Scalable Manufacturing of 4-Hydroxy-2-Methoxybenzaldehyde via Green Ionic Liquid Catalysis
Scalable Manufacturing of 4-Hydroxy-2-Methoxybenzaldehyde via Green Ionic Liquid Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign pathways for synthesizing critical intermediates such as 4-hydroxy-2-methoxybenzaldehyde (CAS 18278-34-7). A pivotal advancement in this domain is detailed in patent CN102557903B, which outlines a novel preparation method that fundamentally shifts the paradigm from traditional corrosive acid catalysis to a greener, ionic liquid-mediated process. This technology addresses the growing demand for high-purity pharmaceutical intermediates while simultaneously mitigating the environmental hazards associated with conventional esterification and formylation reactions. By leveraging the unique solvation properties and catalytic activity of ionic liquids, this method achieves exceptional selectivity and yield under mild conditions, positioning it as a superior alternative for industrial-scale production. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and ensuring the consistent quality of downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of protected phenolic intermediates has relied heavily on the use of strong inorganic acids, such as concentrated sulfuric acid or solid super-acids, to drive esterification reactions. These traditional catalysts present significant engineering challenges, primarily due to their highly corrosive nature which necessitates the use of expensive, specialized reactor materials like glass-lined steel or Hastelloy to prevent equipment failure. Furthermore, the post-reaction workup in conventional processes is notoriously difficult, often requiring extensive neutralization steps that generate large volumes of saline wastewater, thereby increasing the burden on effluent treatment facilities and escalating operational costs. The harsh reaction conditions associated with these mineral acids can also lead to side reactions, such as ether cleavage or polymerization, which compromise the purity of the intermediate and reduce the overall molar yield, typically capping efficiency below 80% in many legacy processes. Additionally, the disposal of spent acid catalysts poses a severe environmental liability, conflicting with modern sustainability mandates and increasing the regulatory compliance overhead for manufacturing sites.
The Novel Approach
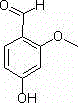
In stark contrast, the methodology described in patent CN102557903B introduces a transformative two-step sequence that utilizes non-toxic ionic liquids, specifically 1-butyl-3-methylimidazolium acetate or bromide salts, as the primary catalyst for the initial esterification. This innovative approach operates under significantly milder thermal conditions, typically between 50°C and 80°C, which not only preserves the integrity of the sensitive aromatic substrate but also drastically reduces energy consumption compared to high-temperature acid catalysis. The use of ionic liquids eliminates the generation of hazardous acidic waste streams, as the catalyst system produces water as the sole byproduct during esterification, and the ionic liquid itself can potentially be recovered and recycled, aligning perfectly with green chemistry principles. Following the protection step, the process employs a Vilsmeier-Haack formylation using phosphorus oxychloride and DMF, cleverly utilizing the subsequent aqueous quench and mild heating (50-55°C) to simultaneously hydrolyze the acetyl protecting group, thereby streamlining the synthesis into a highly efficient telescoped operation. This seamless integration of protection, formylation, and deprotection results in a final product purity exceeding 99.6% with molar yields consistently surpassing 90%, demonstrating a clear technological superiority over legacy methods.
Mechanistic Insights into Ionic Liquid-Catalyzed Esterification and Vilsmeier Formylation
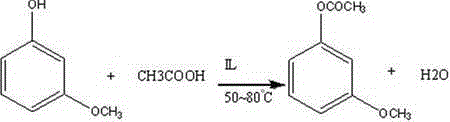
The efficacy of this synthetic route lies in the dual functionality of the ionic liquid catalyst and the strategic manipulation of electronic effects during the formylation stage. In the first step, the ionic liquid, acting as a Lewis base or through hydrogen bonding interactions, activates the carbonyl group of the acetic anhydride or acetic acid, facilitating a nucleophilic attack by the phenolic hydroxyl group of m-hydroxyanisole. This mechanism avoids the protonation of the aromatic ring that typically occurs with strong Brønsted acids, thereby preventing unwanted electrophilic aromatic substitution side reactions and ensuring that the esterification proceeds with high chemoselectivity. The mild acidity of the ionic liquid environment ensures that the methoxy group remains stable, preserving the substitution pattern required for the subsequent regioselective formylation. This precise control over the reaction microenvironment is critical for maintaining the structural fidelity of the intermediate, which is paramount for its application in solid-phase protein synthesis and acid-sensitive resin linking.
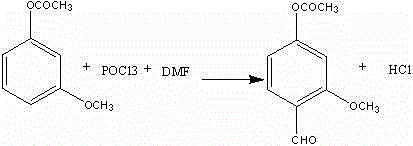
Following the formation of 3-acetoxyanisole, the introduction of the aldehyde moiety is achieved through a classic Vilsmeier-Haack reaction, where the interplay between steric hindrance and electronic activation dictates the outcome. The acetoxy group installed in the first step serves a dual purpose: it protects the phenol from oxidation and, more importantly, exerts a steric influence that directs the incoming electrophilic iminium species to the position ortho to the methoxy group. The reaction is conducted at low temperatures, ranging from -8°C to -2°C, to control the exothermicity and prevent the decomposition of the reactive Vilsmeier complex. Upon completion, the reaction mixture is quenched in water, where the hydrolysis of the iminium salt releases the aldehyde. Crucially, the process leverages the acidic conditions generated during this hydrolysis, combined with a controlled temperature ramp to 50-55°C, to cleave the acetyl protecting group in situ. This tandem hydrolysis-deprotection strategy eliminates the need for a separate saponification step, reducing solvent usage and processing time while delivering the target 4-hydroxy-2-methoxybenzaldehyde with exceptional purity.
How to Synthesize 4-Hydroxy-2-Methoxybenzaldehyde Efficiently
The implementation of this patented protocol requires careful attention to stoichiometry and temperature control to maximize the benefits of the ionic liquid catalyst and the Vilsmeier reagent system. The process begins with the dissolution of m-hydroxyanisole in anhydrous acetic acid, followed by the addition of the ionic liquid catalyst in a weight ratio of approximately 1:0.08 to 0.1 relative to the substrate. Maintaining the reaction temperature between 50°C and 80°C ensures complete conversion to 3-acetoxyanisole, which is then isolated via reduced-pressure distillation to remove excess acetic acid and water. The subsequent formylation step demands strict thermal management, with the addition of phosphorus oxychloride to a mixture of the ester and DMF kept strictly between -8°C and -2°C to stabilize the chloroiminium ion intermediate. Detailed standardized operating procedures regarding mixing rates, quenching protocols, and phase separation techniques are critical for reproducing the high yields reported in the patent literature.
- Perform esterification of m-hydroxyanisole with acetic anhydride using an ionic liquid catalyst at 50-80°C to obtain 3-acetoxyanisole.
- Conduct Vilsmeier reaction on 3-acetoxyanisole using POCl3 and DMF at -8 to -2°C to introduce the aldehyde group.
- Quench the reaction mixture in water and heat to 50-55°C to simultaneously hydrolyze the acetyl group and isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-based synthesis offers profound strategic advantages that extend beyond mere chemical yield. The elimination of corrosive mineral acids from the esterification step translates directly into substantial capital expenditure savings, as manufacturing facilities can utilize standard stainless steel reactors rather than investing in costly corrosion-resistant alloys or glass-lined vessels. This reduction in equipment specification requirements lowers the barrier to entry for contract manufacturing organizations and allows for more flexible allocation of production assets across different product lines. Furthermore, the simplified waste profile, characterized by the absence of spent sulfuric acid and reduced saline effluent, significantly decreases the operational costs associated with environmental compliance and waste disposal, contributing to a leaner and more sustainable cost structure. The high atom economy and molar yields exceeding 90% ensure that raw material utilization is optimized, minimizing the volume of starting materials required per kilogram of finished product and insulating the supply chain against volatility in precursor pricing.
- Cost Reduction in Manufacturing: The replacement of traditional acid catalysts with recyclable ionic liquids removes the need for expensive neutralization agents and reduces the volume of hazardous waste requiring treatment. By avoiding the use of corrosive reagents, the lifespan of production equipment is extended, leading to lower maintenance frequencies and reduced downtime for reactor repairs. The telescoped nature of the second step, which combines formylation and deprotection, further reduces solvent consumption and energy usage by eliminating a discrete hydrolysis unit operation. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The mild reaction conditions and robust nature of the ionic liquid catalyst system enhance the reproducibility of the manufacturing process, reducing the risk of batch failures that can disrupt supply continuity. The use of readily available starting materials such as m-hydroxyanisole and acetic anhydride ensures that the supply chain is not dependent on exotic or single-source reagents that might be subject to geopolitical or logistical constraints. The high purity of the crude product obtained through this method simplifies downstream purification, shortening the overall production cycle time and enabling faster turnaround for customer orders. This reliability is crucial for pharmaceutical clients who require consistent quality and on-time delivery to meet their own regulatory filing and production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to perform effectively from laboratory bench scale to multi-ton commercial production without loss of yield or selectivity. The green chemistry credentials of the method, particularly the reduction in hazardous waste generation and the potential for catalyst recycling, align with increasingly stringent global environmental regulations, future-proofing the manufacturing site against evolving compliance standards. The ability to produce 4-hydroxy-2-methoxybenzaldehyde with purity levels exceeding 99.6% without extensive chromatographic purification supports the economic viability of large-scale operations. This scalability ensures that the supply chain can rapidly respond to surges in demand from the API sector without compromising on quality or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 4-hydroxy-2-methoxybenzaldehyde, derived directly from the proprietary data and experimental results outlined in the relevant patent literature. These insights are intended to clarify the operational parameters and quality benchmarks associated with this advanced manufacturing route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: What are the advantages of using ionic liquids over sulfuric acid in this synthesis?
A: Ionic liquids replace corrosive mineral acids like sulfuric acid, significantly reducing equipment corrosion and waste acid treatment costs while maintaining high yields above 90%.
Q: How is the regioselectivity of the aldehyde group controlled?
A: The process utilizes the steric effect of the acetoxy protecting group and the electronic activation of the methoxy group during the Vilsmeier reaction to ensure ortho-formylation.
Q: What is the purity level achievable with this patented method?
A: The patented method consistently achieves a final product purity of 99.6% to 99.7% with a molar yield exceeding 90%, suitable for high-grade pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxy-2-Methoxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering 4-hydroxy-2-methoxybenzaldehyde with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical applications. Our facility is equipped to handle the specific handling requirements of ionic liquid chemistries and Vilsmeier reactions, guaranteeing a supply of material that is both chemically pure and consistent in its physical properties.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized synthesis can enhance your supply chain resilience. Whether you are developing a new API or optimizing an existing process, our expertise in green chemistry and scalable manufacturing positions us as the ideal partner for your long-term success. Contact us today to discuss how we can support your project with reliable, high-performance chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
