Advanced Spirochiral Rhodium Complexes for Scalable Asymmetric C-H Activation in Fine Chemicals
The landscape of asymmetric synthesis is undergoing a transformative shift with the emergence of advanced catalytic systems described in patent CN105669766B. This intellectual property introduces a novel class of cyclopentadiene rhodium complexes based on a unique spirochiral skeleton, designed specifically to overcome the limitations of traditional catalysts in C-H bond functionalization. For R&D leaders seeking to optimize the construction of axially chiral biaryl compounds, this technology offers a robust pathway to achieve exceptional enantioselectivity without compromising on yield. The strategic implementation of these spiro-ring based ligands allows for precise stereocontrol during oxidative Heck coupling reactions, particularly when dealing with challenging olefinic substrates that often fail under conventional conditions. By integrating this methodology, pharmaceutical manufacturers can access complex molecular architectures essential for next-generation active pharmaceutical ingredients with greater efficiency and reliability. 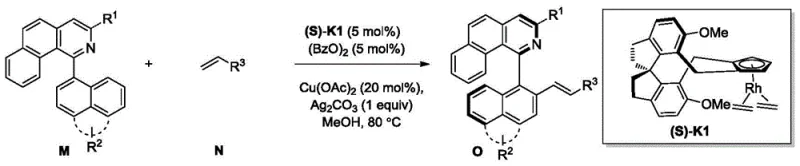
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of rhodium-catalyzed asymmetric C-H bond functionalization has relied heavily on pentamethylcyclopentadiene (Cp*Rh) complexes modified with biotinylated groups or tartaric acid derivatives. While these earlier generations of catalysts demonstrated proof of concept, they frequently suffer from significant drawbacks regarding synthetic complexity and substrate generality. The synthesis of biotinylated catalysts often involves cumbersome bioconjugation steps that are difficult to reproduce on a kilogram scale, leading to batch-to-batch variability that is unacceptable for regulated pharmaceutical manufacturing. Furthermore, the chiral environment provided by these traditional ligands is often too flexible, resulting in moderate enantioselectivity that necessitates costly and time-consuming recrystallization or chromatographic purification steps to meet stringent purity specifications. This inefficiency creates a bottleneck in the supply chain, increasing the cost of goods sold and extending lead times for critical drug intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a rigid spirochiral backbone that locks the catalytic center into a well-defined three-dimensional configuration. This structural rigidity is the key to unlocking superior stereoinduction, allowing the catalyst to differentiate between prochiral faces of the substrate with remarkable precision. The synthesis of these spiro-ligands is streamlined through a series of robust organic transformations, including thermal rearrangements that are inherently scalable and do not require exotic reagents. By eliminating the reliance on fragile biological scaffolds or expensive chiral pool starting materials that are subject to market volatility, this new class of rhodium complexes offers a more sustainable and economically viable solution. The result is a catalytic system that maintains high activity and selectivity across a diverse range of electronic and steric environments, providing process chemists with a versatile tool for late-stage functionalization.
Mechanistic Insights into Spiro-Rhodium Catalyzed Cyclization
The exceptional performance of these catalysts stems from the unique electronic and steric properties imparted by the spiro-diindane skeleton. Upon coordination with the rhodium metal center, the cyclopentadiene ligand forms a stable half-sandwich complex that resists decomposition under oxidative conditions. 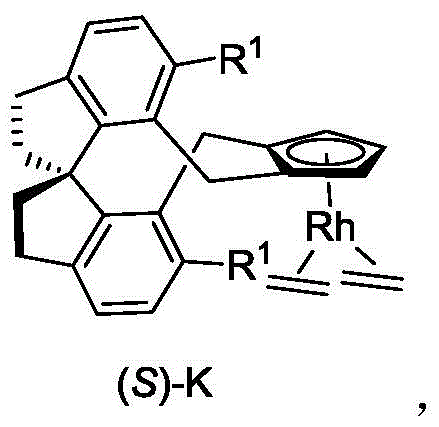 The spiro-junction creates a deep chiral pocket that effectively shields one face of the metal center, forcing the incoming substrate to approach from a specific trajectory. This steric gating mechanism is crucial for controlling the regioselectivity of the C-H activation step, ensuring that the reaction occurs exclusively at the desired position on the aromatic ring. Moreover, the electron-donating or withdrawing nature of the substituents on the spiro-skeleton can be finely tuned to modulate the electrophilicity of the rhodium species, thereby optimizing the reaction rate for different classes of substrates without altering the core catalytic cycle.
The spiro-junction creates a deep chiral pocket that effectively shields one face of the metal center, forcing the incoming substrate to approach from a specific trajectory. This steric gating mechanism is crucial for controlling the regioselectivity of the C-H activation step, ensuring that the reaction occurs exclusively at the desired position on the aromatic ring. Moreover, the electron-donating or withdrawing nature of the substituents on the spiro-skeleton can be finely tuned to modulate the electrophilicity of the rhodium species, thereby optimizing the reaction rate for different classes of substrates without altering the core catalytic cycle.
From an impurity control perspective, the high fidelity of this catalytic system significantly reduces the formation of undesired regioisomers and enantiomers. In traditional processes, the presence of minor stereoisomers can act as potent inhibitors in downstream biological assays or pose toxicity risks, requiring rigorous purification protocols. The spiro-rhodium complex minimizes these risks by driving the reaction towards a single stereoisomer with high enantiomeric excess, simplifying the downstream workup. This reduction in impurity burden not only enhances the safety profile of the final product but also improves the overall mass balance of the process. By reducing the load on purification units such as preparative HPLC or chiral columns, manufacturers can achieve substantial operational savings and increase throughput capacity while maintaining compliance with international quality standards.
How to Synthesize Spirochiral Rhodium Complex Efficiently
The preparation of these high-value catalysts follows a logical and convergent synthetic strategy that is well-suited for industrial adoption. The process begins with the functionalization of a readily available spiro-biindane precursor, which undergoes directed iodination to install reactive handles for subsequent modification. This is followed by a sequence of esterification and selective reduction steps using diisobutylaluminum hydride to generate the necessary alcohol intermediates. The critical step involves a thermal rearrangement that constructs the cyclopentadiene ring directly onto the spiro-skeleton, followed by complexation with a rhodium dimer source. 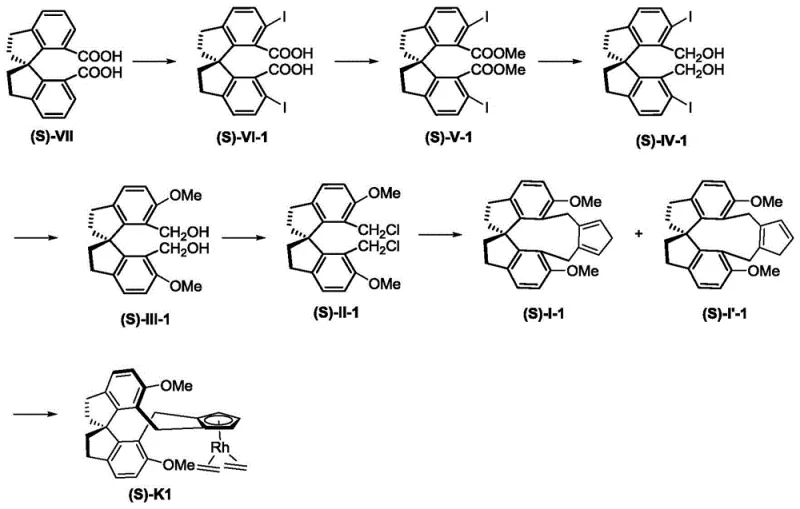 Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Perform iodination of the spiro-biindane skeleton using palladium sources and oxidants to introduce reactive handles.
- Execute esterification and subsequent reduction with DIBAL-H to convert carboxylic acids into primary alcohols.
- Conduct thermal rearrangement and complexation with rhodium precursors to finalize the optically pure catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this spiro-rhodium technology translates into tangible strategic benefits beyond mere technical performance. The streamlined synthesis route reduces the number of unit operations required to produce the catalyst, which directly correlates to lower capital expenditure and reduced operational complexity. By utilizing common organic solvents and avoiding cryogenic conditions for the majority of the steps, the process becomes more energy-efficient and safer to operate in a multipurpose manufacturing facility. This operational simplicity enhances supply chain resilience, as it reduces dependence on specialized equipment or scarce reagents that could become bottlenecks during periods of high demand or global disruption.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers and the reduction in purification steps lead to significant cost optimization in the production of chiral intermediates. Because the catalyst delivers high enantioselectivity directly, the need for costly chiral resolution processes is drastically minimized, lowering the overall cost of goods. Furthermore, the high turnover number of the rhodium complex means that less precious metal is required per kilogram of product, mitigating the financial risk associated with volatile rhodium prices and improving the economic feasibility of the process.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent quality and availability of the catalyst, which is critical for maintaining uninterrupted production schedules. The use of stable, shelf-stable intermediates allows for strategic stockpiling without the risk of rapid degradation, providing a buffer against supply shocks. This reliability empowers procurement teams to negotiate better terms with vendors and plan long-term production campaigns with confidence, knowing that the critical catalytic material will be available when needed to meet commercial deadlines.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents that are easier to recover and recycle, thereby reducing the environmental footprint of the manufacturing operation. The high atom economy of the coupling reaction minimizes waste generation, simplifying waste treatment and disposal compliance. This alignment with sustainability goals not only reduces regulatory burdens but also enhances the corporate social responsibility profile of the supply chain, making it more attractive to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spirochiral rhodium technology in industrial settings. These insights are derived directly from the experimental data and process descriptions within the patent documentation to provide accurate guidance for decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this catalytic system into existing manufacturing workflows and for assessing the potential return on investment.
Q: What is the primary advantage of spirochiral rhodium complexes over conventional Cp*Rh catalysts?
A: The spirochiral skeleton provides a more rigid chiral environment, resulting in significantly higher enantioselectivity and broader substrate tolerance in asymmetric C-H activation reactions compared to traditional biotinylated or tartaric acid-derived ligands.
Q: Are the synthesis conditions for these catalysts suitable for large-scale manufacturing?
A: Yes, the patent describes reaction conditions utilizing common organic solvents and standard temperature ranges, avoiding extreme cryogenic requirements for most steps, which facilitates commercial scale-up and process safety.
Q: How does this technology impact impurity profiles in pharmaceutical intermediates?
A: By achieving high enantiomeric ratios directly during the catalytic step, the need for extensive downstream chiral separation is reduced, leading to a cleaner impurity profile and higher overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirochiral Rhodium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to pilot plant is seamless. We are committed to delivering these complex spiro-rhodium catalysts with stringent purity specifications and support them with our rigorous QC labs to guarantee batch-to-batch consistency. Our infrastructure is designed to handle sensitive organometallic compounds safely, providing you with a secure and reliable source for your most demanding asymmetric synthesis projects.
We invite you to collaborate with us to unlock the full potential of this patented technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our spirochiral rhodium complexes can enhance your process efficiency and reduce your overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
