Industrial Scale Synthesis of Levorotatory Betaxolol Hydrochloride for Global Pharma Supply Chains
The pharmaceutical landscape for ophthalmic treatments demands rigorous standards for chiral purity and manufacturing safety, particularly for beta-blockers used in glaucoma management. Patent CN101012175A introduces a transformative synthesis technique for levorotatory betaxolol hydrochloride that addresses critical inefficiencies found in legacy production methods. This innovative route utilizes p-hydroxyphenethylol and R-epoxyhalopropane as primary starting materials, establishing a robust foundation for high-yield chiral synthesis. By strategically integrating alkylation, amination, and protection steps, the process ensures exceptional stereochemical control without relying on hazardous pyrophoric reagents. For R&D directors and procurement specialists, this technology represents a significant leap forward in producing reliable ophthalmic drug intermediates with minimized environmental impact. The methodology not only enhances product quality but also streamlines the supply chain by reducing reaction times and simplifying purification protocols. As the global demand for high-purity glaucoma medications rises, adopting this advanced synthetic pathway offers a competitive edge in both regulatory compliance and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of levorotatory betaxolol has been plagued by significant operational hazards and economic inefficiencies that hinder large-scale commercialization. Traditional routes often depend on dangerous reagents such as diethylzinc and diiodomethane, which are not only expensive but also pose severe safety risks due to their pyrophoric nature and sensitivity to moisture. These legacy methods typically require stringent anhydrous conditions and nitrogen protection throughout multiple steps, drastically increasing infrastructure costs and operational complexity. Furthermore, prior art processes frequently suffer from low yields in key transformation steps, leading to substantial material loss and inflated production costs. The extended reaction times associated with these older techniques, sometimes exceeding fifty hours for single steps, create bottlenecks that limit manufacturing throughput and extend lead times for API suppliers. Additionally, the use of toxic catalysts necessitates complex downstream processing to remove heavy metal residues, complicating regulatory approval and waste management. These cumulative drawbacks make conventional synthesis routes economically unviable for modern, high-volume pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data offers a streamlined and safer alternative that eliminates the need for hazardous organometallic reagents. By shifting to a chemistry based on stable alkali bases and standard organic solvents, the new method significantly reduces operational risks and simplifies equipment requirements. The process achieves high conversion rates through optimized reaction conditions, utilizing readily available raw materials that ensure supply chain continuity. This modern synthesis strategy effectively bypasses the need for complex chiral resolution by introducing chirality early via R-epoxyhalopropane, thereby preserving optical purity throughout the sequence. The elimination of toxic heavy metals also simplifies the purification workflow, allowing for easier compliance with stringent international pharmaceutical standards. Moreover, the reduced reaction times and improved yields contribute to a more agile manufacturing process capable of responding quickly to market demands. This approach not only enhances safety but also delivers substantial economic benefits through reduced waste and lower raw material consumption.
Mechanistic Insights into Chiral Alkylation and Oxazoline Protection
The core of this synthesis lies in the precise control of stereochemistry during the initial alkylation and subsequent protection steps. The process begins with the reaction of p-hydroxyphenethylol with R-epoxyhalopropane in the presence of a basic compound, forming a chiral epoxide intermediate with high enantiomeric excess. This step is critical as it sets the stereochemical foundation for the entire molecule, ensuring that the final product meets the rigorous purity requirements for ophthalmic applications. Following this, the epoxide undergoes ring-opening amination with isopropylamine, a reaction that must be carefully controlled to prevent racemization. The resulting amino alcohol is then protected using benzene formonitrile or similar reagents to form an oxazoline ring, which serves to mask the reactive amine and hydroxyl groups during subsequent alkylation. This protection strategy is essential for preventing side reactions and ensuring that the cyclopropylmethyl group is introduced selectively at the phenolic oxygen. The use of phase-transfer catalysts in the alkylation step further enhances reaction efficiency, allowing for milder conditions and faster kinetics. Finally, acidic hydrolysis removes the protecting group to reveal the active pharmaceutical ingredient, which is then converted to the hydrochloride salt for stability.
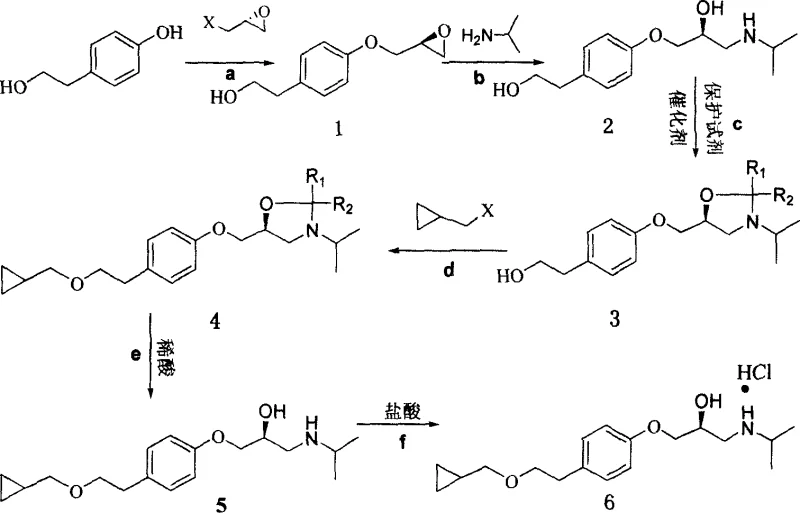
Impurity control is another pivotal aspect of this mechanistic design, ensuring that the final API meets strict pharmacopeial standards. The selection of specific solvents and recrystallization protocols plays a vital role in removing by-products and unreacted starting materials. For instance, the use of ethyl acetate and acetone for recrystallization helps to isolate the intermediate solids with high purity, minimizing the carryover of impurities to subsequent steps. The process also incorporates washing treatments to remove inorganic salts generated during the reaction, ensuring that the final product is free from metal contaminants. By maintaining a closed-loop solvent system where possible, the method reduces the risk of external contamination and ensures consistent batch-to-batch quality. The high optical purity achieved, often exceeding 99% Ee, is a direct result of the chiral integrity maintained during the epoxide formation and the selective nature of the protection-deprotection sequence. This level of control is essential for minimizing side effects in patients and ensuring the therapeutic efficacy of the glaucoma medication.
How to Synthesize Levorotatory Betaxolol Hydrochloride Efficiently
Implementing this synthesis route requires a clear understanding of the sequential chemical transformations and the specific conditions required for each step. The process is designed to be scalable, moving from laboratory benchtop to industrial reactor with minimal modification to the core chemistry. Operators must focus on maintaining precise temperature controls and stoichiometric ratios to maximize yield and purity at each stage. The following guide outlines the critical operational phases, emphasizing the importance of reagent quality and process monitoring. Detailed standard operating procedures should be established to handle the alkylation and protection steps safely, ensuring that all safety protocols are followed. For a comprehensive breakdown of the specific reaction parameters and workup procedures, please refer to the technical guide below.
- Alkylation of p-hydroxyphenethylol with R-epoxyhalopropane to form the chiral epoxide intermediate.
- Ring-opening amination with isopropylamine followed by oxazoline protection of the amino alcohol.
- Final alkylation with cyclopropylmethyl halide, deprotection, and salt formation to yield the hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis technology offers profound advantages for procurement managers and supply chain heads looking to optimize their API sourcing strategies. The elimination of expensive and hazardous reagents translates directly into reduced raw material costs and lower insurance premiums for manufacturing facilities. By utilizing common solvents that can be easily recovered and recycled, the process significantly minimizes waste disposal costs and environmental compliance burdens. The high yields reported in the patent data indicate a more efficient use of starting materials, which reduces the overall cost of goods sold and improves profit margins for manufacturers. Furthermore, the simplified operational requirements mean that production can be scaled up more rapidly without the need for specialized hazardous material handling infrastructure. This flexibility allows suppliers to respond more quickly to fluctuations in market demand, ensuring a steady supply of critical glaucoma medications. The robustness of the process also reduces the risk of batch failures, enhancing supply chain reliability and reducing the need for safety stock.
- Cost Reduction in Manufacturing: The shift away from pyrophoric reagents like diethylzinc eliminates the need for specialized storage and handling equipment, resulting in significant capital expenditure savings. Additionally, the high conversion rates and reduced reaction times lower energy consumption and labor costs per kilogram of product. The ability to recycle solvents further drives down operational expenses, making the process economically superior to traditional methods. These factors combine to create a more cost-effective manufacturing model that can withstand market price pressures.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as p-hydroxyphenethylol and R-epoxyhalopropane ensures that raw material sourcing is not a bottleneck. The simplified process flow reduces the number of unit operations, decreasing the likelihood of equipment downtime or process delays. This streamlined approach allows for more predictable production schedules and shorter lead times for order fulfillment. Suppliers can thus offer more reliable delivery commitments to pharmaceutical clients, strengthening long-term partnerships.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of inorganic salts that can be easily treated or disposed of. The absence of heavy metal catalysts simplifies the regulatory approval process for new drug applications and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles enhances the corporate social responsibility profile of the manufacturer and ensures compliance with increasingly strict environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of levorotatory betaxolol hydrochloride. These answers are derived from the specific technical advantages and operational details outlined in the patent data. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this technology for commercial production. The responses highlight the safety, efficiency, and quality benefits that distinguish this method from conventional approaches.
Q: How does this synthesis route improve optical purity compared to traditional methods?
A: By utilizing R-epoxyhalopropane as the chiral source early in the synthesis and employing specific oxazoline protection strategies, the process maintains stereochemical integrity throughout, achieving Ee values greater than 99% without requiring complex chiral resolution steps.
Q: What are the safety advantages of this manufacturing process?
A: Unlike prior art that utilizes pyrophoric reagents like diethylzinc, this method relies on stable alkali bases and standard organic solvents, significantly reducing fire hazards and operational risks in large-scale production environments.
Q: Is the solvent system suitable for industrial recycling?
A: Yes, the process utilizes common organic solvents such as acetone, toluene, and ethyl acetate, which can be efficiently recovered and reused through standard distillation, minimizing waste discharge and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levorotatory Betaxolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving needs of the global pharmaceutical industry. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN101012175A can be successfully implemented at an industrial level. We are committed to delivering high-purity intermediates and APIs that meet stringent purity specifications through our rigorous QC labs and state-of-the-art manufacturing facilities. Our dedication to quality and safety makes us an ideal partner for companies seeking to optimize their supply chain for ophthalmic drugs. By leveraging our technical expertise, we can help you navigate the complexities of chiral synthesis and bring high-quality medications to market faster.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the viability of this technology for your needs. Together, we can drive innovation and efficiency in the manufacturing of life-saving glaucoma treatments.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
