drastically cuts down on energy costs associated with heating and cooling large reactors. Thirdly, the simplified workup and purification process, devoid of metal scavenging steps, reduces solvent consumption and labor hours. These factors combined contribute to a substantially lower cost of goods sold (COGS), making the final intermediates more attractive for budget-conscious drug development projects.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance to air and moisture means that the process does not require expensive inert atmosphere equipment or strictly anhydrous solvents, which can be logistical bottlenecks. This ease of operation allows for manufacturing in a wider range of facilities, increasing the potential supplier base. Additionally, the short reaction times observed for many substrates (often completing within hours) improve throughput and asset utilization. Faster turnaround times mean that inventory levels can be kept lower, reducing working capital requirements and enhancing the agility of the supply chain to respond to fluctuating demand.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden hazards, but this method appears inherently scalable due to its mild exothermic profile and lack of hazardous gases. The use of DCM and DMSO, while requiring proper handling, is standard in the industry with well-established recovery and recycling protocols. From an environmental standpoint, the atom economy is reasonable given the oxidative nature of the transformation, and the absence of heavy metal waste streams simplifies effluent treatment. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the risk of compliance-related shutdowns and enhancing the green credentials of the manufactured products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenization technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this chemistry into their existing portfolios.
Q: What are the advantages of using PIFA over traditional metal catalysts for selenization?
A: Using PIFA eliminates the need for expensive and toxic transition metal catalysts like silver or copper, simplifying downstream purification and reducing heavy metal contamination risks in the final API.
Q: Can this method be applied to substrates with electron-withdrawing groups?
A: Yes, the patent demonstrates successful selenization of substrates containing nitro, cyano, and halogen groups, although reaction times may vary depending on the electronic nature of the substituent.
Q: Is the reaction sensitive to moisture or oxygen?
A: The process is robust and operates effectively under air atmosphere at room temperature, removing the need for stringent inert gas protection or anhydrous conditions typically required in organometallic chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Seleno-Naphthyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the PIFA-mediated selenization route described in CN113149879A for advancing selenium-based therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to pilot plant is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to detect even trace impurities. Whether you require custom synthesis of novel 4-seleno-naphthyl amides or bulk supply of ester intermediates, our infrastructure is designed to support your growth.
We invite you to collaborate with us to optimize your supply chain and reduce development timelines. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our implementation of this novel chemistry can lower your overall production costs. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in bringing high-quality selenium intermediates to the market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
