Advanced Manufacturing of Tricyclic Lactam Compounds for Commercial Btk Inhibitor Production
Advanced Manufacturing of Tricyclic Lactam Compounds for Commercial Btk Inhibitor Production
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as core structures for kinase inhibitors. Patent CN107108604B introduces a highly efficient method for preparing tricyclic lactam compounds of formula I, which serve as critical intermediates in the synthesis of Bruton's tyrosine kinase (Btk) inhibitors. These inhibitors are pivotal in treating a wide array of inflammatory, immunological, and oncological diseases, including rheumatoid arthritis and B-cell malignancies. The disclosed process represents a significant technological leap, offering a streamlined pathway that enhances both chemical efficiency and commercial viability. By leveraging specific oxidation and cyclization strategies, this method addresses common bottlenecks in heterocycle synthesis, such as low yields and difficult purification, thereby positioning it as a preferred route for industrial adoption. The strategic design of this synthesis ensures that the resulting intermediates possess the necessary structural integrity and purity required for downstream drug development, making it an invaluable asset for any organization focused on high-purity pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing tricyclic lactam scaffolds often suffer from significant inefficiencies that hinder large-scale production. Conventional methods frequently rely on expensive transition metal catalysts or harsh reaction conditions that can compromise the stability of sensitive functional groups within the molecule. These limitations often result in the formation of complex impurity profiles, necessitating rigorous and costly purification steps such as preparative HPLC or multiple recrystallizations. Furthermore, the use of precious metals introduces supply chain vulnerabilities and environmental compliance challenges, as residual metal removal becomes a critical quality attribute that must be strictly monitored. The cumulative effect of these drawbacks is a substantial increase in the cost of goods sold (COGS) and extended lead times, which are unacceptable in the fast-paced environment of generic and proprietary drug manufacturing. Additionally, older methodologies may lack the stereochemical control required for modern drug candidates, leading to racemic mixtures that require further resolution, further diminishing overall process efficiency and yield.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN107108604B utilizes a metal-free cyclization strategy that dramatically simplifies the synthetic sequence. By employing readily available starting materials such as 2-carbonyl cyclic amine compounds and 1,3-keto-lactam compounds, the process achieves high conversion rates under mild conditions. The use of secondary amine reagents like pyrrolidine facilitates the formation of key enamine intermediates, driving the cyclization forward with exceptional selectivity. This method eliminates the need for expensive catalysts, thereby reducing raw material costs and simplifying the workup procedure. The robustness of this chemistry is evidenced by the successful kilogram-scale examples provided in the patent, demonstrating its readiness for commercial scale-up of complex pharmaceutical intermediates. Moreover, the strategic use of protecting groups such as Boc allows for precise control over reactivity, ensuring that the final tricyclic lactam structure is formed with high fidelity and minimal by-product formation, ultimately delivering a superior product profile for downstream applications.
Mechanistic Insights into Cyclization and Oxidation Processes
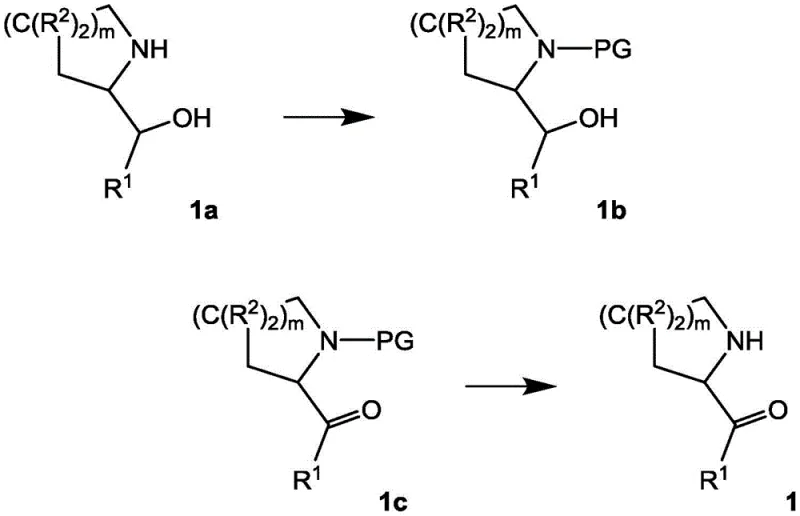
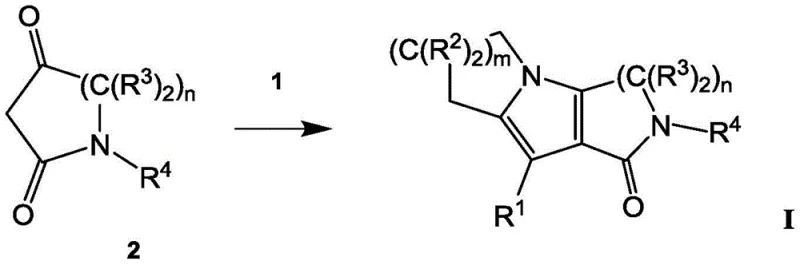
The core of this synthetic innovation lies in the meticulous control of reaction mechanisms, particularly during the oxidation and cyclization phases. The preparation of Intermediate 1 involves a selective oxidation of an alcohol precursor, often utilizing TEMPO-mediated oxidation systems which are known for their high chemoselectivity. This ensures that only the primary alcohol is oxidized to the aldehyde without affecting other sensitive moieties within the cyclic amine structure. Following this, the cyclization step involves a condensation reaction between the aldehyde Intermediate 1 and the 1,3-dicarbonyl Intermediate 2. The presence of a secondary amine catalyst promotes the formation of an enamine species from the 1,3-dicarbonyl compound, which then acts as a nucleophile attacking the electrophilic aldehyde carbon. This cascade reaction leads to the formation of the pyrrole ring fused to the existing cyclic systems, constructing the tricyclic core in a single operational step. The mechanistic pathway is designed to minimize side reactions such as polymerization or over-oxidation, which are common pitfalls in similar heterocyclic syntheses.
Impurity control is intrinsically built into the reaction design through the use of specific protecting groups and controlled addition rates. For instance, the use of tert-butoxycarbonyl (Boc) protection on the nitrogen atoms prevents unwanted N-alkylation or side reactions during the cyclization phase. The patent describes specific temperature controls, such as maintaining reactions between 0°C and 5°C during reagent addition, which is critical for managing exotherms and preventing thermal degradation of intermediates. Furthermore, the final deprotection step using trifluoroacetic acid (TFA) is optimized to remove the Boc group cleanly without inducing hydrolysis of the lactam ring. This level of control ensures that the final product, such as 3,4,6,7,8,9-hexahydropyrido[3,4-b]indolizin-1(2H)-one, meets stringent purity specifications required for pharmaceutical applications. The ability to isolate intermediates as stable salts, such as hydrochlorides, further enhances the purity profile by allowing for crystallization-based purification, effectively removing organic impurities that might persist in oil forms.
How to Synthesize 3,4,6,7,8,9-hexahydropyrido[3,4-b]indolizin-1(2H)-one Efficiently
The synthesis of this high-purity Btk inhibitor intermediate follows a logical sequence designed for maximum efficiency and yield. The process begins with the preparation of the key aldehyde intermediate via protection and oxidation, followed by the crucial cyclization step with a diketone precursor. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and adherence to quality standards. This route is particularly advantageous for manufacturers looking to reduce lead time for high-purity pharmaceutical intermediates, as it avoids lengthy chromatographic purifications in favor of crystallization and extraction techniques. The scalability of each step has been validated through kilogram-scale experiments, confirming that the reaction kinetics and heat transfer properties remain favorable even at larger volumes. By following this protocol, production teams can achieve consistent batch-to-batch quality, which is essential for regulatory compliance and supply chain reliability in the global pharmaceutical market.
- Prepare Intermediate 1 by protecting and oxidizing the corresponding alcohol precursor.
- React Intermediate 1 with 1,3-keto-lactam Intermediate 2 in the presence of a secondary amine.
- Perform deprotection using acidic reagents to yield the final tricyclic lactam compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly impact the bottom line and supply chain resilience. The elimination of transition metal catalysts not only reduces raw material costs but also simplifies the regulatory filing process by removing the need for extensive residual metal testing and clearance protocols. This translates to significant cost savings in manufacturing, as the infrastructure required for metal scavenging is rendered unnecessary. Furthermore, the use of commodity chemicals like pyrrolidine and Boc anhydride ensures a stable and reliable supply of reagents, mitigating the risk of production delays caused by specialized reagent shortages. The high yields reported in the patent examples indicate a material-efficient process, reducing waste disposal costs and improving the overall environmental footprint of the manufacturing operation. These factors combined make this technology a strategic asset for procurement managers seeking cost reduction in API manufacturing without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive, commercially available reagents and avoiding the use of precious metal catalysts. This reduction in material costs is compounded by the high yields achieved at each step, which minimizes the loss of valuable starting materials. Additionally, the simplified workup procedures reduce the consumption of solvents and energy, further driving down operational expenses. The ability to isolate intermediates as stable salts also reduces storage and handling costs, contributing to a leaner and more cost-effective production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: By relying on robust chemistry with widely available starting materials, this method significantly enhances supply chain reliability. The independence from specialized catalysts means that production is less susceptible to geopolitical or market fluctuations affecting rare earth or precious metal supplies. The scalability of the process ensures that supply can be rapidly ramped up to meet demand surges, providing a competitive edge in meeting tight project deadlines. This reliability is crucial for maintaining continuous production schedules and ensuring that downstream drug development programs are not delayed by intermediate shortages.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for scalability, with examples demonstrating successful execution on a multi-kilogram scale. The absence of heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The use of standard organic solvents and reagents facilitates easier recycling and recovery, aligning with green chemistry principles. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile, which is becoming an increasingly important factor in supplier selection processes for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent data to provide accurate and actionable insights for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows. The clarity provided here aims to eliminate uncertainties regarding yield, purity, and scalability, enabling stakeholders to make informed decisions about adopting this advanced synthetic route for their specific project needs.
Q: What are the key advantages of this synthesis method over conventional routes?
A: This method utilizes cheaper reagents and provides higher yields in a more cost-effective manner, specifically avoiding complex purification steps associated with transition metal catalysts.
Q: Is the process scalable for commercial production of Btk inhibitor intermediates?
A: Yes, the patent explicitly describes the use of reagents and conditions suitable for commercial scale production, including kilogram-scale examples with robust isolation procedures.
Q: How is impurity control managed during the cyclization step?
A: Impurity control is achieved through the use of specific protecting groups like Boc and precise temperature control during the addition of reagents, ensuring high purity of the final tricyclic lactam.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,6,7,8,9-hexahydropyrido[3,4-b]indolizin-1(2H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to guarantee that every batch of 3,4,6,7,8,9-hexahydropyrido[3,4-b]indolizin-1(2H)-one meets the highest industry standards. Our expertise in process optimization allows us to identify and mitigate potential scale-up risks early, securing your supply chain and accelerating your time to market.
We invite you to collaborate with us to leverage this advanced technology for your Btk inhibitor programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success. Let us be your partner in achieving chemical excellence and supply chain efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
