Advanced Regioselective Selenization for Commercial Scale-up of Complex Pharmaceutical Intermediates
The landscape of modern organic synthesis is continuously evolving to meet the stringent demands of the pharmaceutical and fine chemical industries, particularly regarding the efficient construction of selenium-containing scaffolds. Patent CN113788774A introduces a groundbreaking preparation method for 4-selenomethoxynaphthalene, a critical intermediate with significant potential in medicinal chemistry and materials science. This innovation leverages a regioselective selenization strategy that bypasses the limitations of traditional thermal methods, offering a pathway to high-purity products under remarkably mild conditions. The technical breakthrough lies in the utilization of PIFA (di(trifluoroacetoxy)iodobenzene) as a hypervalent iodine oxidant, which facilitates the direct functionalization of the carbon-hydrogen bond at the 4-position of 1-methoxynaphthalene. For R&D directors and procurement specialists, this represents a shift towards more sustainable and cost-effective manufacturing protocols that do not compromise on yield or structural integrity. The ability to synthesize diverse 4-seleno methoxy naphthalene derivatives using this protocol opens new avenues for drug discovery and the development of functional materials.
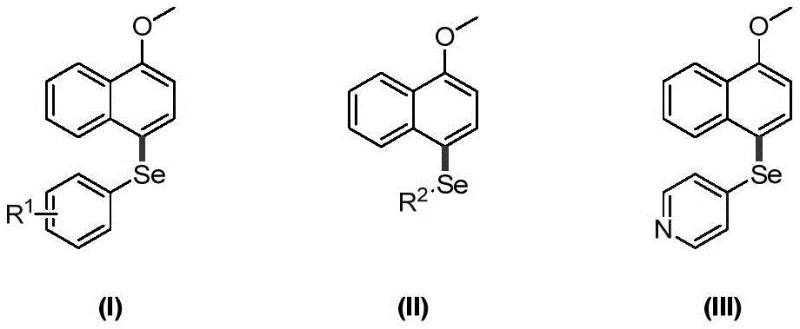
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of selenated naphthalene derivatives has relied heavily on methodologies that impose significant operational and economic burdens on manufacturing facilities. Prior art, such as the method reported by the Borah research group in 2014, necessitates the use of silver hexafluoroantimonate as a catalyst and hydrated copper acetate as an oxidant. These reactions typically require elevated temperatures around 120°C, which not only increases energy consumption but also introduces safety risks associated with high-thermal operations. Furthermore, the reliance on transition metals like silver and copper creates a substantial downstream processing challenge, as removing trace heavy metal residues to meet pharmaceutical purity standards is both technically difficult and expensive. The harsh conditions can also lead to the formation of unwanted by-products and isomeric impurities, complicating the purification process and reducing the overall atom economy of the synthesis. For supply chain managers, these factors translate into longer lead times and higher production costs, making conventional routes less attractive for large-scale commercialization.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a metal-free oxidation system driven by PIFA in dichloromethane (DCM) solvent at room temperature. This methodology fundamentally alters the reaction landscape by eliminating the need for external heating and expensive metal catalysts, thereby drastically simplifying the operational requirements. The reaction proceeds efficiently under an air atmosphere, demonstrating robustness that is highly desirable for industrial applications where inert gas protection might add complexity. By employing diselenide compounds as the selenium source and PIFA as the oxidant, the process achieves high yields with minimal formation of side products. The simplicity of the workup procedure, involving standard aqueous washing and column chromatography, further enhances the practicality of this route for commercial production. This shift from thermal, metal-catalyzed processes to ambient, hypervalent iodine-mediated chemistry represents a significant advancement in green chemistry principles, aligning with global trends towards reducing the environmental footprint of chemical manufacturing.
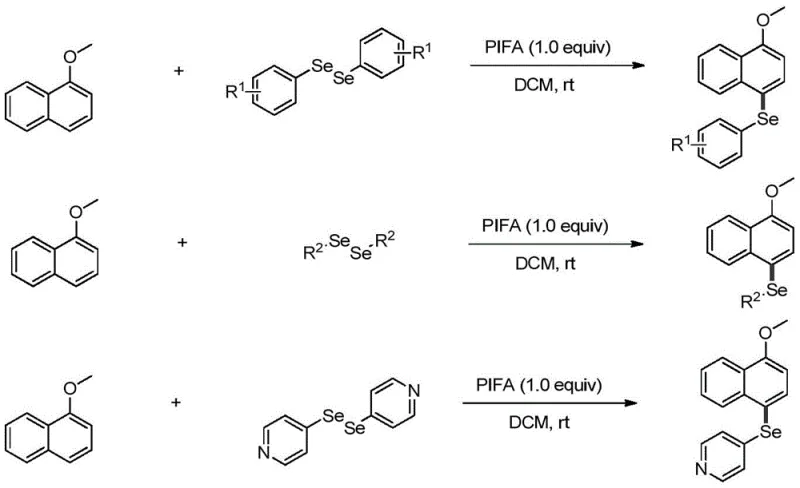
Mechanistic Insights into PIFA-Catalyzed Regioselective Selenization
The mechanistic pathway of this transformation is a sophisticated sequence of radical and ionic steps that ensures high regioselectivity and efficiency. The process initiates with the interaction between PIFA and the diselenide compound, leading to the homolytic cleavage of the selenium-selenium bond and the generation of a reactive selenium radical species alongside a trifluoroacetoxy iodobenzene radical. This selenium radical then engages in an electrophilic attack on the electron-rich aromatic ring of 1-methoxynaphthalene, specifically targeting the 4-position due to the electronic directing effects of the methoxy group. This step forms a seleno radical intermediate, which is a critical juncture determining the success of the functionalization. The mild conditions prevent over-oxidation or degradation of the sensitive naphthalene skeleton, preserving the structural integrity required for downstream applications. Understanding this radical generation and attack phase is crucial for R&D teams looking to optimize reaction parameters or adapt the chemistry to analogous substrates.
Following the radical attack, the seleno radical intermediate undergoes oxidation by the trifluoroacetyloxyiodobenzene radical to generate a seleno cation intermediate. This oxidation step is pivotal as it converts the radical species into a cationic center that is primed for the final aromatization. The subsequent deprotonation of this cationic intermediate restores the aromaticity of the naphthalene system, yielding the final 4-selenomethoxynaphthalene product. This mechanism explains the high purity observed in the examples, as the specific electronic requirements for the cation formation disfavor alternative reaction pathways that would lead to impurities. For quality control professionals, this mechanistic clarity provides confidence in the consistency of the impurity profile, ensuring that the synthesized intermediates meet the rigorous specifications demanded by the pharmaceutical industry. The absence of metal catalysts also means that the impurity spectrum is free from heavy metal contaminants, simplifying the analytical validation process.
How to Synthesize 4-Selenomethoxynaphthalene Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and solvent choices to maximize efficiency. The patent data indicates that maintaining a molar ratio of 1-methoxynaphthalene to diselenide compound at 1:2 is optimal for driving the reaction to completion and improving yield. Similarly, the ratio of substrate to the PIFA oxidant should be kept at 1:1.0 to ensure sufficient oxidative power without excessive reagent waste. Dichloromethane is identified as the preferred solvent, as alternative polar or non-polar solvents have been shown to result in reduced yields or complete failure of the reaction. Operators must monitor the reaction progress via TLC, as completion times can vary depending on the specific diselenide substituent, ranging from minutes to hours. Detailed standard operating procedures regarding safety handling of PIFA and selenium compounds should be followed to ensure personnel safety and environmental compliance.
- Prepare the reaction system by dissolving 1-methoxynaphthalene and the selected diselenide compound in dichloromethane (DCM) solvent under air atmosphere.
- Add PIFA (1.0 equivalent) as the oxidant to the mixture and stir at room temperature until TLC monitoring indicates complete consumption of the starting material.
- Quench the reaction with water, separate the organic layer, wash thoroughly, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of precious metal catalysts such as silver and copper removes a significant cost driver from the raw material bill, while also obviating the need for specialized metal scavenging resins or complex purification steps. This simplification of the downstream processing workflow translates into reduced operational expenditures and shorter manufacturing cycles, allowing for faster turnaround times on customer orders. Furthermore, the ability to conduct the reaction at room temperature significantly lowers energy consumption compared to thermal methods, contributing to both cost savings and sustainability goals. For procurement managers, the availability of cheap and easily obtained raw materials like 1-methoxynaphthalene and PIFA ensures a stable supply chain that is less susceptible to market volatility associated with rare metals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive transition metal catalysts and the reduction in energy infrastructure requirements. By avoiding the use of silver salts and high-temperature heating, manufacturers can achieve significant cost savings on both reagents and utilities. The simplified workup procedure reduces labor hours and solvent usage associated with extensive purification, further enhancing the overall cost-efficiency of the production line. These qualitative improvements in process economics make the final 4-selenomethoxynaphthalene product more competitive in the global market, allowing suppliers to offer better pricing structures to their clients without compromising margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as PIFA and dichloromethane ensures a robust supply chain that is less prone to disruptions. Unlike specialized metal catalysts which may have long lead times or limited suppliers, the key components of this reaction are standard chemicals found in most industrial inventories. This accessibility reduces the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical customers. Additionally, the simplicity of the reaction conditions means that the process can be easily transferred between different manufacturing sites or scaled up without requiring specialized equipment, further strengthening supply continuity.
- Scalability and Environmental Compliance: The ambient temperature operation and metal-free nature of this synthesis align perfectly with modern environmental regulations and scalability requirements. The absence of heavy metal waste simplifies effluent treatment and reduces the environmental burden of the manufacturing process, facilitating easier compliance with strict discharge standards. The straightforward reaction setup allows for seamless scale-up from laboratory grams to multi-ton commercial production without encountering the heat transfer or mixing issues often associated with exothermic metal-catalyzed reactions. This scalability ensures that the technology can meet growing market demand for selenium-containing intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific technical advantages and operational parameters detailed in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers provided reflect the objective data available in the public patent record, ensuring transparency and accuracy for all stakeholders involved in the decision-making process.
Q: What are the advantages of using PIFA over traditional metal catalysts for selenization?
A: Using PIFA eliminates the need for expensive transition metal catalysts like silver or copper, thereby removing the requirement for costly heavy metal removal steps and reducing environmental waste.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the reaction operates at room temperature in common solvents like DCM with simple workup procedures, making it highly suitable for commercial scale-up without specialized high-pressure or high-temperature equipment.
Q: What is the regioselectivity of this selenization reaction?
A: The method achieves high regioselectivity for the 4-position of the 1-methoxynaphthalene skeleton, minimizing the formation of isomeric impurities and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Selenomethoxynaphthalene Supplier
The technical potential of this PIFA-mediated selenization route is immense, offering a pathway to high-value selenium intermediates that are essential for next-generation pharmaceuticals and materials. NINGBO INNO PHARMCHEM stands ready to leverage this innovation, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required by global regulatory bodies. We understand the critical nature of supply chain continuity for our partners and have optimized our processes to deliver consistent quality and reliability. By integrating this advanced synthesis method into our portfolio, we provide our clients with a secure source of high-purity 4-selenomethoxynaphthalene that supports their R&D and commercial manufacturing goals.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this metal-free route for your supply chain. Our experts are available to provide specific COA data and route feasibility assessments to help you make informed decisions. Partnering with us ensures access to cutting-edge chemical technologies backed by a reliable and experienced manufacturing partner dedicated to your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
