Advanced Manufacturing of Fluconazole Intermediates: A Safer, Cost-Effective Epoxidation Strategy
Advanced Manufacturing of Fluconazole Intermediates: A Safer, Cost-Effective Epoxidation Strategy
The pharmaceutical industry continuously seeks robust synthetic routes that balance safety, efficiency, and cost, particularly for high-volume antifungal agents like fluconazole. Patent CN1054853C introduces a transformative methodology for preparing the critical intermediate 1-[2-(2,4-difluorophenyl)-2,3-epoxypropyl]-1H-1,2,4-triazole and its mesylate salt. This innovation fundamentally shifts the paradigm from hazardous, anhydrous conditions to a safer, biphasic aqueous system. By replacing pyrophoric sodium hydride with commercially available aqueous sodium hydroxide and eliminating the need for expensive quaternary ammonium phase-transfer catalysts, this process addresses long-standing pain points in fine chemical manufacturing. For R&D directors and procurement managers alike, this patent represents a viable pathway to significantly lower production costs while enhancing operational safety profiles. The technical breakthrough lies in achieving high conversion rates under moderate temperatures, proving that green chemistry principles can be successfully integrated into complex heterocyclic synthesis without compromising yield or purity standards required for GMP environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key epoxy intermediate relied heavily on the classical Corey-Chaykovsky reaction utilizing trimethylsulfoxonium iodide and sodium hydride in dimethyl sulfoxide (DMSO). This traditional approach presents severe logistical and safety challenges for large-scale operations. The requirement for strictly anhydrous conditions necessitates expensive drying protocols for all solvents and reagents, alongside the continuous use of nitrogen blanketing to prevent moisture ingress and potential ignition of the pyrophoric base. Furthermore, the use of sodium hydride introduces significant explosion risks, mandating specialized explosion-proof equipment and rigorous safety training for operators. Another critical drawback identified in prior art, such as United States Patent 4404216, involves the addition of quaternary ammonium salts like cetyltrimethylammonium bromide to facilitate the reaction. While intended to improve kinetics, these surfactants often lead to persistent emulsions during the workup phase, making phase separation extremely difficult and resulting in poor product recovery. Consequently, yields in these older methods often stagnated around 21%, rendering them economically unviable for modern, cost-sensitive supply chains seeking reliable fluconazole intermediate suppliers.
The Novel Approach
The methodology disclosed in CN1054853C offers a decisive break from these inefficiencies by leveraging a biphasic reaction system driven by aqueous sodium hydroxide. This novel approach completely omits the quaternary ammonium salt catalyst, thereby eliminating the emulsification issues that plagued previous iterations. Instead of relying on hazardous sodium hydride, the process utilizes a 10-50% aqueous sodium hydroxide solution, which is not only safer to handle but also drastically reduces raw material costs. The reaction proceeds smoothly in common organic solvents such as toluene, benzene, or cyclohexane, allowing for a clean separation of the organic layer containing the product from the aqueous waste stream. This simplification of the post-treatment process means that solvent recovery is more efficient, and the overall workflow is streamlined. By optimizing the molar ratios of trimethylsulfoxonium iodide to the ketone substrate to between 0.8 and 1.0, the process minimizes reagent waste while maintaining high conversion efficiency. This strategic redesign of the synthetic route demonstrates how cost reduction in pharmaceutical intermediates manufacturing can be achieved through intelligent process engineering rather than mere resource cutting.
Mechanistic Insights into Biphasic Epoxidation Catalysis
At the heart of this synthesis is a modified Corey-Chaykovsky epoxidation mechanism adapted for biphasic conditions. In the absence of a traditional phase-transfer catalyst, the reaction relies on the interfacial interaction between the organic phase containing the ketone substrate and the aqueous phase containing the hydroxide base. The hydroxide ions deprotonate the trimethylsulfoxonium iodide at the interface or within micro-emulsions formed by vigorous stirring, generating the reactive sulfur ylide species in situ. This ylide then attacks the carbonyl carbon of the 2',4'-difluoro-2-(1H-1,2,4-triazole-1-yl)methyl phenyl ketone to form the betaine intermediate, which subsequently collapses to release dimethyl sulfoxide and form the strained epoxide ring. The success of this metal-free catalysis without quaternary salts suggests that the specific solubility characteristics of the triazole-containing substrate facilitate sufficient contact between phases to drive the reaction forward. Understanding this mechanistic nuance is crucial for scaling up, as agitation speed and interfacial area become critical process parameters (CPPs) that directly influence reaction kinetics and final yield.
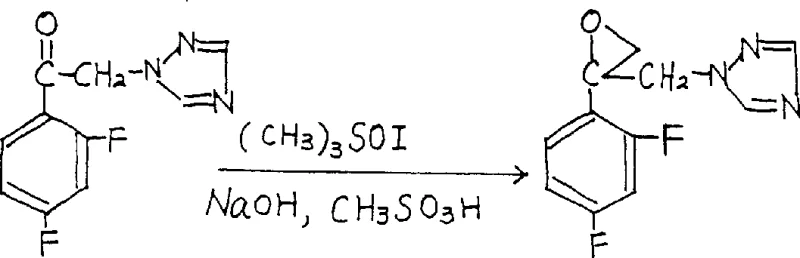
Beyond the primary transformation, impurity control is a paramount concern for any API intermediate supplier. The use of aqueous sodium hydroxide under moderate temperatures (20-100°C) mitigates the risk of thermal degradation or unwanted side reactions that often occur with stronger, non-selective bases at elevated temperatures. In the classical sodium hydride method, trace moisture can lead to hydrolysis of the ylide or the product, generating difficult-to-remove impurities. The new method's tolerance for water inherently stabilizes the reaction environment against such hydrolytic pathways. Furthermore, the elimination of quaternary ammonium salts removes a major source of ionic contamination that can persist through crystallization steps. The final product is isolated as a mesylate salt by adding methanesulfonic acid in ethyl acetate, a step that further purifies the compound by precipitating the desired salt while leaving neutral organic impurities in the mother liquor. This robust purification strategy ensures that the commercial scale-up of complex pharmaceutical intermediates meets the stringent purity specifications required by global regulatory bodies.
How to Synthesize 1-[2-(2,4-difluorophenyl)-2,3-epoxypropyl]-1H-1,2,4-triazole Efficiently
Implementing this synthesis requires precise control over stoichiometry and phase separation dynamics to maximize the reported 60-70% yield. The process begins by charging the reactor with the ketone substrate and a water-immiscible solvent like toluene, followed by the controlled addition of the aqueous base and the sulfonium salt. Maintaining the reaction temperature within the optimal window of 40-60°C ensures rapid ylide formation without decomposing the sensitive epoxide product. Post-reaction, the simplicity of the workup allows for immediate phase separation, where the organic layer is washed and concentrated before salt formation. The detailed standardized operating procedures for this specific route, including exact mixing times and crystallization cooling ramps, are essential for technology transfer teams aiming to replicate these results in a pilot plant setting.
- Prepare the reaction mixture by dissolving 2',4'-difluoro-2-(1H-1,2,4-triazole-1-yl)methyl phenyl ketone in an organic solvent such as toluene or benzene.
- Add aqueous sodium hydroxide solution (10-50% concentration) and trimethylsulfoxonium iodide to the reaction vessel under stirring.
- Maintain the reaction temperature between 20-100°C for 1-6 hours, then separate the organic layer, concentrate, and crystallize with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible economic and operational benefits that extend far beyond the laboratory bench. The most immediate impact is the drastic simplification of the supply chain for raw materials; replacing specialized, hazardous sodium hydride with commodity-grade caustic soda reduces both purchasing complexity and storage costs. Additionally, the removal of expensive quaternary ammonium catalysts, which were previously required in stoichiometric or near-stoichiometric amounts in other methods, directly lowers the bill of materials. The patent explicitly notes a raw material cost reduction of approximately one-third, a figure that significantly improves the margin structure for high-volume production. This cost efficiency is compounded by the improved yield, which jumps from roughly 21% in older catalytic methods to a robust 60-70%, effectively tripling the output per batch for the same input of starting materials. Such improvements in atom economy and throughput are critical for maintaining competitiveness in the generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of pyrophoric reagents and phase-transfer catalysts creates a leaner manufacturing process with lower overhead. By avoiding the need for explosion-proof facilities and strict anhydrous environments, capital expenditure (CAPEX) for new production lines is significantly reduced, while operational expenditure (OPEX) drops due to lower energy consumption for drying and nitrogen purging. The ability to use cheaper solvents like toluene or cyclohexane instead of polar aprotic solvents like DMSO, which are difficult to recover, further enhances the economic viability of the process. These cumulative savings allow suppliers to offer more competitive pricing for high-purity fluconazole intermediates without sacrificing quality.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like aqueous sodium hydroxide and toluene insulates the production schedule from the volatility often seen in the supply of specialized fine chemical reagents. The robustness of the biphasic system means that minor fluctuations in raw material quality have less impact on the final outcome, ensuring consistent batch-to-batch performance. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the risk of batch failures or extended rework cycles. Suppliers can confidently commit to delivery timelines, knowing that the process is forgiving and scalable, thereby strengthening the trust relationship with downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The absence of sodium hydride eliminates the generation of hydrogen gas and the risk of fires, simplifying waste treatment protocols. The biphasic nature of the reaction facilitates easier wastewater management, as the aqueous layer can be treated separately from the organic solvent stream. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures compliance with increasingly stringent global regulations on hazardous waste disposal. The process is inherently designed for commercial scale-up, capable of transitioning seamlessly from 100 kgs to 100 MT annual commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxidation technology. They are derived from the specific advantages outlined in the patent data, focusing on safety, yield, and scalability concerns that typically arise during process validation. Understanding these nuances helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: Why is aqueous sodium hydroxide preferred over sodium hydride in this synthesis?
A: Aqueous sodium hydroxide eliminates the severe safety hazards associated with pyrophoric sodium hydride, removing the need for strict anhydrous conditions and nitrogen blanketing, thereby reducing operational costs and equipment requirements.
Q: How does this method improve yield compared to US Patent 4404216?
A: By omitting the quaternary ammonium salt catalyst which caused emulsification and difficult phase separation in previous methods, this novel approach achieves a significantly higher yield of 60-70% compared to the approximate 21% yield of the prior art.
Q: What are the scalability advantages of this biphasic reaction system?
A: The biphasic system allows for easy separation of the product into the organic layer, simplifying post-treatment and solvent recovery, which is critical for large-scale industrial production ranging from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-[2-(2,4-difluorophenyl)-2,3-epoxypropyl]-1H-1,2,4-triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this safer epoxidation route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of fluconazole intermediate meets the highest international standards. Our commitment to process safety and environmental stewardship aligns perfectly with the green chemistry advantages of this patented method, making us an ideal partner for long-term supply agreements.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of this high-quality intermediate into your antifungal drug production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
