Advanced Bispyrene-Doped Extended Porphyrins for Next-Generation Optoelectronics and Chiral Catalysis
Advanced Bispyrene-Doped Extended Porphyrins for Next-Generation Optoelectronics and Chiral Catalysis
The landscape of functional organic materials is undergoing a significant transformation with the introduction of novel macrocyclic architectures described in patent CN115260205A. This groundbreaking intellectual property discloses a class of bispyrene-doped extended porphyrins and their corresponding dipalladium metal complexes, which represent a substantial leap forward in the design of chiral organic molecules. Unlike traditional porphyrins that often struggle with conformational flexibility and modest chiroptical responses, these new compounds exhibit exceptional stability and remarkably high absorption asymmetry factors. The strategic incorporation of pyrene units, known for their outstanding fluorescence and charge transport properties, into the expanded porphyrin framework creates a synergistic effect that enhances both optical and electronic performance. For R&D directors and procurement specialists in the electronic chemicals sector, this technology offers a reliable pathway to producing high-purity OLED materials and advanced chiral catalysts that were previously difficult to synthesize with consistent quality.
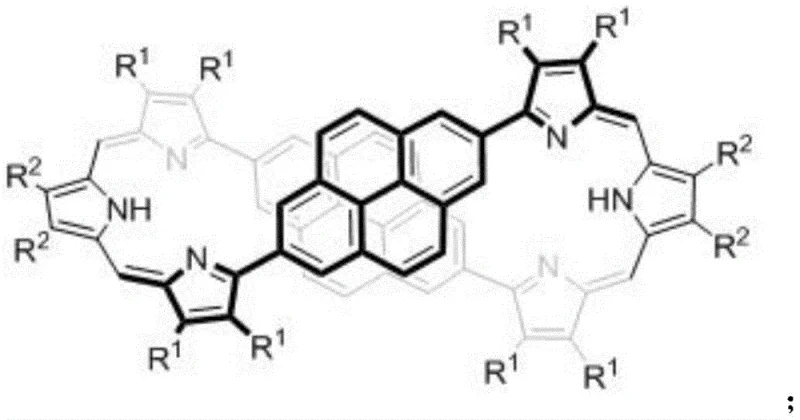
The limitations of conventional methods for synthesizing chiral macrocycles often stem from the inherent difficulty in controlling stereochemistry during the ring-closing steps. Traditional approaches frequently rely on complex chiral auxiliaries or resolution techniques that result in low overall yields and significant material waste. Furthermore, standard porphyrin derivatives typically display absorption asymmetry factors in the range of 10^-4 to 10^-3, which limits their efficacy in high-performance applications such as circularly polarized luminescence devices or sensitive chiral sensors. The novel approach detailed in this patent overcomes these hurdles by utilizing a modular synthetic strategy that builds the chiral environment directly into the rigidified metal-complexed structure. By doping the extended porphyrin skeleton with bispyrene motifs, the inventors have created a system where the steric bulk and electronic properties of the pyrene units pre-organize the molecule, facilitating the formation of stable figure-eight conformations that possess intrinsic chirality.
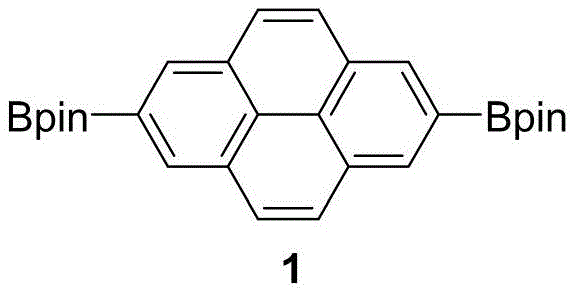
The mechanistic insights into the synthesis of these advanced materials reveal a sophisticated sequence of transition-metal catalyzed transformations designed for maximum efficiency and scalability. The process initiates with an iridium-catalyzed C-H borylation of pyrene, a critical step that functionalizes the inert aromatic core with high regioselectivity to produce the 2,7-diboronate intermediate. This intermediate then undergoes a robust Suzuki-Miyaura cross-coupling reaction with iodopyrrole derivatives, leveraging the versatility of palladium catalysis to form carbon-carbon bonds under relatively mild conditions. Following this, a high-temperature decarboxylation step removes ester protecting groups to reveal the reactive pyrrole units necessary for macrocyclization. The final ring closure is achieved through an acid-catalyzed MacDonald condensation, followed by oxidative aromatization using DDQ, which locks the expanded conjugated system into place.
Subsequent coordination with palladium acetate serves a dual purpose: it not only stabilizes the macrocyclic structure but also induces a rigidification that allows for the separation of enantiomers. The resulting dipalladium complexes, designated as Expr-Pd-1 and Expr-Pd-2, exhibit distinct coordination modes that significantly amplify the chiral optical response, pushing the asymmetry factor to the order of 10^-2. This level of chiral amplification is crucial for applications requiring strong light-matter interactions, such as in next-generation display technologies or asymmetric photocatalysis. The ability to tune the electronic properties through the pyrene dopants while maintaining high chiral integrity makes this class of compounds uniquely suited for integration into complex optoelectronic devices where both charge mobility and polarization control are paramount.
How to Synthesize Bispyrene Extended Porphyrin Efficiently
The synthesis of these high-value electronic chemicals follows a logical progression of well-established organic reactions adapted for macrocyclic construction. The protocol begins with the activation of the pyrene core, followed by the assembly of the pyrrole-pyrene hybrid building blocks. The critical macrocyclization step requires precise control of acid concentration and oxidation potential to ensure the formation of the desired expanded porphyrin rather than linear oligomers. Once the free base porphyrin is obtained, the metallation step with palladium is performed under ambient conditions, yielding the final chiral complexes as stable black solids. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Perform iridium-catalyzed C-H borylation of pyrene with bis(pinacolato)diboron to generate 2,7-diboronate pyrene intermediate.
- Execute Suzuki-Miyaura coupling between the diboronate intermediate and iodopyrrole derivatives followed by alkaline hydrolysis and decarboxylation.
- Conduct acid-catalyzed MacDonald condensation with pyrrole dialdehyde, oxidize with DDQ, and coordinate with Pd(OAc)2 to form the final chiral metal complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic route offers substantial strategic benefits compared to sourcing similar chiral macrocycles from limited specialty suppliers. The reliance on commodity chemicals such as pyrene and standard pyrrole derivatives ensures that raw material availability is high and price volatility is low, mitigating risks associated with supply chain disruptions. Furthermore, the synthetic steps utilize common catalysts like palladium and iridium which, while precious, are used in catalytic amounts and can often be recovered, thereby optimizing the cost structure of the manufacturing process. The elimination of extremely low-temperature conditions or exotic reagents simplifies the engineering requirements for production facilities, allowing for easier technology transfer from laboratory to pilot plant scales.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway significantly reduces the number of purification steps required compared to traditional chiral resolution methods. By achieving chiral resolution through metal complexation rather than preparative chiral HPLC of the free base, the process avoids the high solvent consumption and low throughput associated with chromatographic separations. This operational efficiency translates directly into lower production costs per gram, making high-performance chiral materials more accessible for commercial applications in the display and sensor industries without compromising on purity specifications.
- Enhanced Supply Chain Reliability: The use of robust reaction conditions, such as the Suzuki coupling at 90°C and decarboxylation at 190°C, ensures high conversion rates and minimizes the formation of hard-to-remove byproducts. This reliability in reaction performance leads to consistent batch-to-batch quality, which is critical for long-term supply agreements with electronics manufacturers. Additionally, the intermediates generated during the synthesis are stable solids that can be isolated and stored, providing flexibility in production scheduling and inventory management to meet fluctuating market demands.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like dichloromethane and DMF which are standard in industrial organic synthesis and have established recovery protocols. The high yields reported in the patent examples, particularly in the metal complexation step which reaches approximately 85%, indicate a material-efficient process that generates less chemical waste. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the burden on waste treatment facilities and lowering the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bispyrene-doped extended porphyrins. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating these materials into your specific product development pipelines.
Q: What is the primary advantage of bispyrene-doped extended porphyrins over conventional porphyrins?
A: The primary advantage lies in their significantly enhanced chiral optical properties, specifically achieving absorption asymmetry factors (gabs) up to the order of 10^-2, which is substantially higher than the typical 10^-4 to 10^-3 range found in conventional helical molecules. Additionally, they exhibit strong absorption in the near-infrared II (NIR-II) region.
Q: How does the palladium coordination facilitate chiral resolution in this synthesis?
A: The coordination of Pd(II) with the extended porphyrin framework creates rigid dipalladium metal complexes (Expr-Pd-1 and Expr-Pd-2) with distinct coordination modes. This rigidity and the specific metal-ligand interaction stabilize the chiral conformation, allowing for effective resolution of enantiomers which is otherwise difficult due to the conformational flexibility of the free base porphyrin.
Q: What are the key reaction conditions for the macrocyclization step?
A: The macrocyclization involves an acid-catalyzed condensation between 2,7-dipyrrolylpyrene and pyrrole dialdehyde in dichloromethane at room temperature, followed by oxidation using 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) under aerobic conditions to aromatize the expanded porphyrin ring system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bispyrene Extended Porphyrin Supplier
As the demand for advanced optoelectronic materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex macrocyclic synthesis is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of chiral catalyst or OLED material meets the exacting standards required by global technology leaders. We understand the critical nature of supply continuity in the electronics sector and have optimized our logistics to support just-in-time delivery models.
We invite you to collaborate with our technical team to explore how this patented technology can be adapted to your specific application needs. Whether you require a Customized Cost-Saving Analysis for large-volume procurement or need to evaluate the compatibility of these porphyrins with your existing device architectures, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your innovation cycle with reliable, high-performance chemical solutions.
